神经损伤与修复
-
Figure 1 | PRMT6 is mainly expressed in mouse DRG nociceptive neurons. (A) In DRG neurons, β-tubulin III (green) co-localized with PRMT6 (red). (B) Astroc
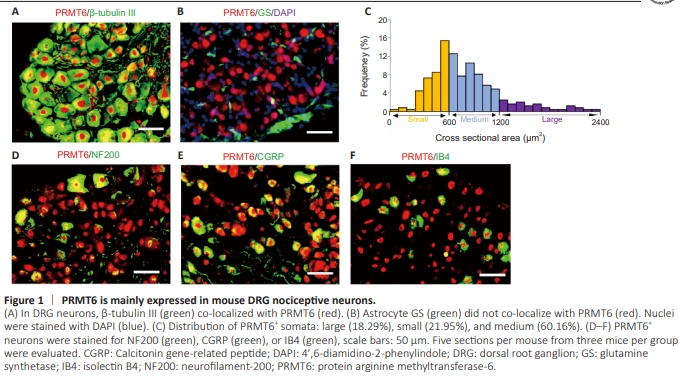
To investigate the correlation between PRMT6 and neuropathic pain, we performed a double/triple-labeled immunofluorescence assay to evaluate PRMT6 distribution in the DRG. We observed co-localization of β-tubulin III (a neuron-specific marker) with PRMT6 within the DRG (Figure 1A); however, there was no significant overlap in staining between PRMT6 and GS (a marker of satellite glial cells; green) (Figure 1B). These results suggest that PRMT6 influences DRG neuronal activation. Furthermore, cross-sectional analysis of neuronal somata showed large (> 1200 μm2 ; 18.29%), medium (600–1200 μm2 ; 60.16%), and small (< 600 μm2 ; 21.95%) PRMT6-labeled neurons (Figure 1C). Subpopulation analysis demonstrated that neurons expressing PRMT6 were also positive for NF200 expression in myelinated Aβ fibers and medium and giant cells (32.70%), CGRP expression in small peptidergic neurons (48.90%), and IB4 expression in small non-peptidergic neurons (41.50%) (Figure 1D–F).
Figure 2 | PRMT6 expression is reduced in the injured DRG in a mouse model of neuropathic pain.
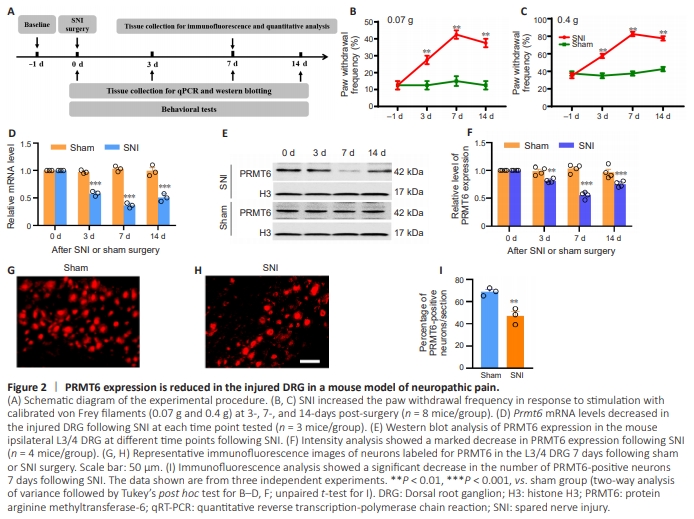
To examine the role of PRMT6 in neuropathic pain progression, we examined PRMT6 expression in the DRG of SNI and shamoperated mice. The experimental design is shown in Figure 2A. Consistent with results from a prior study (Zhang et al., 2023b), SNI, but not sham surgery, induced mechanical allodynia hyperalgesia on the ipsilateral side at 3, 7, and 14 days after surgery (Figure 2B and C). We also examined Prmt6 mRNA expression in the DRG at 3, 7, and 14 days post-SNI surgery and found that it was markedly decreased in the SNI group compared with the sham group (Figure 2D). Furthermore, western blotting showed decreased PRMT6 expression in the L3/4 DRG ipsilateral region of the SNI group (Figure 2E and F). Furthermore, PRMT6 expression in SNI mice was significantly reduced (0.81-fold that seen in the sham group) on day 3, most reduced (0.55-fold that