视神经损伤
-
Figure 2 | Allogeneic transplant of TEG3 rescues axotomized RGCs but activates microglial cells.

First, we tested the neuroprotective potential of intravitreally injected TEG3 cells on axotomized RGCs. TEG3 cells come from Wistar rats and were injected into the vitreous of Wistar (syngeneic mode) or Sprague-Dawley (allogeneic mode) rats right after ONC. Retinas were anatomically analyzed 7 days later, when without treatment > 50% of RGCs have died (Nadal-Nicolás et al., 2015; Vidal-Sanz et al., 2017). We observed that only the allogeneic transplant elicited significant RGC neuroprotection (Figure 2A–C). We, therefore, investigated whether allogeneic TEG3 neuroprotection was still observed after 21 days when 85% of RGCs would have died without treatment. As shown in Figure 2D, there was no difference in the number of RGC between retinas treated either with TEG3 or vehicle. Therefore, TEG3 neuroprotection was transient. Finally, we observed the morphological activation of microglia identified by Iba1 immunodetection. We found that in both syngeneic and allogeneic TEG3-transplanted retinas, microglial cells were more activated than after ONC alone, an activation that increased from 7 to 21 days (Figure 2E and F). In the retina, as in the rest of the central nervous system, Iba1 was specifically expressed by microglial cells (Additional Figure 1A). However, this protein was also expressed by peripheral macrophages. Although most of the Iba1+ cells in the experimental retinas would be microglial cells, we could not exclude macrophage infiltration after TEG3 transplantation, which once infiltrated were indistinguishable from microglial cells.
Figure 3 | The intravitreal graft of TEG3 in intact retinas causes RGC death and activates microglial cells.
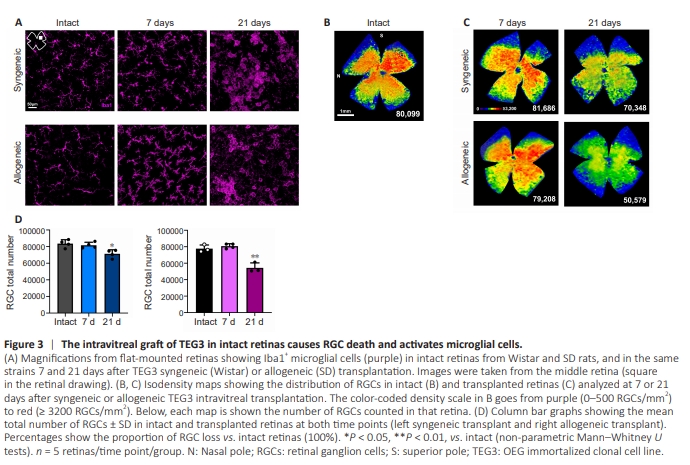
In view of the above results, we wondered whether TEG3 cells elicited an immune or toxic response in intact retinas. Allogeneic and syngeneic TEG3 were transplanted into intact animals, and retinas were analyzed 7 or 21 days later. At 7 days, both transplants induced microglial activation, which was more significant and pronounced in the allogeneic transplants and increased in both scenarios at 21 days (Figure 3A). We then analyzed the RGCs, and to our surprise, both transplants caused significant RGC death after 21 days, which was greater with the allogeneic transplant (Figure 3B–D), despite being this transplant neuroprotectant to axotomized RGCs.
Figure 4 | TEG3 cells form an epimembrane after intravitreal administration without integrating into the retina and express MHCII constitutively.
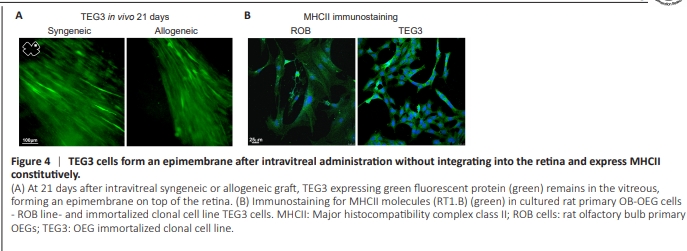
In vivo 21 days after transplantation into intact retinas, GFP+ TEG3 cells were still present in the vitreous, forming an epimembrane without integrating into the retina (Figure 4A). Here, we also described a fact that was probably fundamental to the transplantation effects of OEG: in culture, TEG3 cells, like other OEG cells from olfactory bulb tested – i.e., primary cultured ROB cells – immunostained for MHCII (Figure 4B). Therefore, to our knowledge, OEGs have been unveiled as antigen-presenting cells for the first time. Possible implications of our finding for TEG3 transplantation in our retina experimental paradigms are discussed.
