脑损伤
-
Figure 4 | YL-0919 reduces neuronal damage and alleviates brain edema after TBI.
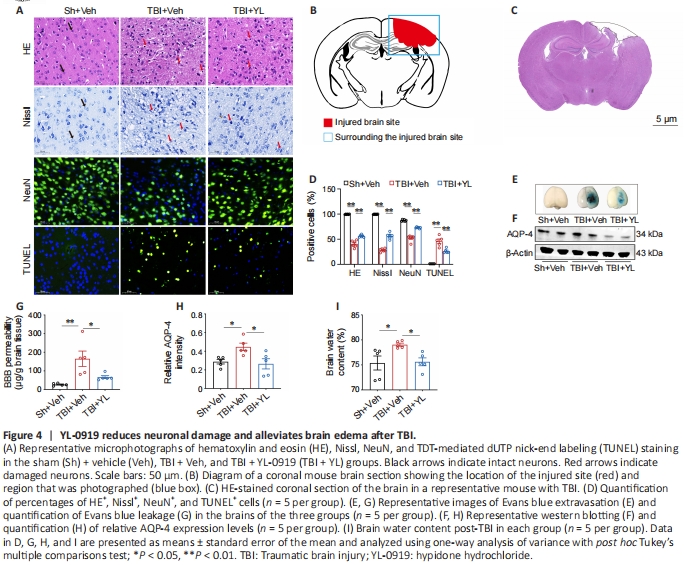
To assess neuronal survival, sections of tissue surrounding the injured region were subjected to HE, Nissl, NeuN, and TUNEL staining (Figure 4A). The drawing and representative image of a coronal brain section shown in Figure 4B and C, respectively, showcase both the injured area and the surrounding region. HE-stained cells exhibited well-defined contours, dense arrangement, and undamaged nucleoli. Compared with the Sh + Veh group, there was also a notable decrease in the percentage of stained cells near the injuredarea in the TBI + Veh group, with YL-0919 treatment leading to the conservation of a great number of intact cells in the TBI+YL group (F(2, 15) = 319.6, P < 0.001; Figure 4D). Nisslstained cells were considered to represent intact neurons with flushed cell bodies, whereas damaged neurons had shrunken cell bodies and pyknotic nuclei. Similarly, the percentage of Nissl-stained cells was significantly increased in the TBI + YL group compared with that in the TBI + Veh group (F(2, 15) = 489.4, P < 0.001; Figure 4D). NeuN-stained cells, which were considered to represent surviving mature neurons, were notably decreased surrounding the injured site in the TBI+Veh group compared with that in the Sh + Veh group. However, this reduction was reversed by YL-0919 (F(2, 12) = 79.46, P < 0.001; Figure 4D). The proportion of TUNELstained cells, indicating apoptosis caused by TBI, was also significantly decreased by the administration of YL-0919 (F(2, 13) = 74.60, P < 0.001; Figure 4D). Dysfunction of the BBB and brain edema post-TBI is widely recognized. To assess the permeability of the BBB, EB stain was administered through the tail vein (Figure 4E), and the mice were euthanized 2 hours later. Quantification of the staining revealed that very little EB passed through the BBB in the Sh + Veh group, with the TBI + Veh group exhibiting the highest extravasation. However, the level of EB was notably diminished in the TBI + YL group compared with that in the TBI + Veh group (F(2, 12) = 8.569, P < 0.001; Figure 4G). The relationship between AQP-4, the balance of brain water, and the formation of edema is strong. Western blot analysis revealed significantly higher expression of AQP-4 in the TBI + Veh group compared to that in the Sh + Veh group (Figure 4F); however, administration of YL-0919 in the TBI + YL group significantly lowered the AQP-4 level (F(2, 12) = 5.727, P = 0.0179; Figure 4H). Finally, we assessed the brain water content as an indicator of the degree of brain edema. Compared to the Sh+Veh group, the TBI + Veh group had elevated brain water content that was reversed by YL-0919 treatment in the TBI + YL group (F(2, 12) = 4.712, P = 0.0309; Figure 4I). These findings suggest that YL-0919 mitigates neuronal damage, improves BBB function, and reduces brain edema resulting from TBI.
Figure 5 | YL-0919 alleviates TBI-induced oxidative stress.
