脊髓损伤
-
Figure 2 | Lentt-ChABC treatment increased CSPG degradatton and reduced glial response affer spinal cord injury.
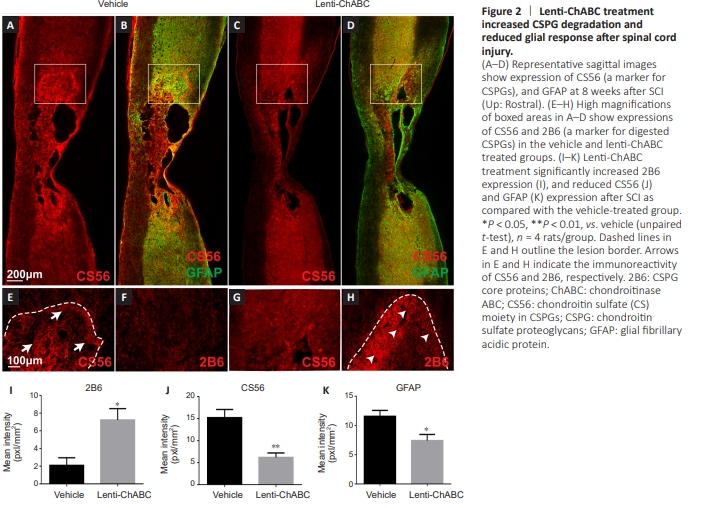
To determine the effect of lent-ChABC on CSPG degradation and glial response, CS56 (a marker for CSPGs), 2B6 (a marker for digested CSPGs), and GFAP (a marker for astrocytes) were examined in spinal cord sections using immunofluorescence double staining. Our results showed that CS56 expression was signiffcantly reduced in the lentt-ChABC group (P < 0.01; Figure 2C, G, and J) compared with the vehicle group (Figure 2A, E, and J) affer SCI. Conversely, 2B6 expression was signiffcantly increased in the lentt-ChABC group compared with the vehicle group affer SCI (P < 0.05; Figure 2F, H, and I). Finally, GFAP expression was significantly reduced in the lenti-ChABC group compared with the vehicle group (P < 0.05; Figure 2B, D, and K). These ffndings suggest that the enzyme not only digested CSPG, but also inhibited astrocyttc glial response.
Figure 3 | Lentt-ChABC treatment promoted the longitudinal migratton of graffed SCs to the host spinal cord affer injury.

Our next step was to determine whether Lentt-ChABC facilitated the migration of grafted SCs from the injury site to the host spinal cord. We found that lentt-ChABC not only digested CSPGs and aided in the reduction of GFAP (Figure 3A–D), but also improved transplanted SCs to migrate. Compared with SCs administered vehicle alone (Figure 3B), SCs treated with the lentt-ChABC migrated for a considerable distance into the rostral and caudal host spinal cord (Figure 3D). Compared with the SC alone group, SCs in the combination group (SC + lenti-ChABC) traveled signiffcantly longer distances when measured from the lesion boundary to the rostral extremity (P < 0.05; Figure 3E). Likewise, signiffcantly longer distances from the lesion boundary to the caudal extremity were found in the SC + lentt-ChABC group compared with the SCs alone group (P < 0.01; Figure 3D and E). Overall, the SC + lenti-ChABC group promoted remarkable SC migratory distances in rostrocaudal directions, almost 4-fold greater than the SCs alone group (P < 0.01; Figure 3E).
To detect the possible effects of the combinatton of SC and lenttChABC on the lesion size and glial scar formation, we examined astrocyttc acttvatton by examining GFAP reacttvity in the spinal cord. Animals that received SCs and lenti-ChABC treatment showed a signiffcant decrease in GFAP expression compared with the vehicle group (P < 0.01) and the lentt-ChABC only group (P < 0.05; Figure 3F). This result suggests that the combination treatment of SCs and lenti-ChABC caused a decrease in glial reactivity. We further examined lesion and cavity volume as indicators of ttssue damage at 8 weeks post-injury. Both the SC alone group (P < 0.05) and the SC + lentt-ChABC group (P < 0.01) showed signiffcantly less lesion volume compared with the vehicle group (Figure 3G). Similarly, the SC alone (P < 0.01) and the SC + lentt-ChABC group (P < 0.01) showed signiffcantly less cavitatton compared with the vehicle or lentt-ChABC groups (Figure 3H). Indeed, most of the animals affer the SC transplantatton showed no cavitatton whatsoever because their lesion sites were fflled with graffed SCs.
