脑损伤
-
Figure 1 | bFGF-HAC gel promotes tissue regeneration and functional recovery after motor cortex injury in mice.
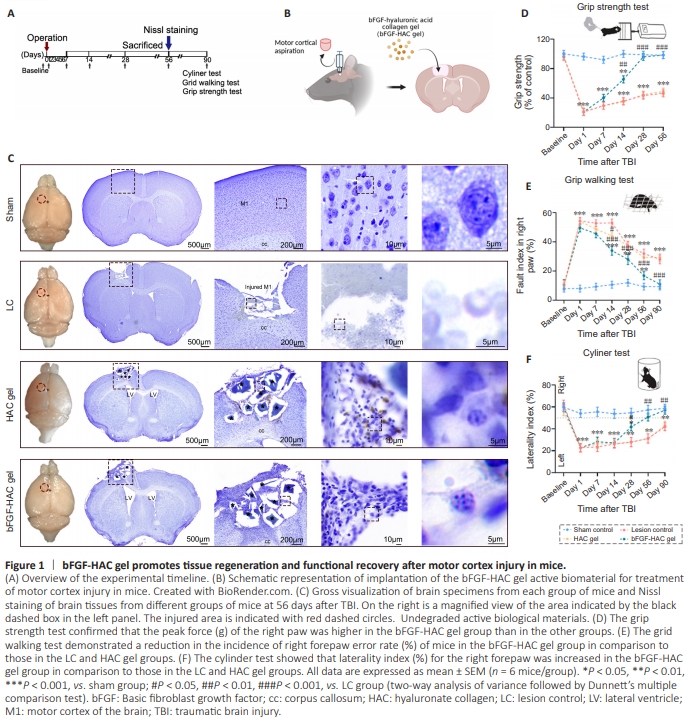
Adult mice were subjected to aspiration injury of the cortex using a homemade biological tissue cutter (Mo et al., 2010; Hao et al., 2017). A piece of brain tissue (1.5 mm × 1.5 mm × 1 mm) was aspirated from the motor cortex (representative area of the forelimb motor cortex) (Tennant et al., 2011), while the corpus callosum was left intact (Figure 1A and B). On the 56th day after the operation, the potential regeneration of cortical injury areas was evaluated in each experimental group using Nissl staining (Figure 1C). In the HAC gel group, a small amount of newly formed tissue and hyaluronic acid collagen gel that had not yet been fully degraded were observed at the cortical injury site. In contrast, the injured area was still clearly visible in the LC group (Figure 1C). Although the injured area in the bFGF-HAC gel group did not appear to have a normal cortical structure, numerous Nissl-positive cells were observed in the injured area (Figure 1C). These results indicated that different interventions resulted in varying degrees of regeneration following cortex injury. The cortical motor regions of the brain play a crucial role in skilled locomotion (Sato et al., 2020). To evaluate the effect of the different interventions on recovery of motor function, forelimb motor function was assessed using the grip strength test, grid test, and cylinder test (Figure 1A and D–F). The results indicated that right forelimb motor function in the LC group was significantly lower than that in the sham group and remained so for 3 months following injury. These findings suggest that injury to the left cortical region can result in long-term impairment of right forelimb motor function, such as grasping ability, motor coordination, and limb preference. Notably, the grip strength in the bFGF-HAC gel group was significantly higher than that in the LC group (P = 0.000; Figure 1D) 14 days postoperatively and recovered to a level similar to that seen in the sham group at 28 days postoperatively (bFGF-HAC gel group vs. sham group, P = 0.998; Figure 1D). In the bFGF-HAC gel group, grid test performance began to improve 14 days postoperatively, exhibiting a significantly lower error rate compared with the LC group (P = 0.000; Figure 1E). By 90 days postoperatively, performance had recovered to normal levels, with no significant difference compared with the sham group (bFGF-HAC gel group vs. sham group, P = 0.647; Figure 1E). At 28 days postoperatively, mice in the bFGF-HAC gel group displayed a significantly higher laterality index for using their right forepaw to support their body weight compared with the LC group (bFGF-HAC gel group vs. LC group, P = 0.001; Figure 1F). Use of the right paw reached a level comparable to that seen in the sham group by 56 days postoperatively (bFGF-HAC gel group vs. sham group, P = 0.556), and this recovery remained stable until 90 days postoperatively (Figure 1F). These findings suggest that, after motor cortex injury, bFGF-HAC gel transplantation significantly increased the laterality index of mice using their right forepaw to support their body weight, significantly decreased the error rate of the right forepaw during spontaneous walking, and restored right forelimb muscle strength to a level comparable to the sham group. These results indicate that bFGF-HAC gel can significantly improve impaired forelimb motor performance in mice after cortical injury.
