神经损伤与修复
-
Figure 1 | Construction of the Rab5 overexpression system and confirmation of overexpression in vivo.
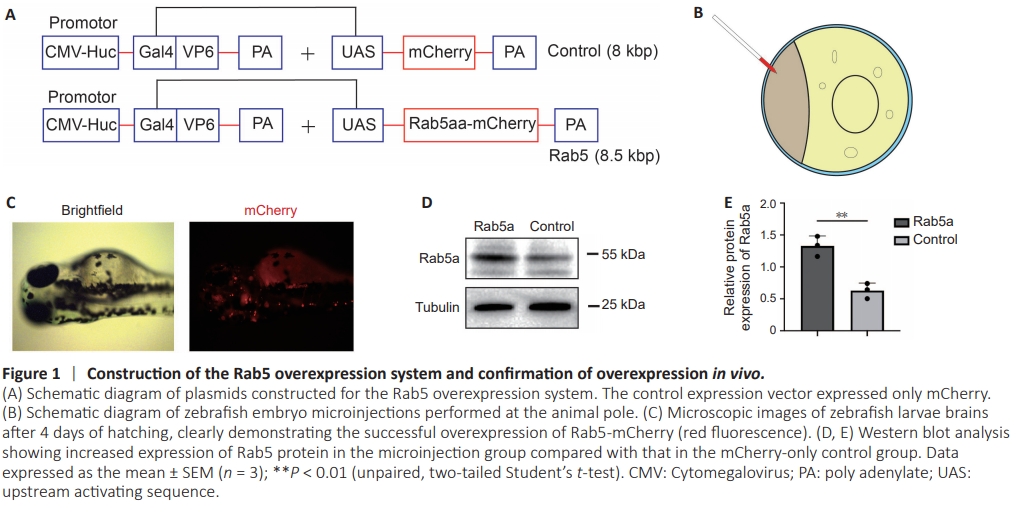
Figure 2 | Effect of Rab5 overexpression on axon regeneration during single-cell electroporation of M-cells.
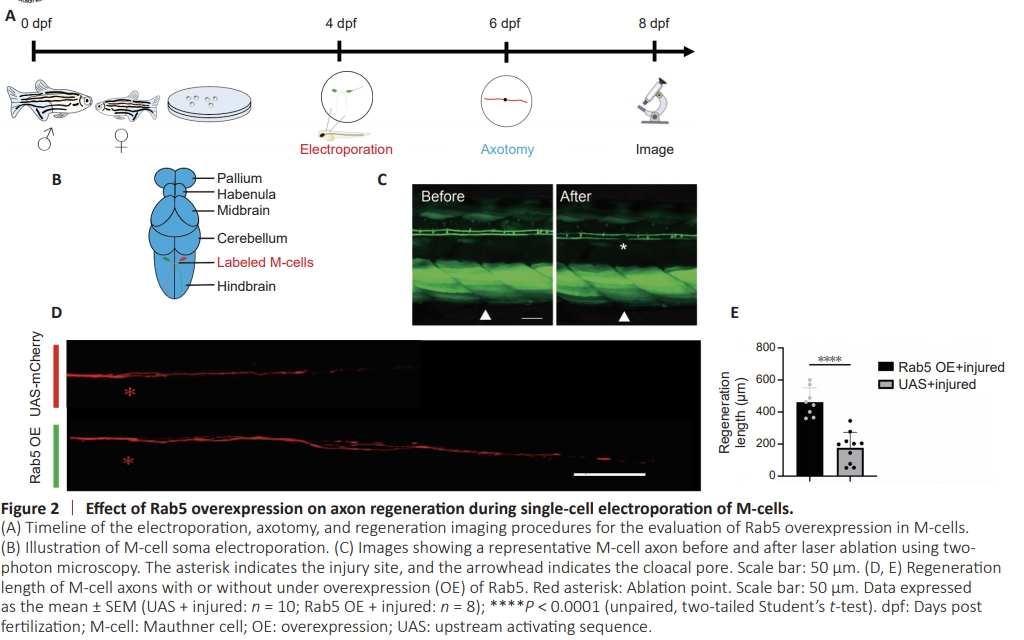
To investigate the role of Rab5 in axonal regeneration in zebrafish, we established an Rab5 overexpression model using a two-plasmid system comprising pUAS-Rab5-mCherry and pCMV/Huc-GAL4-VPL16 (Figure 1A; Korn and Faber, 2005; Huang et al., 2017; Hu et al., 2018; Wang et al., 2022). Following co-microinjection of both plasmids into zebrafish eggs (Figure 1B), we observed red fluorescence in zebrafish larvae at 4 dpf (Figure 1C). Western blot analysis further confirmed the overexpression of Rab5 in these larvae (Figure 1D and E). To introduce the plasmids into the M-cells of zebrafish larvae, we used single-cell electroporation at 4 dpf. At 12 hours post-electroporation, zebrafish larvae with M-cells expressing the mCherry fluorescent protein were selected. At 6 dpf, we performed two-photon disruption of axons in the parallel region near the zebrafish cloacal pore. Axons were then photographed using confocal microscopy at 8 dpf (2 days post axotomy) (Figure 2A and B). Through confocal imaging, we observed that the lengths of regenerated axons of M-cells were significantly increased in zebrafish larvae with Rab5- overexpression compared with those in control larvae (Figure 2C–E)
Figure 3 | Optimization of the transmission electron microscopy sample preparation processes and confirmation of the M-cell position.
