脑损伤
-
Figure 1 | Characterization of hNSCs and hNSC-EVs.

NSCs were successfully isolated from fetal brain tissue and cultured. During the primary passage, there was a noticeable amount of cell debris in the growth medium, and the NSC neurospheres were loose and irregular in shape. By the third passage, NSC neurospheres were of uniform size and regular shape (Figure 1A). Considering that the transcription factor PAX6 and class VI intermediate filament protein (Nestin) are important markers of NSC, the hNSC characteristics wereanalyzed by detecting the presence of Nestin and PAX6 (Figure 1B). Upon induction, hNSC differentiated into neurons (Tuj1) and neuroglia (GFAP) (Figure 1C), indicating that we successfully isolated and cultured hNSCs. Subsequently, the hNSC-EVs were characterized by NTA, TEM, and WB. NTA showed that the hNSC-EVs were homogenous, with diameters between 100 and 150 nm (Figure 1D). TEM further showed that the hNSC-EVs exhibited a double-layered membrane and a cup-like structure (Figure 1E). Expression of hNSC-EVs markers (CD9, CD63, TSG101, and calnexin) was detected by western blotting (WB) (Figure 1F). Next, to confirm that the hNSC-EVs could be internalized by cells, we labeled hNSCEVs with the red fluorescent dye PKH-26 and incubated the labeled hNSC-EVs with different target cells, including human umbilical vein endothelial cells, mouse hippocampal neuronal cells (HT22), and mouse microglial cells (BV2). Internalization of the fluorescently labeled hNSC-EVs was confirmed by microscopic analysis (Figure 1G). Taken together, these results indicate that the isolated and cultured hNSC and the hNSCEVs they produced exhibited typical characteristics and could be used in subsequent experiments.
Figure 2 | Validation of hNSC-EV efficacy in vitro.
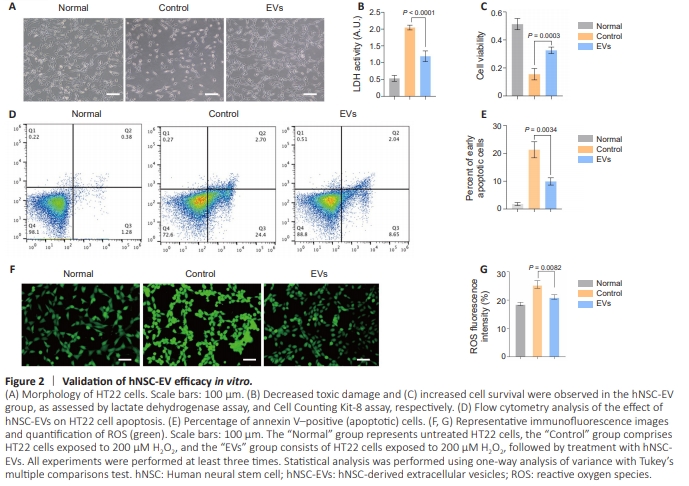
Next, this study utilized an H2O2-induced oxidative stress model to simulate the pathological environment of ischemic stroke, examining the mechanism of action of hNSC-EVs in neuronal repair processes under stressful conditions in vitro. We observed apoptosis-related morphological changes in HT22 cells induced by H2O2 under a microscope, and these morphological changes were partially reversed by treatment with hNSC-EVs (Figure 2A). Furthermore, lactate dehydrogenase (Figure 2B) and CCK8 (Figure 2C) assays showed that hNSC-EVs reduced the damage caused by H2O2-induced oxidative stress, confirming that the hNSCEVs promoted neuronal repair, reduced toxic damage, and increased cell survival in the context of oxidative stress. Flow cytometry analysis showed a decreased proportion of apoptotic cells in the hNSC-EV group compared with the control group, confirming the role of hNSC-EVs in reversing neuronal apoptosis (P < 0.01; Figure 2D and E). In addition, lower ROS levels were detected in the hNSC-EV group compared with the control group, confirming the antioxidant capacity of hNSC-EVs (P < 0.01; Figure 2F and G).
Figure 4 | hNSC-EVs improved neurological function after ischemic stroke in vivo.

