脊髓损伤
-
Figure 2 | CerS5, GSDMD, and GSDMD-N expression levels are significantly higher 14 days after SCI.
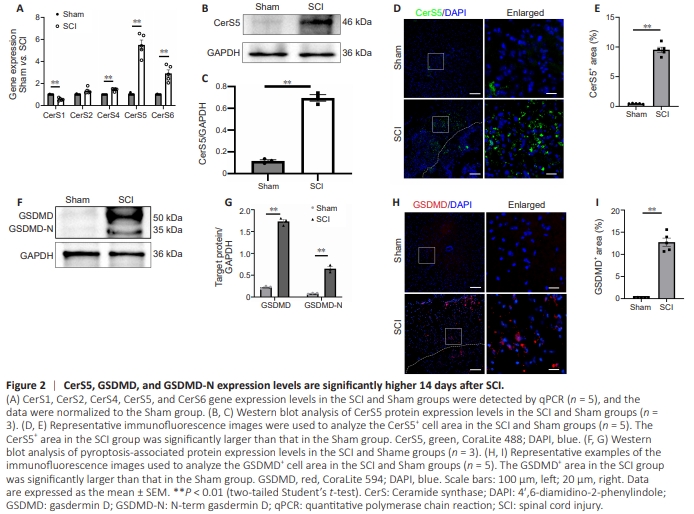
Next, we performed qPCR analysis to investigate the expression of CerS family members in mice at the site of SCI and in the corresponding position in the spinal cords of mice in the Sham group, 14 dpi, to confirm that sphingolipid metabolism was altered following SCI. The results revealed no statistically significant difference in CerS2 expression between the SCI and Sham groups (P > 0.05). However, a 0.6-fold decrease in CerS1 expression and 1.5-fold, 5.55- fold, and 2.9-fold increases in CerS4, CerS5, and CerS6 expression were observed in the SCI group compared with the Sham group, respectively (Figure 2A). To further validate the change in CerS5 protein expression level, WB assay and immunofluorescence staining were performed on spinal cord tissue collected 14 days after SCI. The results were consistent with the findings from scRNA-seq analysis and qPCR, confirming the upregulation of CerS5 expression after SCI (Figure 2B and C). Notably, the number of CerS5+ cells was significantly increased at the injury site in mice from the SCI group compared with at the corresponding area of the spinal cord in mice from the Sham group (P < 0.01; Figure 2D and E). Cell pyroptosis is increased after spinal cord injury Next, we investigated the expression and tissue localization of the pyroptosis-associated protein GSDMD at the injury site 14 days after SCI and in corresponding spinal segments in the Sham group to confirm that pyroptosis occurred after SCI. When compared with the Sham group, western blot analysis showed a significant increase in the expression of both GSDMD and its N-terminal fragment, GSDMD-N in the SCI group (P < 0.01; Figure 2F and G). Additionally, immunohistochemical staining demonstrated a significant increase in the number of GSDMD-positive cells at the injury site compared with the Sham group (P < 0.01; Figure 2H and I). These findings were in line with the results from the scRNAseq dataset analysis.
Figure 3 | CerS5 knockdown inhibits LPS-induced BV2 cell pyroptosis in vitro.

Although we observed elevated levels of microglial sphingolipid metabolism and cellular pyroptosis after SCI and validated these findings in a mouse model of SCI, the underlying relationship between sphingolipid metabolism and cellular pyroptosis remained unclear. To address this topic, we first established a neuroinflammatory model by stimulating BV2 cells with LPS to mimic microglial activation after SCI. Subsequently, we inhibited CerS5 expression of using an antisense RNA (Figure 3A and B), which led to a significant decrease in the expression of GSDMD, a protein associated with cell death (Sborgi et al., 2016; Figure 3C and D).
Figure 4 | CerS5 knockdown inhibits microglial pyroptosis in the spinal cord in a mouse model of SCI.

The central nervous system exhibits a high degree of complexity, with various cell types interacting in an intricate and orderly manner (Lénárt et al., 2024). Consequently, the microglial changes observed after SCI are likely to be influenced by the local tissue environment. To determine whether modulating CerS5 expression would inhibit microglial pyroptosis in a mouse model of SCI, we inhibited CerS5 expression in the injured spinal cord by intrathecal injection of si.CerS5 (Figure 4A and B). Subsequently, we examined GSDMD expression at the site of SCI to assess the impact of CerS5 inhibition on microglial pyroptosis. Both immunofluorescence and western blot results were consistent with in the results from the in vitro experiments, showing that inhibition of CerS5 expression resulted in a reduction in the number of GSDMD+ cells and in GSDMD expression compared with the SCI group (Figure 4C and D).