seen in the sham group) on day 7, and still low (0.75-fold that seen in the sham group) on day 14 (Figure 2E and F). To investigate the role of PRMT6 in chronic pain, we also examined PRMT6 expression in the DRG of CCI and BCP animal models. PRMT6 was also markedly decreased in the CCI and BCP group compared to with the sham group (Figure 1 in Additional file 2). Immunohistochemistry analysis revealed a 21.85% (P < 0.01) reduction in the number of PRMT6-labeled neurons in the SNI group 7 days postoperatively compared with the sham group (Figure 2G–I). We also assessed changes in the types of cells expressing PRMT6 in the SNI mice. The results showed that approximately 75% of β-tubulin III-positive neurons are labeled for PRMT6 in the sham mice. SNI surgery decreased the colocalization of β-tubulin III with PRMT6 to 51%. The distribution proportion of different neuron types did not change significantly. Approximately 48% of PRMT6- labeled neurons were positive for CGRP, 32% for IB4 and 23% for NF200 in Sham mice. While in SNI mice, approximately 52% of PRMT6-labeled neurons were positive for CGRP, 33% for IB4 and 20% for NF200 (Figure 2A and B in Additional file 2). These results indicate that PRMT6 protein and mRNA expression levels in the DRG correspond specifically to peripheral nerve injury, suggesting that PRMT6 could potentially be involved in neuropathic pain.
Figure 4 | Prmt6–/– mice exhibit normal innervation patterns and sensory neuron numbers.
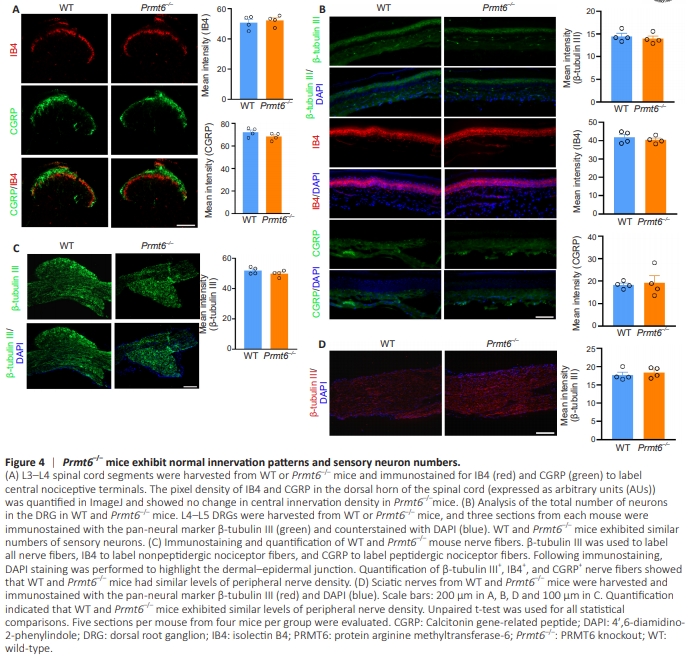
To explore whether the observed hyperalgesia occurred because of changes to the neural architecture in the transgenic mice, we immunostained L3–L4 spinal cord segments, DRG neurons, hindlimb skin, and sciatic nerve fibers to detect neuronal markers and examine innervation patterns and sensory nerve counts. Mice lacking Prmt6 showed no alterations in central or peripheral innervation density or total DRG neuron count (Figure 4A–D).
Figure 5 | Effect of Prmt6-siRNA microinjection into the DRG on nociceptive thresholds in na?ve mice.
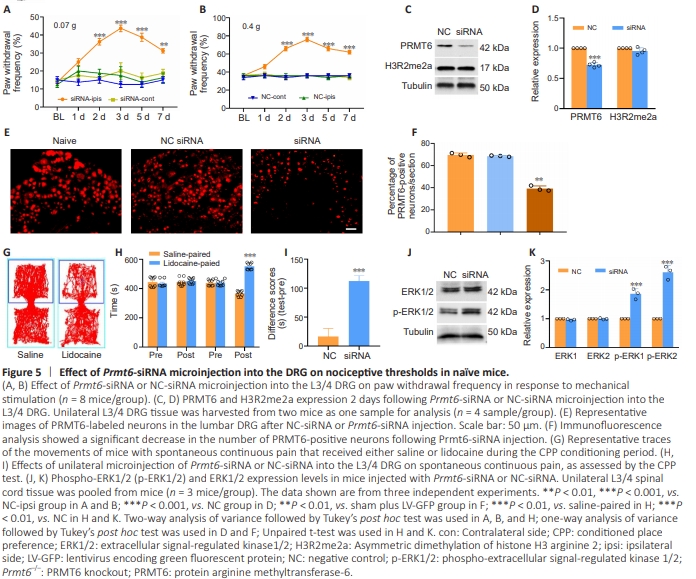
Figure 6 | Effects of PRMT6 overexpression on neuropathic pain induced by SNI.
