脊髓损伤
-
Figure 1|Characteristics of studies in ClinicalTrials.gov.
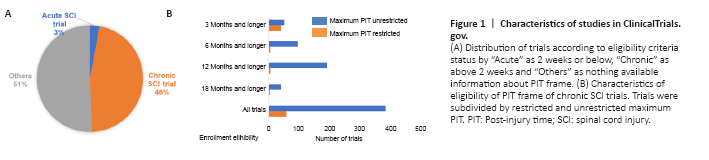
In order to observe how researchers are defining the enrollment criteria for chronic SCI trials, we analyzed 957 trials registered in ClinicalTrials.gov registry. Of these trials, 46% (445) of trials were for chronic SCI, 3% (28) for acute SCI and the remaining 51% of trials were missing or did not restrict the PIT in the eligibility criteria (Figure 1A). Among the chronic SCI trials, we observed 87% (385) that did not limit the maximum PIT while only 13% (60) of trials restricted maximum PIT for their eligibility. The range of maximum PIT was 1 month to 10 years. We also observed higher trends of unrestricted trials in each subcategory (Figure 1B). It means that there is a high probability of participants enrolled with a wide range of PITs.
Figure 2|PRISMA flow diagram of search strategy and study selection.

To understand the range of time-to-enrollment in clinical trials, we performed a literature search which revealed a total of 508 studies (Figure 2). Following inclusion and exclusion criteria evaluation, 19 studies remained and were evaluated for individual patient data after a primary screening of article title and abstracts. Of the 19 studies, nine clinical trials which presented individual patient data were included in the final analysis (Table 1; Grijalva et al., 2010; Lima et al., 2010; Rao et al., 2013; Chen et al., 2014; El-Kheir et al., 2014; Mendon?a et al., 2014; Shin et al., 2015; Hur et al., 2016; Vaquero et al., 2016). Median (range) time-to-enrollment was 18.5 months (0.5–321). Specifically, five studies (Grijalva et al., 2010; Lima et al., 2010; Chen et al., 2014; Mendonca et al., 2014; Vaquero et al., 2016) included participants who had a wide range of time-to-enrollment (Figure 3). This wide variability could potentially affect the outcome of chronic SCI.