Figure 4 | SCs combined with lentt-ChABC promoted the growth of 5-HT+ axons into and beyond the lesion site.
Since serotoninergic (5-HT+ ) axons are considered critical to functional recovery, we examine the growth of serotonergic axons into the SC graft environment after SCI (Takiguchi et al., 2022). More 5-HT+ axons were observed to grow into the SC graffs and caudal to the injury in the combined group (SC + lenttChABC) at 8 weeks affer SCI (Figure 4A–B4). Within the SC graff, both the SCs alone and SC + lenti-ChABC groups significantly increased the numbers of ingrowth 5-HT+ axons compared with the vehicle group (P < 0.01; Figure 4D and E) A higher number of 5-HT+ axons were found in the SC + lentt-ChABC group compared with the lenti-ChABC group (P < 0.05; Figure 4C–E), indicating that SCs facilitated 5-HT+ axonal ingrowth. In the spared tissue, the number of 5-HT+ axons in the SC + lenti-ChABC group was signiffcantly higher compared with that in the vehicle (P < 0.01) and the lentt-ChABC (P < 0.05) groups (Figure 4C and F). In the SC + lentt-ChABC treated group, the number of 5-HT+ axons were significantly higher compared with the other treatment groups at various distances from the lesion/SC graft site (Figure 4D). For example, the number of 5-HT+ axons at 500 μm caudal to the lesion epicenter in the SC + lentt-ChABC group exceeded the number observed in the vehicle group (P < 0.01), the SC + vehicle group (P < 0.05) or the lentt-ChABC only group (P < 0.05; Figure 4C and D). Overall, both SC transplantatton groups (SC + vehicle and SC + lentt-ChABC) show signiffcant 5-HT+ axonal growth into (Figure 4E), around (Figure 4F), and beyond (Figure 4D) the SC graft/lesion cavity as compared with the vehicle group. A close correlation was found between the 5-HT+ axons at the lesion epicenter and BBB scores at 8 weeks affer SCI (P < 0.01; Figure 4G). Similarly, a close correlatton was found between the 5-HT+ axons at 500 μm caudal to the lesion epicenter and BBB scores at 8 weeks affer SCI (P < 0.05, Figure 4H).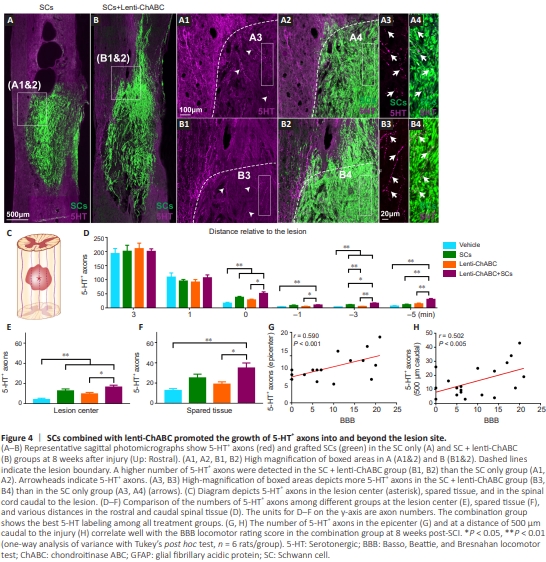
Figure 5 | SCs combined with lentt-ChABC promoted the growth of TH+ axons within the graffs and caudal to the injury.
 To test whether SCs combined with lentiChABC promote TH+ axonal regeneration, we examined the presence of TH+ axons at and beyond the lesion. At 8 weeks post-injury, TH+ axons were found to grow extensively into the SC graffs (Figure 5A–B6). However, signiffcantly more TH+ axons were found to grow into the SC grafts of the SC + lenti-ChABC group (P < 0.01) compared with the SC + vehicle group (Figure 5C and D). Additionally, the SC + lenti-ChABC group showed a stronger TH immunoreactivity compared with that of the lentiChABC alone group (P < 0.01; Figure 5C and D). The number of TH+ axons at the spared spinal cord ttssue were also signiffcantly higher in the SC + lentt-ChABC group compared with the vehicle (P < 0.05) or the lentt-ChABC alone group (P < 0.05; Figure 5F). The total number of TH+ axons were signiffcantly higher in the SC + lentt-ChABC group compared with that in the vehicle (P < 0.01) or lentt-ChABC alone group (P < 0.01; Figure 5E). In the distal host spinal cord at 500 μm caudal to the lesion border, a signiffcantly greater number of TH+ axons were found in the SC + lentiChABC group compared with the vehicle (P < 0.01), SCs only (P < 0.05), or lentt-ChABC only group (P < 0.05; Figure 5I). Further, a significant correlation between the numbers of the TH+ axon immunoreacttvity and BBB outcomes was found at 8 weeks postinjury (P < 0.05; Figure 5J).
