脊髓损伤
-
Figure 1 | Tryp inhibits microglia-derived inflammation through shifting polarization to the M2 phenotype.
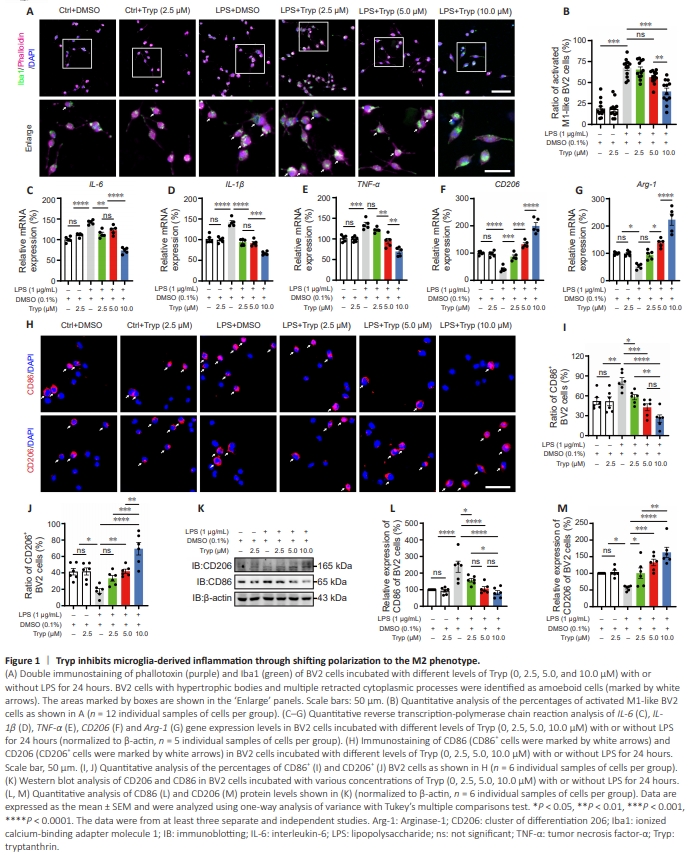
To examine the effects of Tryp on microglia-derived inflammation and its potential mechanisms, the LPS-induced BV2 cell model was used to mimic the characteristics of microglia located in the spinal cord lesion area after SCI in vitro (Liu et al., 2021b). CCK8 assay showed that Tryp was not cytotoxic at concentrations lower than 10.0 μM within 24 hours (Additional Figure 3A). Additionally, incubation with LPS (1 μg/mL) did not cause cytotoxic effects on BV2 cells (Additional Figure 3B). We next evaluated the phenotypes of BV2 cells based on their morphology by coimmunostaining of Iba1 and phalloidin, a marker of F-actin. As shown in Figure 1A and B, LPS significantly induced BV2 cells to transform into an amoeboid-like morphology, the M1-like phenotype. Tryp inhibited the morphological changes of LPSinduced BV2 cells in a dose-dependent manner. Furthermore, qRTPCR analysis showed that the mRNA expression of M1 microglial markers (IL-6, IL-1β, and TNF-α) were significantly upregulated by LPS treatment, and were significantly suppressed by incubation with Tryp (Figure 1C–E). The mRNA levels of M2 microglial markers (CD206 and Arg-1) were significantly upregulated after Tryp treatment (Figure 1F and G), which indicated that Tryp promoted microglial polarization to the M2 phenotype. Immunostaining and western blot analyses of CD86 and CD206 further showed that Tryp promoted the shift of microglia from the M1 to M2 phenotype in a dose-dependent manner (Figure 1H–M). Moreover, we evaluated the protein levels of the apoptosis-related markers Bcl-2 and Bax (Murphy et al., 2000, Wei et al., 2001) in BV2 cells after Tryp treatment. As shown in Additional Figure 4A–C, no significant differences were found in the expression of Bcl-2 or Bax between groups, suggesting that Tryp treatment did not induce the apoptosis of BV2 cells. Taken together, these results suggested that microglia-derived inflammation was inhibited by Tryp through modulating microglial polarization to the M2 phenotype rather than apoptosis.
Figure 2 | Tryp promotes the M2 polarization of microglia through the cGAS/STING/NF-κB pathway.