Figure 5 | Microglial inhibition with minocycline or systemic immunosuppression rescues RGCs from TEG3 toxicity in intact retinas.
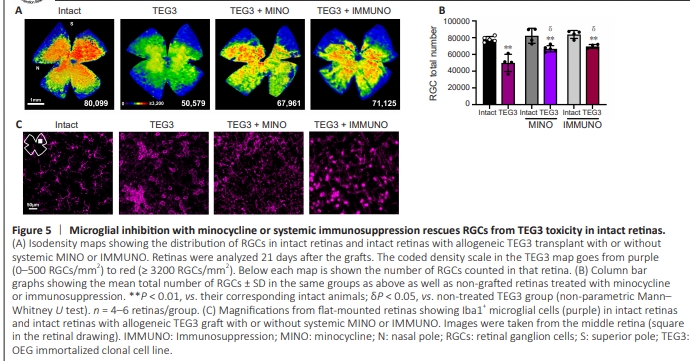
Microglial cells in the transplanted retinas were highly activated and we thought that this activation may play a role in RGC death through an uncontrolled inflammatory response. Thus, the next group of intact animals treated with intravitreal TEG3 or vehicle was systemically treated with minocycline. This tetracycline antibiotic selectively inhibits microglial cells (Kobayashi et al., 2013) and was analyzed at 21 days, when we observed that the TEG3 transplants caused RGC loss. In the syngeneic transplant, microglial inhibition did not rescueRGCs (Additional Figure 1B and C). However, in allogeneic transplantation, RGC loss decreased from 33% to 19% (Figure 5A and B), similar to the percentage of loss observed in syngeneic transplantation with or without minocycline treatment. Microglial cells in TEG3+ minocycline intact retinas showed a lower level of anatomical activation than in TEG3 intact retinas, in which microglia appeared more ameboid than after minocycline treatment (Figures 5C and Additional Figure 1A). We then analyzed the effect of systemic immunosuppression on RGC survival 21 days after allogeneic TEG3 transplantation in intact retinas. We found that the rescue effect was no different from that observed with minocycline (Figure 5A and B). Microglial cells in TEG3 immunosuppressed intact retinas had a less activated morphology than in TEG3-intact retinas and a different morphology than in the minocycline group (Figures 5C and Additional Figure 1A).
Figure 6 | Microglial inhibition with minocycline or systemic immunosuppression rescues RGCs but does not synergize with TEG3 neuroprotection.
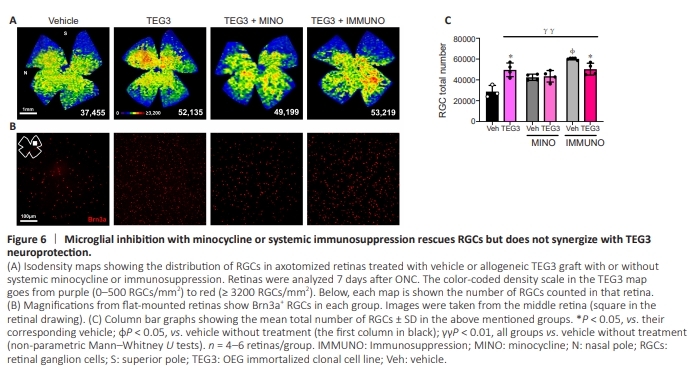
Since both treatments rescued RGCs in allogeneic transplanted intact retinas, we investigated whether these treatments improved TEG3-induced RGC neuroprotection after ONC. Neither treatment synergized with TEG3 neuroprotection at 7 days. However, both systemic therapies were able to neuroprotect RGCs as well (minocycline) or better (immunosuppression) than TEG3 alone (Figure 6A–C).