Figure 2 | bFGF-HAC gel promotes neoplastic cell proliferation at the injury site after TBI.
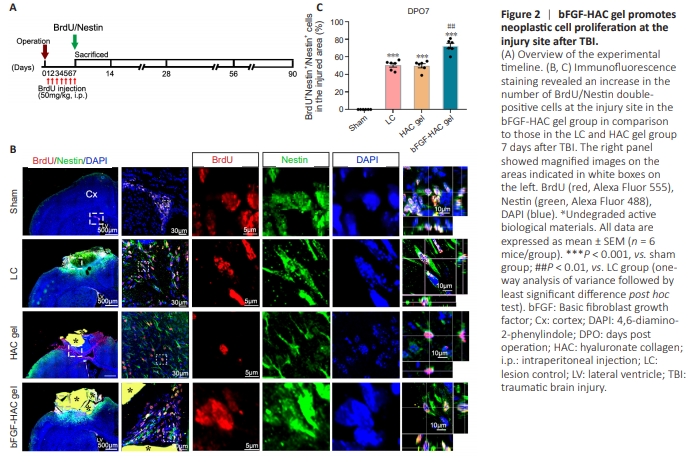
To investigate the effects of different interventions on neural stem progenitor cells in the SVZ and DG following cortical injury, BrdU/ Nestin immunofluorescence double-labeling was performed. At 7 days after the operation, no BrdU/Nestin double-positive cells were observed in the corresponding cortex area of the sham group, although some were observed in the ipsilateral SVZ region (Figure 2A). In the LC and HAC gel groups, BrdU/Nestin doublepositive cells, indicating activated NSCs, were observed in the injured cortex (Figure 2B). Compared with the LC and HAC gel groups, significantly more newborn BrdU/Nestin double-positive cells were detected at the injury site in the bFGF-HAC gel group (bFGF-HAC gel group vs. LC group, P = 0.0002; bFGF-HAC gel group vs. HAC gel group, P = 0.0088; Figure 2B and C). These findings suggest that bFGF-HAC gel implantation may activate ipsilateral NSCs and facilitate their migration to the injured area.
Figure 3 | bFGF-HAC gel promotes newborn neuron regeneration at the site of injury after TBI.
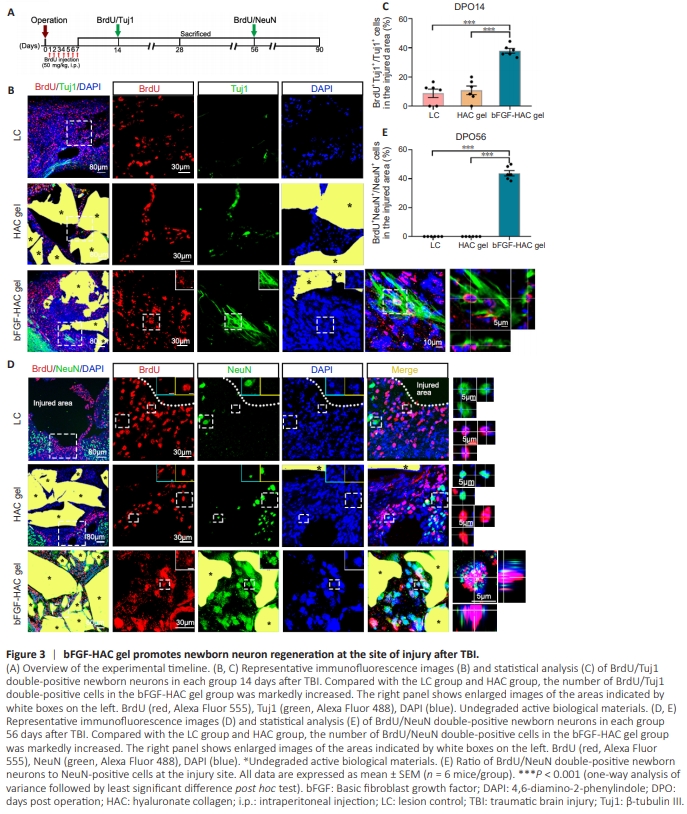
In the LC and HAC gel groups, a limited number of newborn Tuj1- positive cells were observed at the junction of the injured area and normal brain tissue 14 days after the operation (approximately 16% Tuj1-positive cells in the LC group were BrdU-positive; approximately 21% Tuj1-positive cells in the HAC gel group were BrdU-positive; Figure 3A–C). By 56 days postoperatively, no BrdU/ NeuN double-positive cells were detected at the injury site in the LC and HAC gel groups (Figure 3D and E), indicating that the BrdUpositive cells generated in these groups at 7 days postoperatively did not further develop into mature neurons. However, at 14 days after bFGF-HAC gel transplantation, a significant number of Tuj1- positive cells were observed within and surrounding the injured area, and 38% of these Tuj1-positive cells were also BrdU-positive (Figure 3B and C). The number of BrdU/Tuj1 double-positive cells at the injury site continued to increase in the bFGF-HAC gel group over time, and many of these cells were still observed at the injury site up to 90 days postoperatively. Additionally, some of the newborn neurons exhibited the characteristic cell morphology of cortical pyramidal neurons, featuring elongated cell protrusions and distinct branching (Additional Figure 1A–C). Six months after cortical injury, newborn BrdU/Tuj1 double-positive neurons were still observed, and their morphology closely resembled that of normal pyramidal neurons, with numerous branches (Additional Figure 1D). These findings suggest that bFGF-HAC gel implantation following cortical injury can consistently and robustly promote the generation of newborn neurons in the