Figure 5 | CerS5 knockdown inhibits activation of the classical NLRP3 pyroptosis pathway after SCI.

Our earlier results showed that CerS5 expression levels in microglia after SCI were closely related to pyroptosis. However, the molecular mechanism linking these two phenomena remained unclear. To investigate the molecular mechanism by which CerS5 inhibits pyroptosis, we examined activation of the classical NLRP3 cell death pathway after SCI. Immunofluorescence staining results showed a decrease in the number of NLRP3+ cells after inhibition of CerS5 expression in vivo (Figure 5A). Furthermore, western blot analysis of NLRP3, ASC, Caspase1, cleaved-Caspase1, IL1β, and IL-18 expression at the injury site showed that the expression of all NLRP3 pyroptosis classical pathway-related proteins was reduced after inhibition of CerS5 expression (Figure 5B). These results demonstrate that CerS5 inhibits microglial pyroptosis after SCI by inhibiting activation of the NLRP3 signaling pathway.
Figure 6 | CerS5 knockdown inhibits Pla2g7/p50 expression in the spinal cord after SCI.
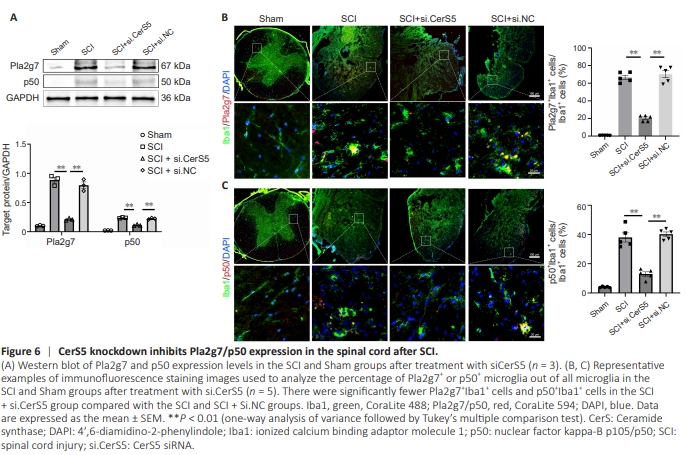
In addition to pyroptosis-associated proteins, the scRNAseq dataset analysis results that Pla2g7 and p50 were highly expressed in MG1 microglia after SCI (Figure 1F). Therefore, we used a mouse model of SCI to investigate whether inhibiting CerS5 expression in vivo would affect Pla2g7 and p50 expression. Western blot analysis showed that inhibiting CerS5 effectively reversed the upregulation in Pla2g7 and p50 expression seen after SCI (Figure 6A). Immunofluorescence staining confirmed that there was a significant increase in the number of Pla2g7+ and p50+ microglia in the SCI group compared with the Sham group (P < 0.01), and that inhibiting CerS5 expression reversed this effect (Figure 6B and C).
Figure 7 | Pla2g7 is essential for mediating the regulatory effects of CerS5 on pyroptosis.
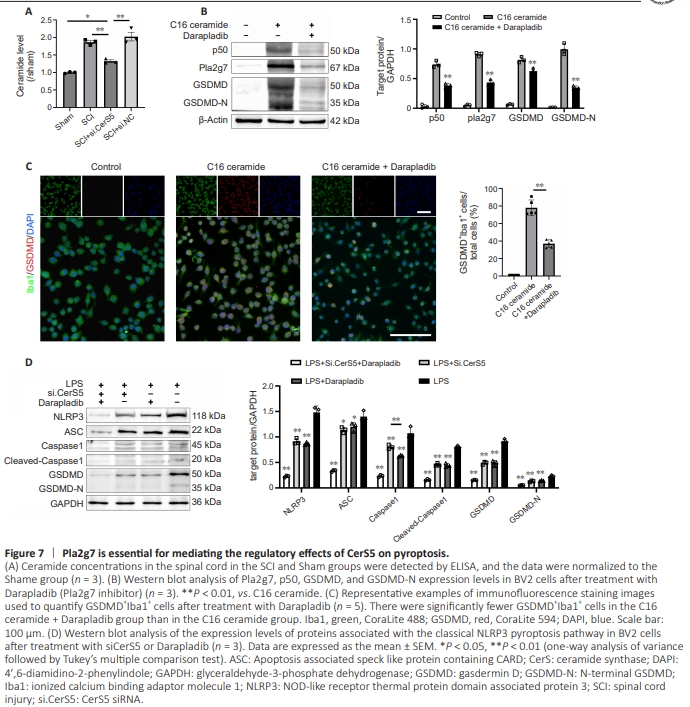
Figure 8 | Darapladib inhibits microglial pyroptosis in the spinal cord after SCI.
