脑损伤
-
Figure 1 | The interaction between blood vessels and NSCs in the telencephalon of adult zebrafish.
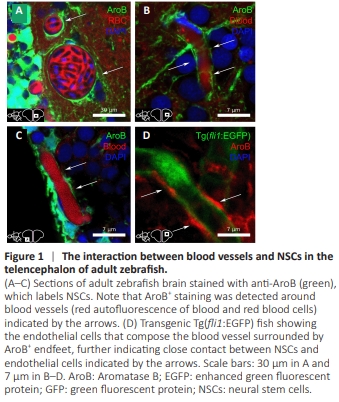
In mice, under physiological conditions, NSCs interact with blood vessels during development and adulthood, and several growth factors and chemokines secreted by vessels can regulate NSC activity (Ottone et al., 2014; Karakatsani et al., 2019). In the brain of adult zebrafish, we demonstrated the close link between NSCs and blood vessels (Figure 1), consistent with previous studies (Diotel et al., 2018; Sulliman et al., 2021; Pellegrini et al., 2023). AroB immunostaining revealed NSC endfeet around and along the brain vasculature in the telencephalon (Figure 1A–C). We also noted this direct contact when performing AroB immunostaining in Tg(fli1:EGFP) zebrafish, which allows labeling of endothelial cells (Figure 1D). Together, these data show a close association between NSC endfeet and blood vessels, suggesting that endothelial factors and factors supplied by the blood flow could impact NSC activity.
Figure 2 | Regenerative angiogenesis following telencephalic stab wound injury.

In pathological conditions such as traumatic brain injury, blood vessel rupture and cell death occur, leading to the activation of regenerative processes (Xiong et al., 2010; Sawada et al., 2014). Indeed, a decrease in the oxygen level within the tissue and the need to replenish the damaged area leads to the formation of new blood vessels. To investigate this process in teleosts, we subjected Tg(fli1:EGFP x mpeg1.1:mCherry) zebrafish to telencephalic stab wound injury and sacrificed them at different times after the lesion. We analyzed several angiogenic parameters, including the blood vessel surface, the GFP transgene intensity, and the blood vessel diameter. We compared these parameters with the contralateral hemisphere, which serves as an internal control. They were upregulated between 2 and 5 dpl in the stab-wounded hemisphere compared with the contralateral hemisphere (Figure 2A–E). The blood vessel surface (fli1:EGFP+ area) showed a 1.8-fold increase at 3 dpl before decreasing to the basal level at around 14 dpl, indicating a transient increase in the number of blood vessels and/or the surface covered by blood vessels (Figure 2A and C). The blood vessel diameter and the GFP transgene intensity around the lesion site also increased, peaking at 2 and 4 dpl, respectively. Interestingly, although the diameter decreased from 3 dpl, it remained significantly higher than in the contralateral hemisphere even at 21 dpl (Figure 2B, D, and E). Consequently, a telencephalic lesion activates regenerative angiogenesis, as denoted by the increased number of blood vessels, their vasodilation, and the upregulation of specific genes/ transgene.
Figure 4 | Regenerative neurogenesis following telencephalic stab wound injury.
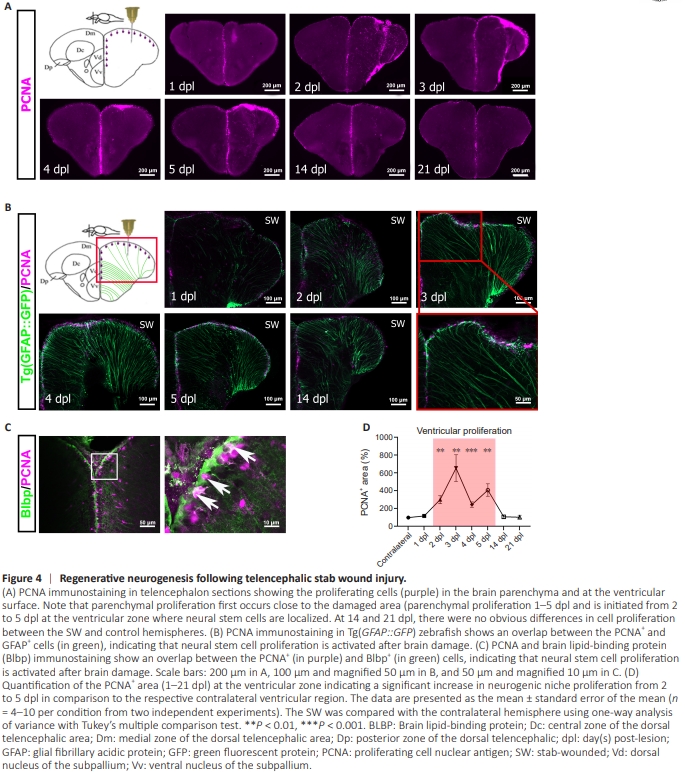
At the ventricular surface, the proliferative cells are known to correspond mostly to NSCs and to a lesser extent to neuroblasts (committed progenitors) (M?rz et al., 2010; Diotel et al., 2020). Moreover, ventricular proliferation is known to increase after a stab wound injury, notably in the telencephalon, to replace the dead neurons (Dash et al., 2001; Chirumamilla et al., 2002; M?rz et al., 2011; Diotel et al., 2020). Thus, we aimed to correlate regenerative angiogenesis with NSC/progenitor cell proliferation. For that, we counted the number of proliferative cells (PCNA+ ) in the stab-wounded and contralateral hemispheres at the ventricular