It was recently shown that LPS-induced cellular dyshomeostasis may lead to cytoplasmic dsDNA accumulation, which then activates the cGAS-STING pathway to shift microglia to the M1 phenotype (Li et al., 2022a). Thus, we next examined whether Tryp promoted microglia (BV2 cell) M2 polarization by inhibiting cGAS-STING signaling in vitro. Western blot analyses showed that cGAS and p-STING were significantly upregulated in BV2 cells treated with LPS + DMSO (LD-treated group), compared withthose in the Ctrl group (P = 0.0372). The upregulation of cGAS and p-STING was significantly inhibited by Tryp treatment (LTtreated group), when compared with the LD-treated group (P = 0.0002; Figure 2A–C). Furthermore, the levels of both p-p65 and p-IκBα were significantly reduced in the LT-treated group compared with those in the LD-treated group (Figure 2D–H). These results suggested that the cGAS/STING/NF-κB pathway was inhibited in Tryp-treated BV2 cells. To confirm whether Tryp promoted microglial M2 polarization through inhibiting the cGAS-STING pathway, SR-717, an agonist of STING, was used to activate STING signaling (Chin et al., 2020). As shown in Additional Figure 5, the effective concentration of SR-717 was approximately 20 μΜ, and this concentration was selected for the following experiments. Western blot analyses showed that cGAS, p-STING, p-p65, and p-IκBα were significantly upregulated in BV2 cells treated with LPS + Tryp and SR-717 (LT + SR-717 group; Figure 2I–P). The qRT-PCR (Figure 2Q–U) and immunostaining analyses (Figure 2V–W) further showed that SR717 partially reversed Tryp-induced M2 polarization of BV2 cells. Consistent with the results of BV2 cells, immunostaining analyses of primary cultured microglia also showed that Tryp promoted M1-to-M2 polarization, and the effect of Tryp was partially reversed by SR-717 (Figure 2V and X). Taken together, these results suggested that Tryp promoted microglial polarization to the M2 subtype through inhibiting the cGAS/STING/NF-κB pathway.
Figure 3 | Tryp shifts microglia from M1 to M2 phenotype after spinal cord injury by targeting the cGAS/STING/NF-κB pathway.

To investigate whether targeting the cGAS/STING/NF-κB pathway by Tryp would polarize microglia to the M2 phenotype in vivo, we established the clip-compressive SCI model (Additional Figure 6). As resident innate immune cells in spinal cord, microglia play a vital role in the development of secondary inflammation after SCI (Devanney et al., 2020). As shown in Figure 3A and B, the expression levels of cGAS and p-STING were significantly decreased in Tryp-treated mice at 3 dpi compared with those in control-treated mice. Moreover, the coimmunostaining analyses of cGAS/Iba1 (Figure 3C and D) and p-STING/Iba1 (Figure 3E and F) showed that the microglial cGAS-STING pathway was inhibited in spinal cord of Tryp-treated mice at 3 dpi. Subsequently, we assessed the expression of p-p65, p65, p-IκBα, and IκBα at 7 dpi and found a significant inhibition of NF-κB activation in the Tryptreated group (Figure 3G and H). To further evaluate the role of Tryp in regulating the polarization and activation of microglia after SCI, CD86 and CD206 were each coimmunostained with CD68, which is highly expressed in activated microglia, near the spinal cord lesion area. As shown in Figure 3I–L, compared with those in the Sham group, the percentages of CD68+ cells were remarkably increased in the spinal cord of both control-treated and Tryp-treated mice at 7 dpi. There were no significant differences between controltreated and Tryp-treated mice, which indicated that Tryp did not suppress microglial activation at the early stage after SCI. The ratio of activated M1 microglia (CD68+ CD86+ cells) was significantly reduced and the ratio of activated M2 microglia (CD68+ CD206+ cells) was substantially increased in Tryp-treated mice compared with those in control-treated mice at 7 dpi (Figure 3I–L). Moreover, consistent with the immunostaining results, western blot analyses of the protein levels of CD206 and CD86 in lesion sites at 7 dpi (Figure 3M–O) showed that microglia were significantly shifted to the M2 phenotype in Tryp-treated mice compared with control-treated mice. Furthermore, qRTPCR analysis of IL-6, IL-1β, TNF-α, CD206, and Arg-1 showed that a proinflammatory response was dominant at 7 dpi, and that Tryp promoted an anti-inflammatory response (Figure 3P–T). These results suggested that inhibition of the cGAS/STING/NFκB pathway by Tryp promoted microglial polarization to the M2 subtype and suppressed microglia-derived neuroinflammation after SCI.
Figure 4 | Tryp promotes functional recovery and tissue repair after SCI.