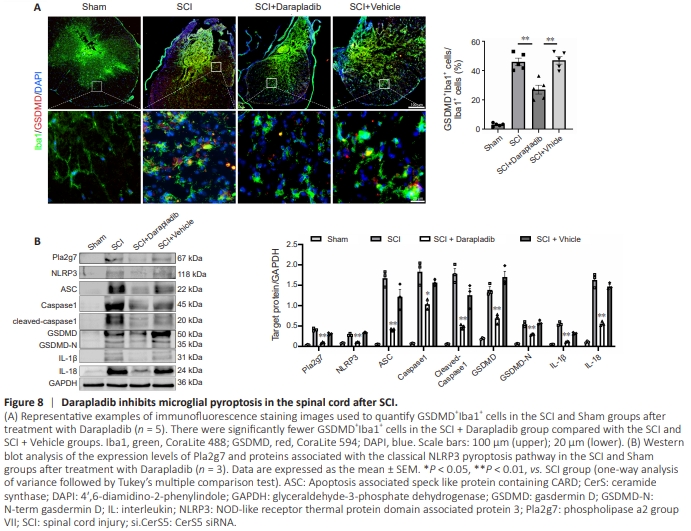
While we demonstrated that inhibiting CerS5 expression after SCI can affect expression of the pyroptosis-associated proteinsPla2g7 and p50, the underlying regulatory relationship remained unclear. To further investigate the relationships among these molecules, we first examined the expression of C16 ceramide, a product of CerS5 metabolic pathway, after SCI. The results showed that the C16 ceramide content of spinal cord tissue was significantly elevated after SCI. However, inhibiting CerS5 expression in vivo resulted in a marked reduction in C16 ceramide levels (Figure 7A). To simulate this phenomenon in vitro, we stimulated BV2 cells with C16 ceramide and then treated the cells with the Pla2g7 inhibitor Darapladib. The results demonstrate that treating BV2 cells with C16 ceramide induced high levels of p50, Pla2g7, and GSDMD expression in microglia after injury. Notably, inhibiting Pla2g7 expression suppressed p50 and GSDMD expression (Figure 7B). Immunofluorescence staining results further confirmed that treatment with the Pla2g7 inhibitor attenuated the ability of C16 ceramide, a downstream product of the CerS5 metabolic pathway, to promote microglial pyroptosis (Figure 7C). To determine whether Pla2g7 is essential for CerS5-induced pyroptosis, we compared the effects of CerS5 and Pla2g7 inhibition on pyroptosis in BV2 cells stimulated with LPS. The results showed that both interventions effectively suppressed the expression of pyroptosis-associated proteins, thus confirming that Pla2g7 plays a crucial role in regulating the relationship between CerS5 and pyroptosis (Figure 7D). To further validate the role of Pla2g7 in the relationship between CerS5 and pyroptosis in vivo, we treated a mouse model of SCI with Darapladib and found that inhibiting Pla2g7 in vivo resulted in a significant reduction in the number of GSDMD+ microglia (Figure 8A), as well as decreased expression of NLRP3, ASC, Caspase1, Cleaved-Caspase1, GSDMD, IL-1β, and IL-18 (Figure 8B). These findings demonstrate that inhibiting Pla2g7 in vivo effectively attenuated microglial pyroptosis after SCI.
Figure 9 | CerS5 knockdown inhibits neuronal apoptosis in the spinal cord after SCI.

We investigated whether inhibiting CerS5 expression would play a neuroprotective role in the context of SCI. To address this, we first stimulated normal BV2 cells and BV2 cells in which CerS5 expression was inhibited with LPS. We then incubated PC12 cells in conditioned media from both cell types and observed the effects on PC12 cell apoptosis. We found that inhibiting CerS5 expression in BV2 cells reduced the ability of the conditioned BV2 cell medium to induce apoptosis in PC12 cells (Figure 9A). Using a mouse model of SCI we found that inhibiting CerS5 expression markedly reduced neuronal apoptosis in vivo (Figure 9B). Furthermore, hematoxylin-eosin staining demonstrated that inhibiting CerS5 in vivo effectively reduced the area of the injured region (Figure 9C). Finally, the BMS scores indicated that inhibiting CerS5 expression improved motor recovery in mice after SCI (Figure 9D).