surface. Similarly to regenerative angiogenesis, NSC/ progenitor cell proliferation increased following a stab wound injury, with the highest level from 2 to 5 dpl compared with the contralateral hemisphere (Figure 4A–D). At 14 dpl, ventricular proliferation had returned to the basal level (Figure 4D). To confirm that the ventricular PCNA+ cells were indeed NSCs/progenitor cells, we used transgenic Tg(GFAP::GFP) zebrafish (Figure 4B) and performed BLBP immunostaining (Figure 4C). GFAP and BLBP correspond to well-characterized NSC markers in zebrafish (M?rz et al., 2011; Rodriguez Viales et al., 2015; Diotel et al., 2016). Our results indicate that a telencephalic stab wound injury in zebrafish activates regenerative neurogenesis.
Figure 5 | Increased vegfaa and vegfbb gene expression following a telencephalic stab wound injury.
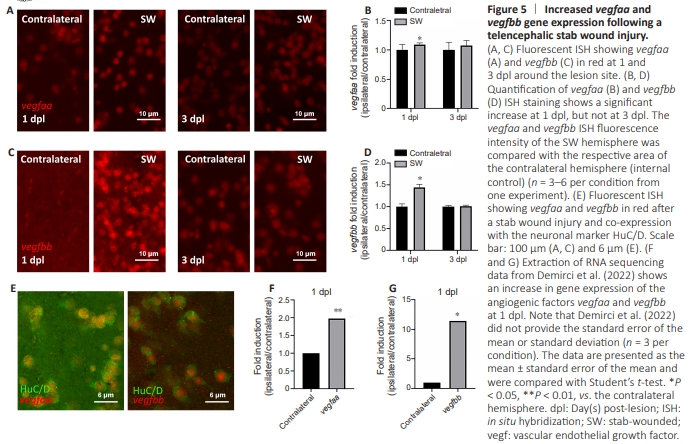
Given that Vegf signaling is important for angiogenesis, we wondered if vegf expression is modulated after stab wound injury of the telencephalon. By performing fluorescent ISH against vegfaa and vegfbb at 1 and 3 dpl, we observed a transient increase in the expression of both genes at 1 dpl close to the damage site, with a return to the basal level at 3 dpl (Figure 5A– D). Next, we extracted RNA sequencing data from (Demirci et al., 2022) and confirmed vegfaa and vegfbb upregulation at 1 dpl (Figure 5F and G). Finally, by performing HuC/D immunostaining after vegfaa and vegfbb ISH, we demonstrated that the cells overexpressing these genes after injury correspond to neurons (Figure 5E). Although the reason for such a transient increase remains to be explored, we speculate that certain factors and signals that occur during brain injury, such as hypoxia/hypoxiainducible factors, inflammatory factors, and growth factors, could transiently increase vegf gene expression, as has already been demonstrated in mammals.
Figure 6 | Regenerative angiogenesis and neurogenesis are disrupted after tivozanib injection.
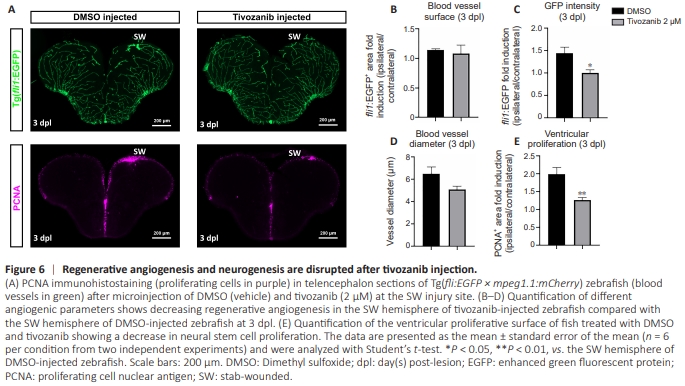
To investigate the impact of angiogenesis on brain regeneration, we microinjected tivozanib at the lesion site. We decided to perform a local microinjection using tivozanib at a concentration of 2 μM at the site of the lesion for several reasons: (i) tivozanib is more potent than vatalanib (total inhibition of developmental angiogenesis at 1 and 5 μM, respectively); (ii) tivozanib is known to inhibit VEGFR1, VEGFR2, and VEGFR3 (Nakamura et al., 2006), whereas vatalanib mainly inhibits VEGFR1 and VEGFR2; and (iii) the injected concentration was doubled compared with treated larvae to compensate for the dilution of the inhibitor following injection. We analyzed angiogenesis and neurogenesis at 3 dpl and injection, considering that both processes were increased at this time point. After an intraparenchymal injection of tivozanib at the lesion site, several parameters of angiogenesis such as the blood vessel diameter and surface tended to decrease, and the GFP transgene intensity that was significantly lower when compared to the vehicle (DMSO)-injected fish (Figure 6A–D).In parallel, we noticed a significant decrease in NSC/progenitor cell proliferation around the ventricular area at 3 dpl (Figure 6A and E). These data showed that inhibition of the VEGF signaling pathway is important for both regenerative angiogenesis and neurogenesis during brain repair. Note that we obtained similar results with the co-injection of tivozanib and vatalanib (1 μM of each), strengthening these findings (data not shown).
