脑损伤
-
Figure 3 | Glutamatergic CYLD deficiency enhances neuronal activation in the BLA and decreases the slope of current-voltage curve of the BLA pyramidal neurons.

Optically stimulating BLA pyramidal neurons as an US produced tone-cued fear memory, suggesting a key role for the activity of BLA pyramidal neurons in modulating cued fear (Johansen et al., 2010; Yiu et al., 2014). Thus, we next determined whether exaggerated cued fear expression in male cKO mice was related to altered neuronal activation, neuronal excitability, or synaptic transmission in the BLA. Initially, we examined the number of activated cells in the BLA of cKO and WT mice at 90 minutes after fear retrieval by labeling cells with c-Fos (Figure 3A). The results suggested a significant increase in the number of c-Fos+ cells in the BLA of cKO mice following the tone-cued test (P = 0.035; Figure 3B). This indicates enhanced neuronal activation in the BLA of cKO mice after cued fear memory recall, and prompted us to further identify any changes in neuronal excitability, intrinsic properties, or synaptic transmission in BLA pyramidal neurons of cKO mice using whole-cell patch-clamp experiments. Biocytin labeling confirmed the pyramidal morphology of patched cells in the BLA (Figure 3C). Pyramidal neurons from cKO mice showed decreased input resistance (P = 0.031; Table 1) and decreased slopes of current–voltage curves (P < 0.0001; Figure 3D and E), implicating higher excitability of BLA pyramidal neurons in cKO mice (Bjorefeldt et al., 2015; Hong et al., 2016; Tonomura and Gu, 2022). Other electrophysiological properties were unchanged (Table 1).
Figure 6 | Deletion of CYLD in glutamatergic neurons leads to over-activation of microglia in the BLA.

Figure 7 | Glutamatergic neuron-specific knockout of CYLD does not cause microglial activation in CA1 and CA3 regions of the hippocampus.
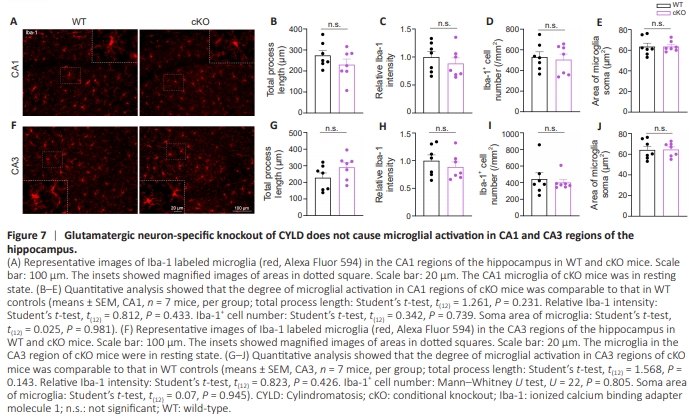
It has become abundantly clear that the function and development of synapses are greatly affected by neural– immune interactions (Zajicek and Yao, 2021). Numerous studies have observed that reactive microglia are critically involved in the functional brain changes and behavioral alterations (reflecting fear memory) associated with PTSD (Hori and Kim, 2019; Li et al., 2021b; Lee et al., 2022; Zhu et al., 2023). Our previous work uncovered a positive function of CYLD in preventing over-activation of microglia and neuroinflammation (Han et al., 2020). Consequently, we sought to identify from a different perspective another mechanism that might account for augmented fear expression of male cKO mice, namely, the regulatory role of CYLD in microglial function. To gain initial insight into the relationship between CYLD and microglial activation, we determined whether resident microglia in the BLA were activated in naive Cyld–/– mice. A significant increase in the number of Iba-1+ cells was observed (P = 0.009), indicating that microglia in the BLA of male Cyld–/– mice were in an over-activated state (Figure 6A– D). Consistently, selective deletion of Cyld in glutamatergic neurons also evoked over-activation of microglia in the BLA of cKO mice. Although the intensity of Iba-1 as well as the number and soma size of Iba-1+ cells did not change (Figure 6E–H), we observed significantly decreased total process lengths (P = 0.0012; Figure 6I) and branch points of microglia in the BLA of cKO mice (P < 0.0001; Figure 6J). Moreover, we labeled Iba-1+ cells with CD68, a maker of activated microglia, and performed 3D reconstructions of microglia. Significant increases in CD68 puncta (P = 0.036; Figure 6K) and volume (P < 0.0001; Figure 6L) per microglia were observed in the BLA of cKO mice, further confirming over-activation of microglia. Notably, in line with unchanged CFM, which is hippocampusdependent (Baldi and Bucherelli, 2015; Chaaya et al., 2018), no significant difference occurred in microglial morphology in the hippocampus of cKO mice (Figure 7A–J). Altogether, these results show that CYLD is essential to the physiological activities of glutamatergic neurons and microglia, and provides insight into the biological mechanisms of exaggerated cued fear memory.