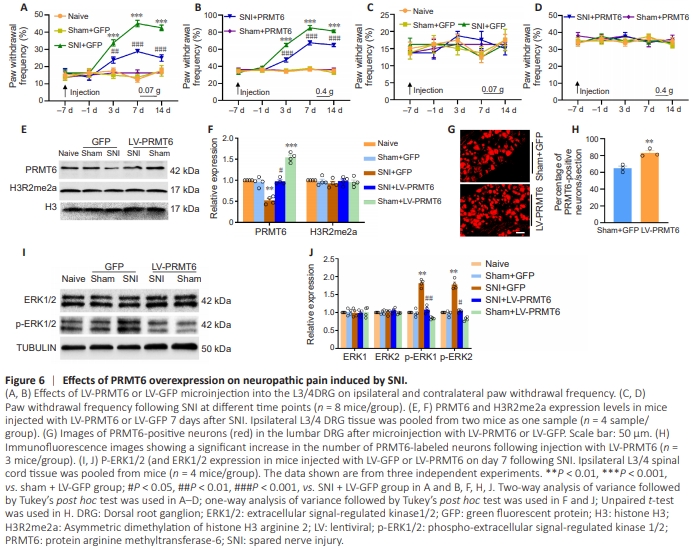
Intrathecal injection of the PRMT6 inhibitor EPZ020411 may lack anatomical specificity. Therefore, to determine whether PRMT6 in the DRG alone alters nociceptive thresholds, we microinjected Prmt6-specific siRNA unilaterally into the L3/4 DRG of naive mice. The experimental design is shown in Figure 5A in Additional file 2. Behaviorally, microinjection of Prmt6-siRNA, but not a negative siRNA (NC-siRNA), resulted in mechanical allodynia, as evidenced by an ipsilateral increase in the frequency of paw withdrawal in response to mechanical stimulation (Figure 5A and B). As expected, PRMT6 expression levels in the ipsilateral L3/4 DRG of Prmt6-siRNA–microinjected mice were reduced compared with NC-siRNA–microinjected mice 2 days post-siRNA microinjection (Figure 5C–F). Next, we performed a conditioned place preference test to assess whether knocking down Prmt6 expression in the DRG induced spontaneous continuous pain. Prmt6-siRNA– microinjected mice spent more time in the lidocainepaired chamber (Figure 5G–I), suggesting that they were experiencing stimulus-independent pain. As expected, NCsiRNA-microinjected mice demonstrated no significant preference for either the saline- or the lidocaine-paired chamber (Figure 5G–I). Furthermore, compared with the NCsiRNA group, p-ERK1/2 levels were significantly increased in the ipsilateral L3/4 dorsal horn in the Prmt6-siRNA group (Figure 5J and K). These findings suggest that decreasing PRMT6 expression in the DRG evoked spontaneous pain hypersensitivity, a typical clinical manifestation of neuropathic pain, despite the absence of nerve damage. Protein arginine methyltransferase-6 overexpression in dorsal root ganglions alleviates spared nerve injury–induced pain hypersensitivity We then examined whether PRMT6 overexpression affects SNI-induced neuropathic pain. The experimental design is shown in Figure 5B in Additional file 2. Seven days before SNI or sham surgery, lentiviruses expressing full-length Prmt6 mRNA (LV-PRMT6) or a control (LV-GFP) were microinjected unilaterally into the L3/4 DRGs. Ipsilateral L3/4 DRGs were harvested to assess PRMT6 expression 14 days following sham or SNI surgery. Consistent with our previous observations, SNI significantly increased the frequency of ipsilateral paw withdrawal in response to mechanical stimuli from day 3 to day 14 following surgery compared with baseline (Figure 6A and B). In contrast, compared with LV-GFP–treated SNI mice, LV-PRMT6–treated SNI mice showed a lower ipsilateral paw withdrawal frequency from day 3 to day 14 post-SNI (Figure 6A and B). LV-PRMT6 microinjection did not alter the basal paw withdrawal response of sham mice to mechanical stimulation (Figure 6A and B). Neither LV-PRMT6 nor LV-GFP affected either contralateral basal responses (Figure 6C and D) or motor function (Table 2).expression was significantly reduced in the ipsilateral L3/4 DRG on day 14 post-SNI compared with mice microinjected with LV-GFP and subjected to sham surgery (Figure 6E and F). This pattern of decreased expression was not observed in mice microinjected with LV-PRMT6 and subjected to SNI (Figure 6E and F). Furthermore, the expression basal PRMT6 expression level in in the ipsilateral L3/4 DRG was significantly increased in mice microinjected with LV-PRMT6 and subjected to sham surgery (Figure 6E and F). Considering that PRMT6 catalyzes H3R2 methylation and represses gene transcription, we next assessed H3R2me2a levels following microinjection of LV-GFP or LV-PRMT6 into the DRG. No marked alteration in H3R2me2a expression was detected in either group (Figure 6E and F), indicating that the effect of PRMT6 on peripheral neuropathic pain is independent of H3R2 methylation. Furthermore, compared with the Sham + GFP group, the number of PRMT6-positive cells was significantly increased following microinjection ofLV-PRMT6 (1.35-fold; Figure 6G and H) into the L3/4 DRG, as detected by immunofluorescence staining. The role of DRG PRMT6 in neuropathic pain was further supported by increased expression of biomarkers of cell activity in the spinal cord dorsal horn. Levels of p-ERK1/2, a marker of neuronal hyperactivation, were significantly reduced in the ipsilateral L3/4 dorsal horn of SNI mice microinjected with LV-PRMT6 compared with those microinjected with LVGFP at 7 days post-microinjection (Figure 6I and J). These data indicated that PRMT6 overexpression in the DRG alleviates the pain hypersensitivity induced by SNI.
Figure 8 | hnRNP-F and PRMT6 colocalize in DRG neurons.
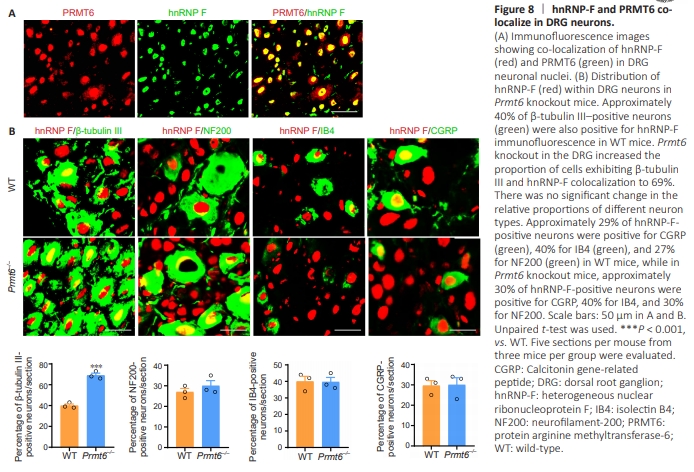
To explore the relationship between hnRNP-F and PRMT6 in the genesis and development of neuropathic pain, hnRNP-F and PRMT6 co-localization in DRG neurons was assessed by immunofluorescence staining (Figure 8A). Approximately 40% of β-tubulin III-positive neurons were also positive for hnRNP-F. Prmt6 knockout in the DRG increased the proportion of cells expressing both β-tubulin III and hnRNP-F to 69% (Figure 8B). The proportions of different neuron types did not change significantly. Consistently, approximately 29% of hnRNP-F-positive neurons were positive for CGRP, 40% were positive for IB4, and 27% were positive for NF200 in wild-type mice (Figure 8B), while in Prmt6–/– mice approximately 30% of hnRNP-F-labeled neurons were positive for CGRP, 40% were positive for IB4, and 30% were positive for NF200 (Figure 8B). These findings further verify that hnRNP-F is upregulated following Prmt6 knockout in the DRG. These results suggested that PRMT6 plays a critical role in the occurrence and development of neuropathic pain via hnRNP-F