周围神经损伤
-
Figure 1|Ultrastructure morphological change in the endoplasmic reticulum (ER) of retinal ganglion cells (RGCs) in response to optic nerve crush (ONC).
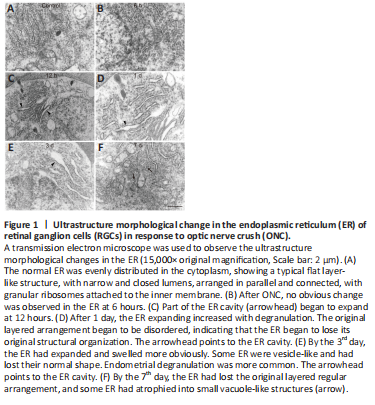
ER stress can be detected in axotomized RGCs, which might lead to the activation of apoptotic signaling pathways (Hu et al., 2012). To investigate the potential role of UPR signaling in this phenomenon, we used transmission electron microscopy to evaluate changes in the ultrastructure morphology of the RGC-ER over time after ONC. Normal ER was evenly distributed in the cytoplasm, showing a typical flat layer-like structure, with narrow and closed lumens, arranged in parallel and connected, with granular ribosomes attached to the inner membrane (Figure 1A). No obvious change was observed in the ER after 6 hours (Figure 1B). However, part of the ER cavity (Figure 1C) began to expand by 12 hours post-ONC. After 1 day, the number of expanded ER with degranulation (Figure 1D) increased and the original layered arrangement was disordered, indicating that the ER began to lose its original structural organization. After 3 days, the ER had expanded and swelled more obviously (Figure 1E). Some ER became vesicle-like and lost their normal shape, and endometrial degranulation was more common. After 7 days, the ER had lost the original regular layered arrangement, and some had atrophied into small vacuole-like structures (Figure 1F). These results suggest that the morphological changes in the ER is an important signal that indicates the time frame of RGC apoptosis.
Figure 2|Valproate (VPA) decreases the number of apoptotic retinal ganglion cells (RGCs) in the rat model of optic nerve crush (ONC).
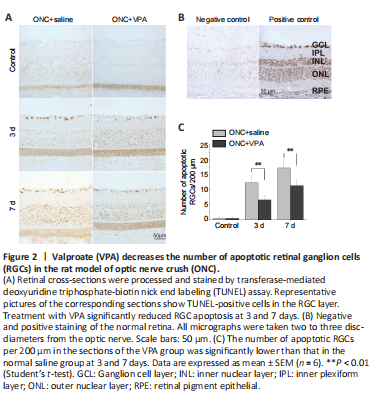
Twice daily subcutaneous VPA injection has been reported to promote the survival of RGCs after ONC (Zhang et al., 2012b). To investigate whether a single dose of VPA reduces RGC apoptosis in the current study, rats received an intravitreal injection immediately after ONC. TUNEL assay was subsequently performed on retina sections and TUNEL-positive cells were observed in the RGC layer (Figure 2A and B). RGC apoptosis was significantly lower in the VPA group than that in the control group (P < 0.01; Figure 2C). These data demonstrate that intravitreal injection is a safe and reliable option for VPA delivery.
Figure 3|Valproate (VPA) decreases retinal thinning and the loss of retinal ganglion cells (RGCs) in the rat model of optic nerve crush (ONC).
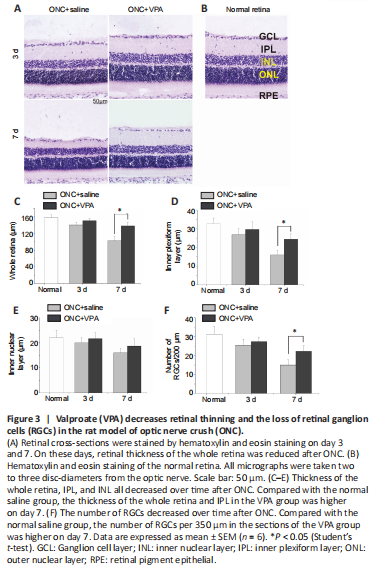
We also examined retina sections to evaluate whether intravitreal VPA can prevent retinal thinning and reduce the number of RGCs that are lost after ONC. RGC dendrites synapse with axon terminals of bipolar cells in the inner plexiform layer (IPL) of the retina. RGC injury might change the function of bipolar cells (Zhang et al., 2022). Therefore, the thickness of the whole retina, IPL, and inner nuclear layers were evaluated. The average thickness of the normal whole retina was 158.2 ± 6.76 μm. ONC injury reduced the thickness of the whole retina in a time-dependent manner, with or without VPA treatment (Figure 3A and B). However, after 7 days, thinning of the whole retina and the IPL were attenuated after VPA treatment (P < 0.01; Figure 3C and D). No significant difference in the thickness of the inner nuclear layer was observed between the VPA and normal saline groups at 3 and 7 days post-ONC (Figure 3E). The average number of RGCs in normal retina sections was 31.2 ± 4.12 per 350 μm. Although ONC injury resulted in a time-dependent decrease in the number of RGCs regardless of group, the number of RGCs was significantly higher 7 days post-ONC in the VPA group than in the control group (P < 0.05; Figure 3F). Thus, we confirmed the protective effect of VPA on the retinal neurons.
Figure 4|Glucose-regulated protein 78 (GRP78) is expressed in retinal ganglion cells (RGCs).
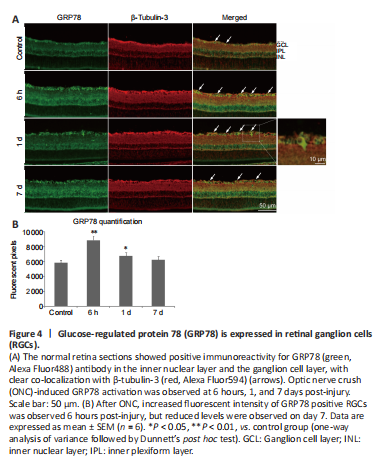
GRP78 is a master chaperone during ER stress and regulates the signaling of UPR in neurodegenerative disorders (Gorbatyuk and Gorbatyuk, 2013). We examined the location of GRP78 in the retinal sections and observed immunoreactivity in the ganglion cell layer and inner nuclear layer of normal controls. The positive expression of GRP78 was co-localized primarily with β-tubulin-3 (RGCs marker; Jiang et al., 2015) and surrounded the RGC nuclei (Figure 4A). We found that GRP78 expression had increased by 6 hours after ONC, but levels decreased by day 7 (Figure 4B).
Figure 5|C/EBP homologous protein (CHOP) is expressed in retinal ganglion cells (RGCs).
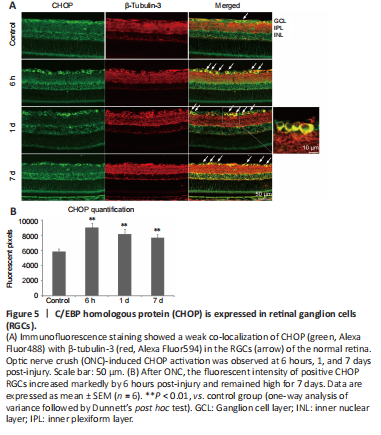
CHOP is another key mediator of the ER-stress response and an important pro-apoptotic signaling molecule after axonal injury (Syc-Mazurek et al., 2017). To investigate the location of CHOP in the retina sections, immunofluorescence staining was performed. We found slight activity in the ganglion cell layer of the control rats, which was co-localized with β-tubulin-3 in the RGCs (Figure 5). High expression of CHOP in the RGCs was induced by ONC and remained for 7 days.