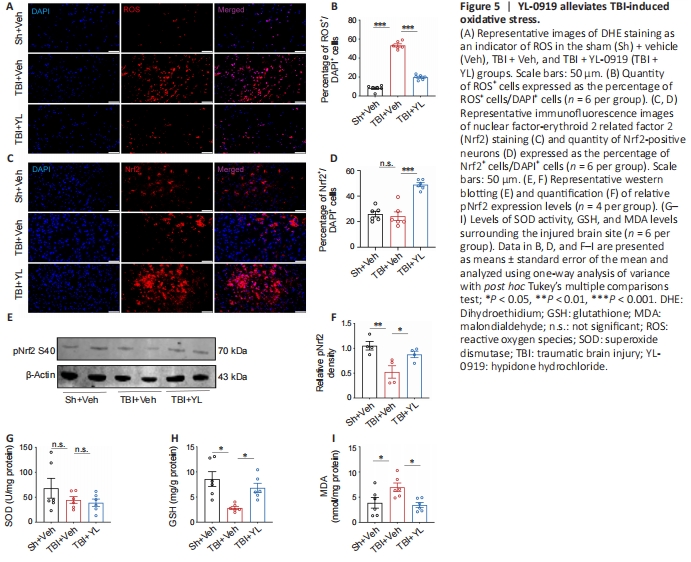
Given the importance of oxidative stress in the pathological process of TBI, we proceeded to validate the antioxidative properties of YL-0919 in TBI model mice. Initially, DHE staining was employed to assess ROS levels. The TBI + Veh group exhibited a notable rise in the proportion of ROS-positive cells compared to the Sh + Veh group; however, the administration of YL-0919 in the TBI + YL group reduced this increase in ROSpositive cells (F(2, 15) = 298.0, P < 0.001; Figure 5A and B). Nrf2 is an important regulator of the antioxidant system. The phosphorylated form is translocated into the nucleus, where it induces the expression of GSH and antioxidant enzymes such as SOD (Wang et al., 2020). Immunofluorescence staining showed that the expression of Nrf2 was markedly increased in the TBI + YL group compared with that in the TBI + Veh group (F(2, 15) = 30.65, P < 0.001; Figure 5C and D). Western blotting analysis showed that the level of phosphorylated Nrf2 (Ser40) was decreased in the TBI + Veh group compared with that in the Sh + Veh group, but was notably increased by YL-0919 (F(2, 9) = 7.971, P = 0.0102; Figure 5E and F). In addition, we measured the activities of SOD and GSH, along with the level of MDA, as an additional measure of oxidative stress. The SOD activity level did not show any notable variation among the three groups (F(2, 15) = 1.439, P = 0.2680; Figure 5G), whereas the GSH level was significantly lower in the TBI + Veh group than in the Sh + Veh group, but was increased following YL-0919 treatment (F(2, 15) = 8.422, P = 0.0035; Figure 5H). By contrast, the TBI + Veh group exhibited a notable rise in the MDA level compared with the Sh + Veh group, an alteration that was reversed by YL0919 treatment (F(2, 15) = 5.325, P = 0.0179; Figure 5I). These findings indicat that reduction in oxidative stress may be one ofthe beneficial effects by which YL-0919 protects against TBI.
Figure 6 | YL-0919 inhibited H2O2-induced oxidative stress in HT22 cells.
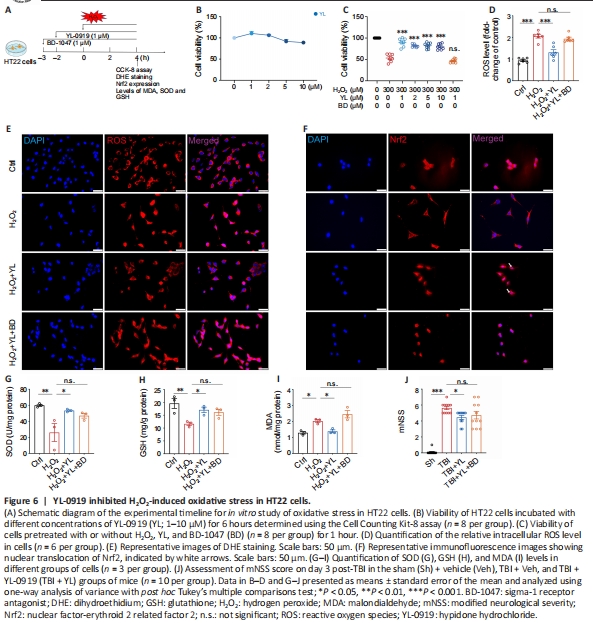
We employed H2O2 to establish a representational model of oxidative stress in HT22 cells as previously described by Singh et al. (2016), and assessed cell viability at different time intervals after exposure to different concentrations of H2O2. As shown in Additional Figure 4, cell viability decreased with increasing H2O2 concentration and time. Cell Counting Kit‐8 was used to determine the viability of HT22 cells incubated with 300 μM H2O2 at different concentrations of YL-0919 (0.1–10 μM) for 6 hours (Additional Figure 5). To investigate the antioxidative mechanisms of YL-0919, HT22 cells were incubated with 1 μM BD-1047 for 1 hour, then treated with 1–10 μM YL-0919 for 2 hours and subsequently stimulated with 300 μM H2O2 for 4 hours prior to analysis (Figure 6A). While treatment with YL-0919 alone did not have any impact on cell viability (F(4, 35) = 7.379, P > 0.05; Figure 6B), pretreatment with YL-0919 at 1–10 μM increased the viability of cells stimulated with H2O2. This protective effect of YL0919 was reversed when BD-1047 was added (F(6, 47) = 68.11, P < 0.001; Figure 6C). DHE staining of these cells showed that H2O2 markedly increased ROS levels, while YL-0919 attenuated ROS levels (Figure 6E). This attenuation was also reversed by BD-1047 (F(3, 20) = 36.25, P < 0.001; Figure 6D). When we assessed Nrf2 expression using immunofluorescence staining, interestingly the results showed that YL-0919 promoted the nuclear translocation of Nrf2 (Figure 6F). Moreover, YL0919 treatment significantly increased the levels of SOD and GSH, but BD-1047 reversed these effects (F(3, 8) = 6.295, P = 0.0168, Figure 6G; and F(3, 8) = 6.477, P = 0.0156, Figure 6H). The administration of YL-0919 or BD-1047 had the opposite effect of MDA levels (F(3, 8) =15.66, P = 0.001; Figure 6I). Finally, we found that 4 mg/kg BD-1047 also reversed the YL-0919- mediated improvement in mNSS of mice on day 3 post-TBI (F(3, 40) = 74.20, P < 0.001; Figure 6J). In summary, YL-0919 appears to reduce the accumulation of oxidative substances and enhance antioxidant defense, an effect that can be blocked by an σ-1R antagonist.