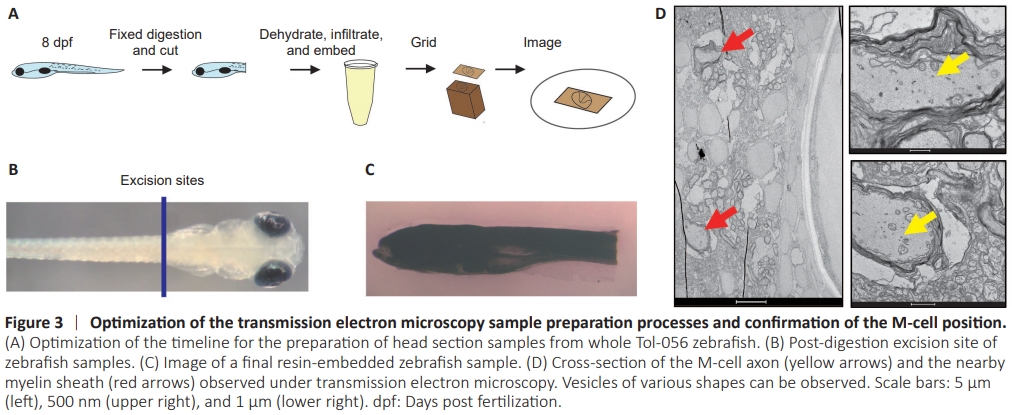
Figure 4 | Effects of axonal injury and Rab5 overexpression (OE) on the number of transport vesicles within the M-cell axon.
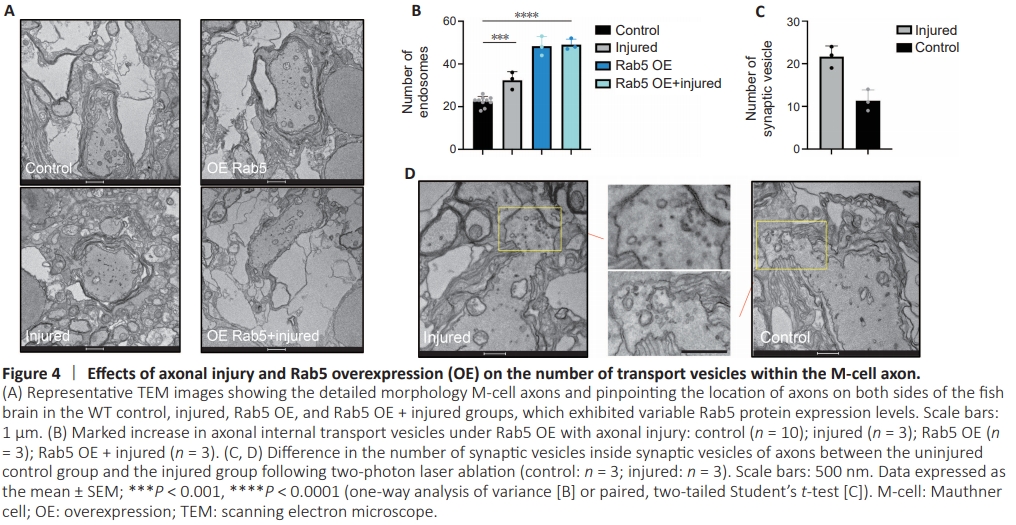
Previous studies have demonstrated the regenerative capacity of M-cells (Korn and Faber, 2005; Huang et al., 2017; Hu et al., 2018). Rab5 is primarily involved in cytokinesis (Dodson et al., 2013; Kenyon et al., 2015; Blacque et al., 2018; Zhang et al., 2019; Yuan and Song, 2020). Cytokinesis involves extensive transport of substances and spatiotemporal regulation, and Rab5 plays a crucial role in endosome generation and transport processes. We hypothesized that Rab5 may play a role in the transportation of materials within axons. To investigate the function of the Rab5 in axonal regeneration following injury, we employed TEM to observe intracellular material transport in M-cell axons of zebrafish (Hildebrand et al., 2017; Lam et al., 2021). Conventional specimen preparation techniques often hinder the penetration of imaging techniques through the skin, limiting the visibility of internal structures. To ensure optimal sample preparation, we gently digested the head section before isolating it from whole zebrafish and fixing it with osmium tetroxide (Figure 3A), which enabled us to visualize the brain structures more clearly (Figure 3B and C). Using TEM images, we were able to observe M-cell axons and their myelin sheath (Figure 3D). Comparing WT control zebrafish with injured zebrafish, with and without Rab5 overexpression, we found a notable increase in the number of vesicles (excluding synaptic vesicles) within injured M-cell axons, particularly in the Rab5–overexpression group (Figure 4A and B). However, it is important to note that axonal injury did not lead to a further increase in vesicle numbers in the Rab5-overexpression group, suggesting that their vesicular transport capacity may have reached saturation (Figure 4C). Moreover, our findings indicated that axons close to the injury site had higher numbers of vesicles and more active synapses in both the overexpression group and the injury group (Figure 4C and D). These findings suggested that further exploration is needed to understand the role of Rab5 in axon regeneration and vesicular transport following injury.