injured area. At 56 days postoperatively, only a few NeuN-positive cells were observed at the injury site in the LC group and HAC gel group. Conversely, a significant number of NeuN-positive cells were detected at the cortical injury site in the bFGF-HAC gel group, and approximately 44% of these NeuN-positive cells were also BrdU-positive (Figure 3D and E). These findings suggest that bFGF-HAC gel implantation can stimulate the generation of mature neurons in the injured cortical area. At 90 days post–cortical injury, MAP2/BrdU doublepositive cells were observed at the injury site in the bFGF-HAC gel group (Additional Figure 1E), further confirming that bFGF-HAC gel implantation may facilitate the sustained production of mature neurons at the injured area.
Figure 4 | bFGF-HAC gel encourages newborn neuron differentiation to reconstruct the original layered architecture of the cerebral cortex in the injured area after TBI.

The laminar organization of the neocortex is a fundamental feature of the mammalian central nervous system (D’Souza and Burkhalter, 2017; Carton-Leclercq et al., 2023). The motor cortex of the brain exhibits a distinct laminar structure and well-defined cellular configurations. FOXP2 serves as a marker for cortico– thalamic projection neurons in layer VI (Druart et al., 2020), CTIP2 is a marker for subcortical projection neurons in layer V of the cerebral cortex (Harb et al., 2022), and SATB2 identifies cortico– cortical (ipsilateral and corpus callosum) projection neurons distributed across layers II to V (Saito et al., 2011). Together, these diverse cortical neurons form a network that is a crucial component of the cerebral cortex. To characterize the distribution of newborn neurons within the injured cortical area, we stained brain sections with antibodies to FOXP2, CTIP2, SATB2, and BrdU (Figure 4A). At 90 days postoperatively, FOXP2/BrdU doublepositive cells, CTIP2/BrdU double-positive cells, and SATB2/BrdU double-positive cells were observed in the injured cortex of the bFGF-HAC gel group (Figure 4B). These results suggest that bFGF-HAC gel may facilitate the remodeling of layers II to VI with newborn cortical neurons. Approximately 70% to 80% of neurons in the mouse brain were excitatory glutamatergic projection neurons, while only 15% to 20% were inhibitory GABAergic nonpyramidal interneurons (Hendry et al., 1987; Beaulieu, 1993; Tremblay et al., 2016). At 90 days postoperatively, newborn neurons in the injured cortex of the bFGF-HAC gel group includedboth BrdU/Tuj1/vGluT1 triple-positive glutamatergic neurons (Figure 4C) and BrdU/Tuj1/GAD67 triple-positive GABAergic neurons (Figure 4C). These findings suggest that bFGF-HAC gel can promote the generation of new glutamatergic and GABAergic neurons in the injured cortex, thereby reconstructing the laminar cellular architecture of the original cerebral cortex.