Figure 7 | Analysis of regenerative angiogenesis and neurogenesis after VEGF165 injection.
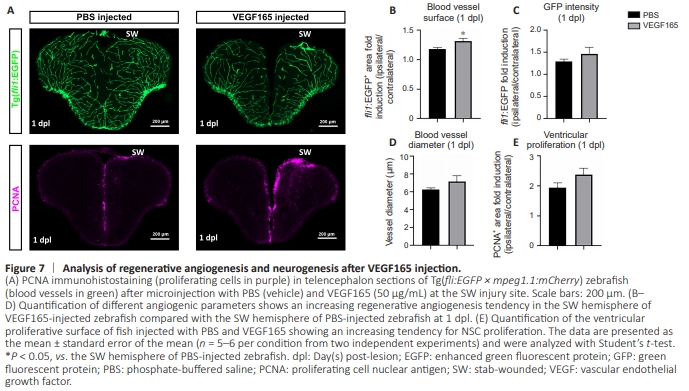
To reinforce the possible role of VEGF signaling in these regenerative processes, we took the opposite approach by activating this signaling pathway through VEGF microinjection at the lesion site. It resulted in a significant increase in the surface area covered by blood vessels from 1 dpl, and in an increasing trend in GFP transgene intensity and the blood vessel diameter compared with the PBS-injected control (Figure 7A–D). VEGF injection also induced a trend toward increased ventricular proliferation, as denoted by PCNA immunostaining at 1 dpl (Figure 7A and E). Based on these findings, promoting angiogenesis through VEGF signaling seems to impact regenerative neurogenesis positively in our experimental conditions, even if this effect is moderate.
Figure 8 | DEX incubation leads to a decrease in angiogenesis and brain regeneration.
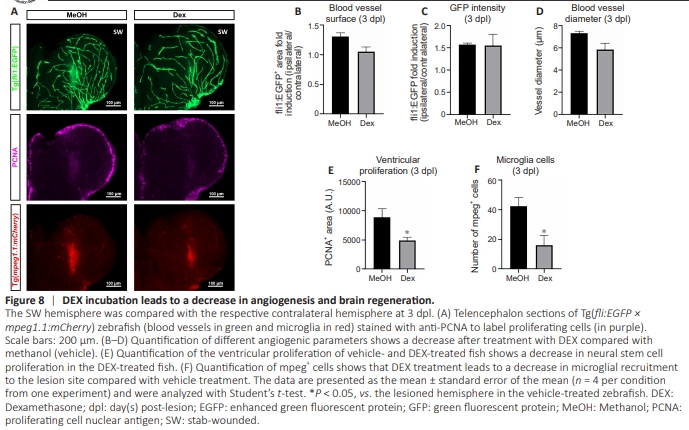
Figure 9 | Regenerative angiogenesis and neurogenesis are disrupted after clodronate liposomes injection.