Figure 5 | Axonal regeneration in zebrafish M-cells treated with Rab5 inhibitors.
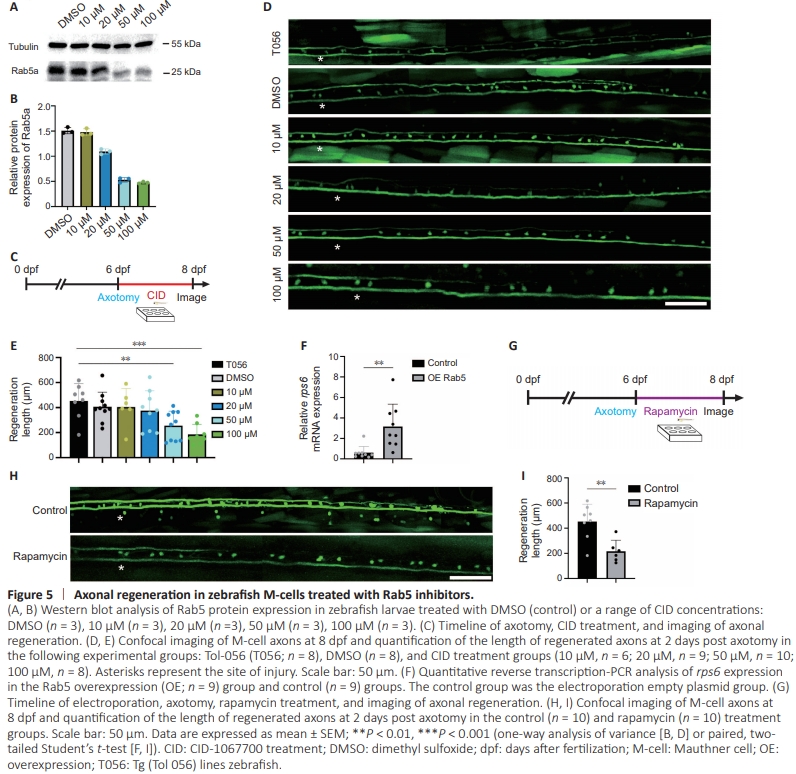
To verify the role of Rab5 in axonal regeneration, we employed the Rab inhibitor CID (Kaveh et al., 2022; Wang et al., 2022). Western blot monitoring of Rab5 expression in zebrafish larvae treated with CID showed a gradual decrease in Rab5 expression with increasing drug concentration (Figure 5A and B). Next, we treated zebrafish larvae with CID for 48 hours following two-photon disruption at 6 dpf andevaluated the length of regenerated axons at 8 dpf using confocal microscopy (Figure 5C and D). Statistical analysis indicated that post-disruption axonal regeneration markedly decreased with increasing inhibitor concentration (Figure 5E). Our findings thus far clearly indicate that the Rab5 inhibitor CID had a suppressive effect on axonal regeneration, whereas Rab5 itself promoted the process of axonal regeneration.
Previous studies have shown that Rab5 and phosphatidylinositol 3-kinase (PI3K) are involved in promoting liver regeneration in zebrafish, indicating their role not only in intracellular material transport, but also in promoting axonal regeneration through the activation of signaling pathways (Chen and Wang, 2001; Capalbo et al., 2011; Clark et al., 2011; Runyan et al., 2012; Law et al., 2017; Cheng Mm et al., 2019; Jung et al., 2021; Luo et al., 2022). The ribosomal protein S6 kinase belongs to the threonine/serine kinase family and is responsible for regulating the phosphorylation of ribosomal S6. This phosphorylation event is vital in transcription, cell cycle progression, and cell survival (Kenyon et al., 2015). Phosphorylated S6 (pS6) serves as a marker of PI3K activation (Vieira et al., 2003; Whitecross and Anderson, 2017; Yang et al., 2018; Wang et al., 2020; Zhang et al., 2020). Our qRT-PCR analysis revealed increased expression levels of rps6 in zebrafish larvae after axonal injury (Figure 5F), providing evidence for the concurrent activationof the PI3K pathway. To further support this finding, we administered rapamycin, an inhibitor of mTOR, to zebrafish larvae immediately following axotomy and captured images of the fish 48 hours later (Figure 5G). Notably, we observed a significant reduction in axonal regeneration in the rapamycintreated group compared to that in the control group (Figure 5H and I). These findings indicated that the PI3K-mTOR-pS6 pathway may be activated during axonal regeneration in zebrafish.