Figure 5 | bFGF-HAC gel promotes synapse formation by newborn neurons at the injury site after TBI.
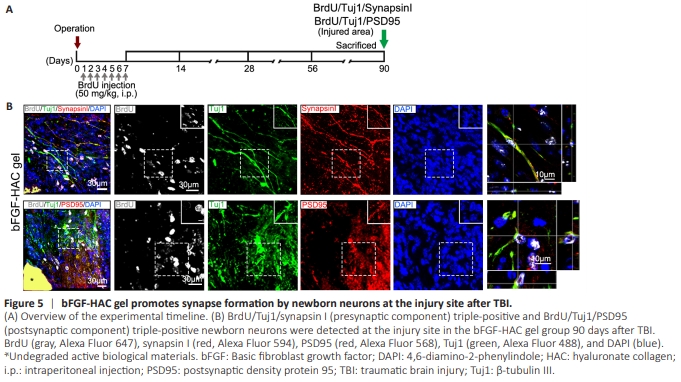
Given that the adult mammalian brain is composed of various laminar structures containing specific cells types, and of the cortex is organized into layers I to VI (Ferland et al., 2003; Arlotta et al., 2005; Molyneaux et al., 2007; Harris et al., 2019), repairing disrupted neuronal networks following cortical injury poses a significant challenge. Synapses, which are crucial for transmitting information among neurons, are fundamental for neuronal function (Di Maio et al., 2021). To investigate whether synaptic connections are established among newborn neurons and between newborn neurons and host neurons after cortical injury, we stained brain sections with synapsin I, a marker for presynaptic components (Zhang et al., 2022), and PSD95, a marker for postsynaptic components (Shi et al., 2023) to observe co-labeling with newborn neurons (Figure 5A). At 90 days post– cortical injury, a substantial number of Tuj1-positive newborn neurons and their protrusions were observed at the injury site in the bFGF-HAC gel group. These neurons expressed both synapsin I and PSD95, suggesting that they had formed synapselike connections with other neurons (Figure 5B). These findings suggest that bFGF-HAC gel can induce newborn neurons to receive synaptic inputs or send synaptic outputs.
Figure 6 | bFGF-HAC gel promotes integration of newborn neurons into existing neural circuits at the injury site after TBI.
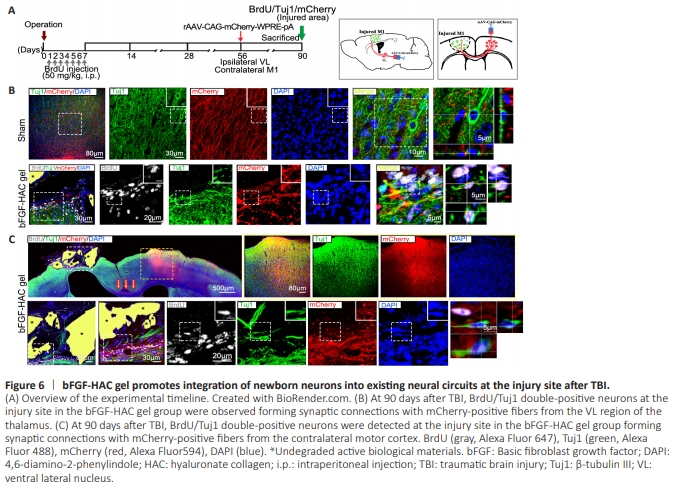
The M1 contains axonal projections from the VL thalamus (Strick and Sterling, 1974; Kuramoto et al., 2009; Guo et al., 2018). At 56 days post–cortical injury, the viral vector rAAV-CAG-mCherryWPRE-pA encoding an anterograde tracer was injected into the VL ipsilateral to the injury site. Four weeks later, Tuj1-positive neurons in the M1 area of the cortex were observed forming synapse-like structures with mCherry-positive nerve fibers originating from the VL in the sham group. Similarly, BrdU/Tuj1 double-positive newborn neurons in the injured cortex area developed synaptic-like structures with mCherry-positive nerve fibers from the VL in the bFGF-HAC gel group (Figure 6A and B). Callosal projection neurons reside in the superior cortical layers (II and III), where their axons traverse the midline to the contralateral cerebral hemisphere, facilitating integration of information between homologous regions of both hemispheres (Molyneaux et al., 2007; Lodato et al., 2015). At 56 days post– cortical injury, the viral vector rAAV-CAG-mCherry-WPRE-pA encoding an anterograde tracer was injected into the upper layers of the normal brain motor cortex (II and III) contralateral to the injury site. Four weeks later, BrdU/Tuj1 double-positive newborn neurons in the injured cortex of the bFGF-HAC gel group had formed synapse-like structures with mCherry-positive fibers originating from the contralateral brain motor cortex (Figure 6A and C). These findings indicate that the newborn neurons in the injured cortex had reestablished information transmission pathways with both the contralateral cerebral cortex and the ipsilateral thalamus, helping to integrate neural circuits.