We further investigated the therapeutic efficacy of hNSCEVs in a rat MCAO model. First, we evaluated infarct volume 24 hours after MCAO. The infarct volume was significantly larger in rats from the control group than in rats from the hNSC-EV group, suggesting that hNSC-EVs alleviate cerebral ischemic injury in vivo (Figure 4A). Furthermore, treatment with hNSC-EVs inhibited neuronal apoptosis in vivo after stroke (Figure 4B). To investigate the effects of hNSC-EVs on the PI3K/AKT/mTOR pathway following ischemic stroke, WB was performed to examine PI3K, p-PI3K, mTOR, p-mTOR, AKT, and p-AKT expression. The level of PI3K (p-PI3K/PI3K), AKT (p-AKT/AKT), and mTOR (p-mTOR/mTOR) in the hNSC-EV group was significantly higher than that in the control group (P < 0.01; Figure 4C). These results demonstrate that hNSC-EV treatment may exert neuroprotective effects by regulating the PI3K/AKT/mTOR pathway in a rat model of ischemic stroke. Next, we evaluated recovery of neurological function in MCAO rats using a series of behavioral tests. The modified neurological severity score showed that neural damage in the hNSC-EV group was reduced (Figure 4D). In the gait test, a skewed gait, prolonged swaying time, and shortened walking distance were observed in the control group; however, hNSCEV treatment returned these gait parameters to normal levels (Figure 4E). In the open field test, the total distance traveled by rats in the control group was significantly shortened, and the hNSC-EV group exhibited results similar to those seen in the sham group; however, the total distance traveled by rats in the sham group and hNSC-EV group tended to stabilize on day 7 (Figure 4F). Moreover, evaluation of neural function via the grip test (Figure 4G), cylinder test (Figure 4H), and rotarod test (Figure 4I) on days 3, 5, 7, 10, and 14 showed that the neurological function of rats in the hNSC-EV group improved significantly, demonstrating the effectiveness of hNSC-EVs in treating ischemic stroke.
Figure 6 | Assessment of hNSCEV toxicity.
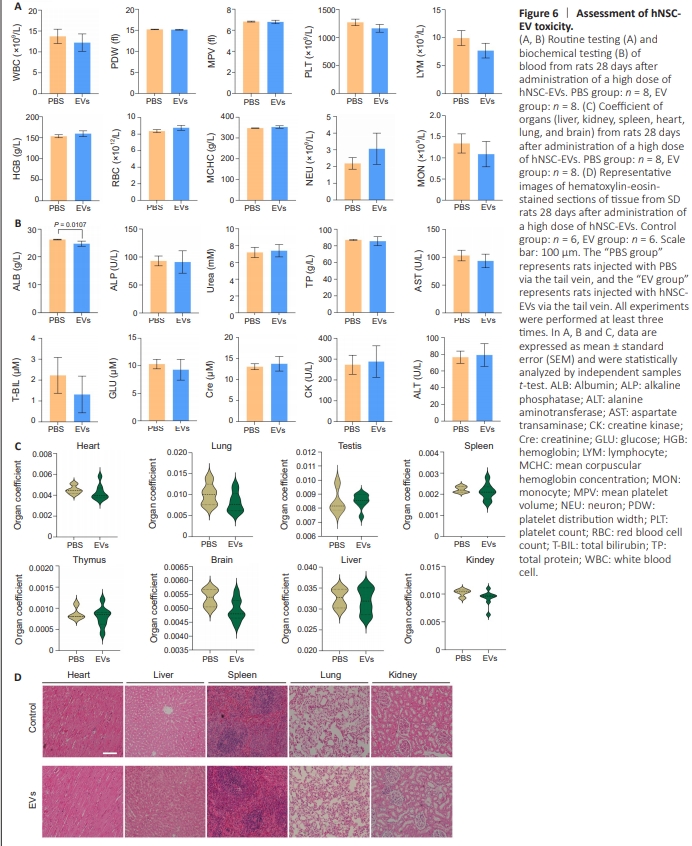
To explore the possibility of using hNSC-EVs in a clinicalcontext, we assessed their immunoreactivity and toxicity. First, we evaluated the immunoreactivity associated with intravenous administration of PBS and a high dose (five times the therapeutic dose) of hNSC-EVs in SD rats by evaluating the peripheral blood lymphocyte subtypes of the two groups on days 1 (Figure 5A) and 14 (Figure 5B). Differences in CD3+ CD161+ NKT cell counts were observed between the two groups on day 1, which could have resulted from the xenotransplantation with hNSC-EVs; however, these differences were not present on day 14. Other indicators such as proportions of lymphocytes, T cells, CD4+ T cells, CD8+ T cells, CD4+ CD8+ T cells, CD45RA+ B cells and CD161+ NK cells out of total leukocytes did not change significantly, suggesting that hNSC-EVs were not immunoreactive. Next, we evaluated the potential toxicity associated with intravenous administration of high-dose hNSC-EVs for 28 days in our rat model. Hematological and chemical analyses of the blood (Figure 6A and B) and organ coefficients (Figure 6C), as well as indepth histopathological evaluation of several different tissues (Figure 6D), did not reveal any hNSC-EV treatment-associated abnormalities. Thus, the immunoreactivity and toxicity results indicate that the hNSC-EVs had a good safety profile in vivo.