To test whether SCs combined with lentiChABC promote TH+ axonal regeneration, we examined the presence of TH+ axons at and beyond the lesion. At 8 weeks post-injury, TH+ axons were found to grow extensively into the SC graffs (Figure 5A–B6). However, signiffcantly more TH+ axons were found to grow into the SC grafts of the SC + lenti-ChABC group (P < 0.01) compared with the SC + vehicle group (Figure 5C and D). Additionally, the SC + lenti-ChABC group showed a stronger TH immunoreactivity compared with that of the lentiChABC alone group (P < 0.01; Figure 5C and D). The number of TH+ axons at the spared spinal cord ttssue were also signiffcantly higher in the SC + lentt-ChABC group compared with the vehicle (P < 0.05) or the lentt-ChABC alone group (P < 0.05; Figure 5F). The total number of TH+ axons were signiffcantly higher in the SC + lentt-ChABC group compared with that in the vehicle (P < 0.01) or lentt-ChABC alone group (P < 0.01; Figure 5E). In the distal host spinal cord at 500 μm caudal to the lesion border, a signiffcantly greater number of TH+ axons were found in the SC + lentiChABC group compared with the vehicle (P < 0.01), SCs only (P < 0.05), or lentt-ChABC only group (P < 0.05; Figure 5I). Further, a significant correlation between the numbers of the TH+ axon immunoreacttvity and BBB outcomes was found at 8 weeks postinjury (P < 0.05; Figure 5J).Figure 9 | SCs combined with lentt-ChABC promoted long-term SCs survival, migratton and facilitated axonal growth at 6 months post-injury when SCs were graffed at 1 week and 3 months affer SCI.
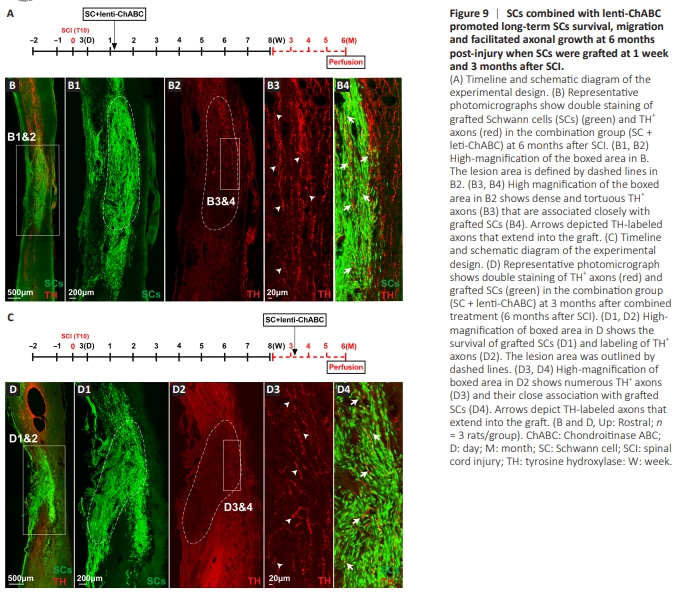
To determine whether the combination of SCs and lentiChABC promotes long-term survival of grafted SCs and axonal regeneration, we kept the animals to survive for 6 months. We observed that SCs continue to survive and migrate beyond the lesion cavity up to 6 months affer injury and graffing (Figure 9A and B). Importantly, descending dopaminergic (TH+ ) axons were found to grow and elongate longitudinally along SC graffs in the combined treatment group (Figure 9B–B4), indicating that the combination of SCs and lenti-ChABC can promote persistent SC survival, migration, and supportive of axonal growth across the lesion of a SCI.
We determined whether the combined treatment of SC and lenti-ChABC could promote SC survival and axonal growth in a chronic SCI injury model. We administered the combined SC and lentt-ChABC treatment at 3 months affer SCI and evaluated the results at 6 months after SCI (Figure 9C and D). We found that SCs survived well, migrated beyond the lesion cavity, and supported the growth of TH-posittve axons following chronic SCI (Figure 9D–D4).