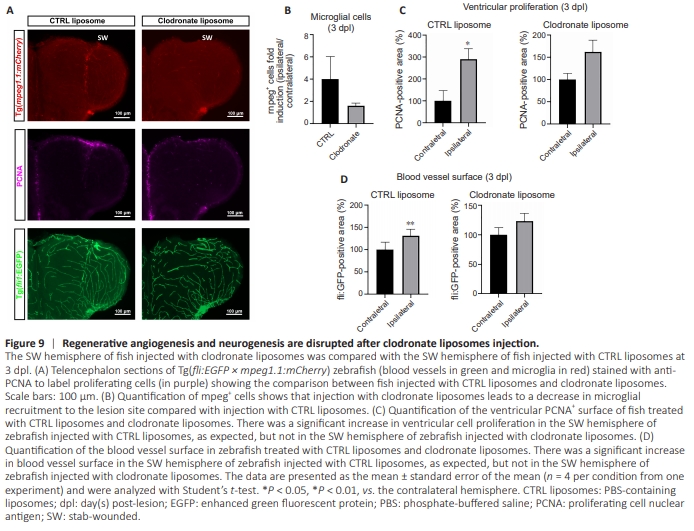
It is well known that inflammation and microglia-mediated inflammation after brain injury in zebrafish are key actors of regenerative neurogenesis (Kyritsis et al., 2012; Palsamy et al., 2023). However, the role of inflammation in regenerative angiogenesis has not yet been elucidated in zebrafish during telencephalic injury. We explored this phenomenon by incubating adult zebrafish in water containing DEX dissolved in methanol (15 mg/L) as described previously to inhibit inflammation and microglial recruitment (Kyritsis et al., 2012; Palsamy et al., 2023). To avoid compensatory mechanisms that might arise after long-term exposure to DEX, we pretreated the zebrafish for 1 day prior to stabbing, and maintained DEX treatment until the end of the experimental procedure (3 days after stabbing). We treated control fish similarly but with the vehicle (0.1% methanol, final concentration). As expected, DEX treatment significantly decreased recruited microglia/immune cells (mpeg+ cells) at the injury site (Figure 8A and F) and blunted reactive neurogenesis (Figure 8A and E), consistent with previous studies (Kyritsis et al., 2012; Caldwell et al., 2019). We investigated angiogenesis parameters to determine the impact of such anti-inflammatory treatment on regenerative angiogenesis. There was a significant decrease in the blood vessel surface and diameter following DEX treatment in the stab-wounded hemisphere compared with the vehicle-treated hemisphere (Figure 8A–D). Interestingly, DEX treatment did not have an impact on the blood vessel surface, the fli:EGFP transgene intensity, and the blood vessel parameters in the contralateral hemisphere compared with the vehicletreated fish (Additional Figure 2). So far, it appears that the modulation of inflammatory processes during brain injury impacts regenerative angiogenesis in our experimental conditions (Figure 8A–E). We next evaluated the precise role of microglial activation in regenerative angiogenesis by using clodronate-containing liposomes to deplete microglia during the development of the brain lesion (Palsamy et al., 2023). In control fish microinjected with PBS-containing liposomes, we observed the expected increase in ventricular cell proliferation and regenerative angiogenesis (Figure 9). In contrast, fish injected with clodronate-containing liposomes show reduced microglial recruitment to the injured hemisphere (Figure 9A and B) and a lack of injury-induced proliferation in the ventricular zone (Figure 9A and C), consistent with the findings reported by Palsamy et al. (2023). These treated fish also showed no injury-induced angiogenesis (Figure 9A and D). Taken together, these experiments demonstrated the role of inflammation, and specifically the recruitment of microglia, in promoting injuryinduced angiogenesis.
Figure 10 | Microglial recruitment is regulated by Vegf signaling.
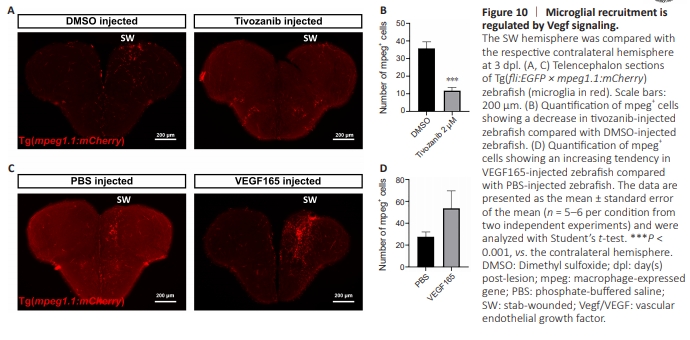
Figure 11 | Microglial and endothelial cell proliferation following brain lesion and modulation of Vegf signaling.

In the literature, experimental data show that microglia express Vegf receptors and that Vegf signaling contributes to their activation (Forstreuter et al., 2002; Couturier et al., 2014; Uemura et al., 2021). To further appreciate the possible role of VEGF signaling on the recruitment of microglia after a telencephalic stab wound injury in zebrafish, we counted the number of mpeg+ cells after an injection of tivozanib or VEGF in Tg(fli1:EGFP × mpeg1.1:mCherry) zebrafish. Compared with the vehicle, tivozanib microinjection decreased the number of mpeg+ cells in the injured hemisphere (Figure 10A–D) and decreased microglia proliferation (Figure 11). Consistently, VEGF microinjection increased the number of recruited microglia compared with zebrafish treated with the corresponding vehicle (Figure 10A– D), as well as the number of proliferative microglia (Figure 11). These findings are consistent with the previous data and suggest a potential mechanism of interaction between regenerative angiogenesis and neurogenesis through direct or indirect activation of microglia orchestrated by Vegf signaling.