周围神经损伤
-
Figure 2|Fibrin-collagen hydrogel biocompatibility.
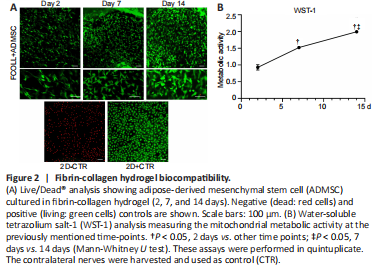
Biocompatibility is one of the most important criteria that should be verified when preparing a biomaterial. The in vitro evaluation of the cultured ADMSC in the fibrin-collagen hydrogel at different time points (2, 7, and 14 days) demonstrated the high biocompatibility of the prepared hydrogel. During the entire time frame monitored starting at 2 days till 14 days, dead cells were completely absent; moreover, an increase in cellular density was observed (Figure 2A), indicating that fibrin-collagen hydrogel supports cell viability and proliferation. This result is in accordance with WST-1 assay that showed a statistically significant increase in metabolic activity at 7 and 14 days compared with 2 days (P < 0.05), and at 7 days compared with 14 days (P < 0.05; Figure 2B).
Figure 3|Histological and biochemical results of ex vivo nerve-derived cell migration.
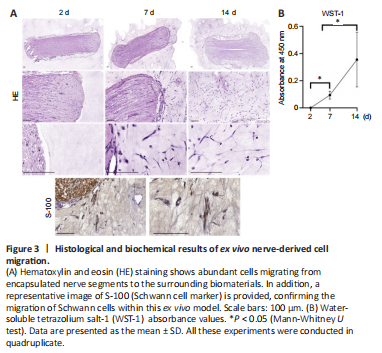
Histology revealed that cells from the nerve segments encapsulated into the hybrid hydrogels were able to progressively grow and migrate into the surrounding biomaterials. These results were especially evident between 7 and 14 days in culture. These cells showed an elongated shape indicating a positive interaction with the biomaterials used. Furthermore, immunohistochemistry for S-100 confirmed that most cells corresponded to Schwann cells (Figure 3A). WST-1 assay showed a significant increase in cell metabolic activity over time, confirming the biocompatibility and permeability of the hybrid biomaterial ex vivo (P < 0.05; Figure 3B).
Figure 7| Distal nerve stump morphological analysis 15 weeks post nerve repair.
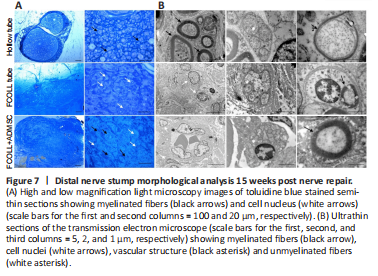
Toluidine blue semi-thin sections of the rat sciatic nerve distal portion were prepared to evaluate the general morphology of the regenerated nerves following 15 weeks post-injury. As shown in Figure 7A, only the hollow tube group presented numerous myelinated regenerating fibers, while the FCOLL –/+ ADMSC group showed few myelinated fibers and a lot of isolated cells. Moreover, their regenerated nerve had an ununiform outer circumference. The ultrastructural analysis using transmission electron microscopy (Figure 7B) confirmed the presence of myelinated fibers in the hollow tube group showing also unmyelinated fibers and small fibers in the early phases of myelination. Moreover, in the FCOLL –/+ ADMSC groups, few unmyelinated and small myelinated fibers were detectable. Numerous vascular structures were found in the FCOLL + ADMSC group consistent with the previous molecular analysis where high expression of VEGF-A was detected.
Figure 8| Histological and immunohistochemical assessment of regenerated nerve inside the conduit 15 weeks post-injury.
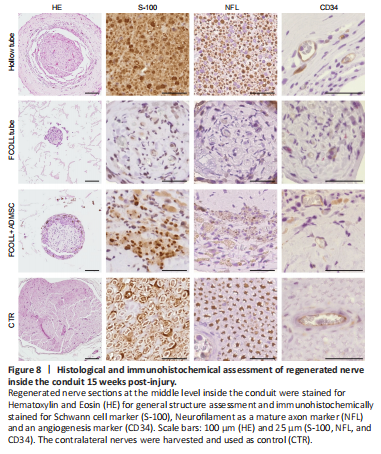
To demonstrate the nerve tissue regeneration process, hematoxylin and eosin and immunohistochemistry results from the middle part of implanted conduits are shown (Figure 8). Hematoxylin and eosin staining showed a consistent tissue formed in the middle portion of implanted grafts. This tissue was considerably more abundant, occupying more intraluminal area, in animals treated with hollow conduits (85.72 ± 12.69 × 103 μm2). Moreover, a similar pattern and amount of tissue were observed in the FCOLL + ADMSC group (59.63 ± 29.99 × 103 μm2). In contrast, the amount of tissue in the FCOLL group (18.37 ± 8.57 × 103 μm2) was scarce (Figure 8). Interestingly, histology confirmed that no connective tissue was formed between the newly formed component and the inner wall of the chitosan tube. Furthermore, immunohistochemical analysis showed that newly formed tissue presented Schwann cells (S-100) and regenerated axons (NFL) confirming the nerve tissue regeneration process. These results were more favorable in the hollow tube group followed by the FCOLL + ADMSC group. In contrast, these markers were not positive in the FCOLL group (Figure 8). In addition, associated with the nerve tissue regeneration process, clear signs of neovascularization were observed in all groups, and blood vessels were confirmed by immunohistochemistry for CD34 protein.