Figure 7 | bFGF-HAC gel promotes integration of newborn neurons into the CST circuit of at the injury site after TBI.

Corticospinal motor neurons, a specialized subset of neurons in the cerebral cortex, collect, integrate, translate, and transmit excitatory and inhibitory cortical inputs as a unified message to distal spinal cord targets (Jara et al., 2014). We next asked whether cortical layer IV to V newborn neurons at the injury sitein the bFGF-HAC gel group could re-establish CST neural networks with the distal spinal cord after surgery. To address this, EGFPlabeled and mRuby-labeled retrograde virus PRV (capable of crossing multiple synapses) (Chen et al., 2018; Yang et al., 2021) was injected into the right and left biceps brachii of experimental mice respectively 85 days after injury. Five days later, PRV-EGFPand/or PRV-mRuby–positive motor cortex neurons were observed in the sham group (Figure 7A–C). Although neurons labeled with PRV-EGFP and PRV-mRuby were present in the contralateral cortex, they were rarely observed at the injury site in the LC group. However, in the bFGF-HAC gel group, BrdU/Tuj1 double-positive neurons in the injured cortex area co-labeled with both PRV-EGFP and PRV-mRuby (Figure 7C). These findings suggest that newborn neurons in the injured area were integrated into the motor circuit.
Figure 8 | bFGF-HAC gel promotes Nestin+ NSC activation and ectopic migration.
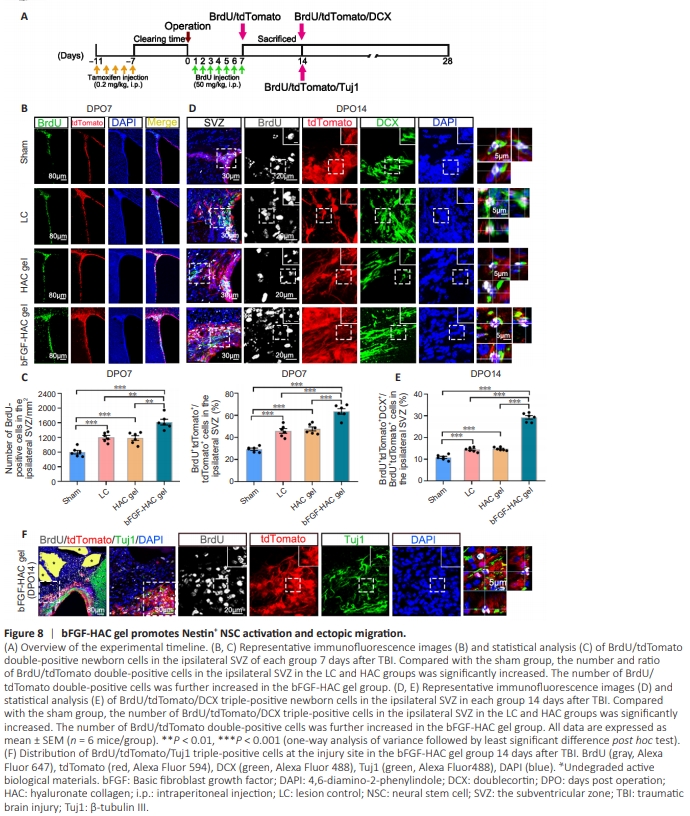
To further elucidate the impact of bFGF-HAC gel on endogenous neurogenesis following cortical injury and to investigate the origin of the newborn neurons observed at the site of cortical injury, we injected Nestin CreERT2:Rosa26-tdTomato transgenic lineage-tracing mice with tamoxifen intraperitoneally for 5 consecutive days prior to cortical injury. One week after the injection (Additional Figure 2A–D), the mice were subjected to motor cortex injury, as previously described (Bai et al., 2023). At 7 days post–cortical injury, a limited number of Nestin/tdTomato double-positive cells were observed in the ipsilateral SVZ and DG in both the LC and HAC gel groups, and some of these cells were also BrdU-positive (Figure 8A–C and Additional Figure 3A–C). However, in the bFGFHAC gel group, a significant increase in Nestin/tdTomato doublepositive cells was noted at the injury site in the ipsilateral SVZ and DG. Notably, 64% of the tdTomato-positive cells in the SVZ and 53% of the tdTomato-positive cells in the DG were also BrdUpositive (Figure 8B, C and Additional Figure 3B, C). No significant differences were observed in the number of BrdU-positive cells in the contralateral SVZ or DG among the groups (Additional Figures 2E and F, 3B and C, and 4). These findings suggest that the bFGFHAC gel effectively activated endogenous Nestin-positive NSCs in the ipsilateral SVZ and DG of the injured area. To determine whether activated tdTomato-positive NSCs originating in the SVZ and DG could migrate to the injured area of the cortex, brain sections were subjected to immunocytochemistry double labeling for BrdU and DCX, markers of migrating NPCs (Couillard-Despres et al., 2005). In the sham group, DCX/tdTomato double-positive cells were primarily localized to the ipsilateral SVZ adjacent to the injured area at 14 days post–cortical injury, with cell protrusions extending in the direction of the rostral migration stream. In the LC and HAC gel groups, DCX-positive cells in the SVZ migrated toward the site of injury, and a small proportion of these cells were also BrdUand tdTomato-positive (Figure 8D and E). In the bFGF-HAC gel group, a significant increase in the number of DCX-positive cells was observed in the ipsilateral SVZ at the injury site. These cells exhibited a shuttle-shaped soma and protrusions extending towards the site of injury. Notably, the majority of these DCXpositive cells migrated from the SVZ to the injured cortex, and 29% of them were also BrdU- and tdTomato-positive (Figure 8D and E). However, interestingly, there was no significant difference in the number of BrdU/tdTomato/DCX triple-positive cells in the DG among the groups (Additional Figure 4A–C). Although a number of BrdU-positive cells were also observed in the ipsilateral DG of the bFGF-HAC gel group, the DCX-positive neurons did not migrate from the DG. Additionally, they were not tdTomato-positive and did not demonstrate a trend towards migrating to the injured area following motor cortex injury (Additional Figure 4B). These findings suggest that the SVZ is the most potential niche for generating newborn neurons following cortical injury, and that Nestin-positive cells in this region can be further activated by the implanted active biomaterial.To further investigate whether tdTomato/DCX double-positive cells differentiate into neurons at the site of cortical injury, multiplexed immunofluorescence labeling was performed. The results demonstrated the presence of tdTomato/BrdU/Tuj1 triplepositive cells in the injured area at 14 and 28 days post–cortical injury. At 14 days post-injury, some tdTomato-positive cells in the vicinity of the corpus callosum in the bFGF-HAC gel group were observed to also be BrdU- and Tuj1-positive (Figure 8F and Additional Figure 5). By 28 days post-injury, an increased number of tdTomato-positive cells at the site of cortical injury were found to also be BrdU- and Tuj1-positive (Additional Figure 6A and B). Collectively, these findings suggest that motor cortex injury alone can activate Nestin-positive NSCs in the ipsilateral SVZ to a limited extent, prompting them to respond to the injury. However, very few of these activated NSCs migrate out of the SVZ. In contrast, bFGF-HAC gel transplantation significantly enhanced the NSC mobilization in the ipsilateral SVZ in response to injury.