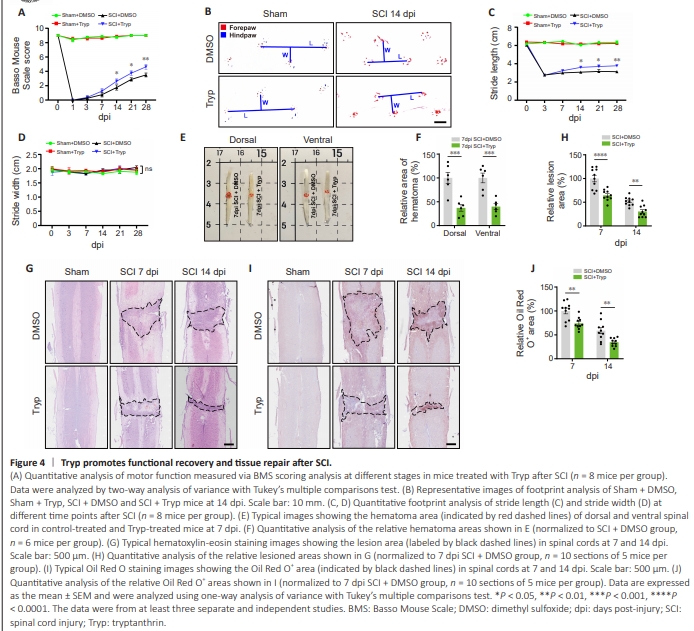
To examine the effects of Tryp on functional recovery after SCI, footprint analysis and BMS scoring were performed. As shown in Figure 4A, the BMS score was significantly increased in Tryptreated mice compared with that in control-treated mice (14 dpi, P = 0.0179; 21 dpi, P = 0.0179; 28 dpi, P = 0.0011). Footprint analysis showed that the stride length was significantly improved in Tryp-treated mice compared with that in control-treated mice at 14 dpi, whereas no significant difference was detected in the stride width between groups (Figure 4B–D). After SCI in mice, spinal cord lesions are detrimental to the reconstruction of neural networks across the epicenter and to the functional recovery of hindlimbs (Huang et al., 2021). Thus, we evaluated the histological and morphological changes in spinal cord tissue after SCI. Compared with that in control-treated mice, the hematoma area was decreased in both dorsal (P = 0.0002) and ventral (P = 0.0003) spinal cord in Tryp-treated mice at 7 dpi (Figure 4E and F). HE staining further showed that the lesion size in Tryp-treated mice was significantly decreased at both 7 and 14 dpi (7 dpi, P < 0.0001; 14 dpi, P = 0.0045) compared with those in controltreated mice (Figure 4G and H). Additionally, Oil Red O staining analyses showed that on 7 and 14 dpi, the diffusion of lipid-rich debris, which indicated the extent of inflammatory infiltration (Zhou et al., 2020b), was restricted in spinal cord tissue of Tryptreated mice but not in control-treated mice (Figure 4I and J). These results suggested that Tryp promoted functional recovery and tissue repair after SCI in mice.
Figure 5 | Tryp inhibits neuronal loss after spinal cord injury.

Recent studies have shown that the locomotor functional recovery after SCI is strongly correlated with the retention of ventral motor neurons (VMNs) and the regeneration of axons within and nearby the spinal cord lesion area (Chen and Shifman, 2019, Huang et al., 2021). Moreover, some studies have shown that oligodendrocyte-derived myelin sheaths support the recovery of axon conduction after SCI (Llorens-Bobadilla et al., 2020). Both Nissl staining (P = 0.0122; Figure 5A and B) and NeuN immunostaining (P = 0.0090; Figure 5C and D) showed Tryp significantly reduced the injury-induced loss of VMNs compared with control-treated mice. Moreover, western blot analysis of Bcl-2 and Bax showed that compared with that in controltreated mice, Bcl-2 expression was significantly downregulated, whereas Bax expression was not changed significantly (Figure 5E– G). These results suggested that Tryp inhibited the apoptosis of VMNs. We also defined each NF+ axonal punctum colocalized with MBP as a myelinated axon, and counted the ratio of MBP+ NF+ axon puncta to total NF+ axon puncta. The loss of myelin sheaths was significantly inhibited in the spinal cord lesion area in the Tryptreated mice compared with control-treated mice (Figure 5H and I). As shown in Figure 5J–L, the fluorescence intensities of both NF and MBP in the epicenter of injured spinal cord in the Tryptreated mice were higher than those in the control-treated mice. These results suggested that Tryp alleviated the loss of VMNs in the lesion area after SCI.
Figure 6 | Microglia treated with tryptanthrin suppresses neuronal apoptosis through ER stress–related signaling.

We next investigated whether the improvement of microgliaderived inflammatory microenvironment induced by Tryp promoted neuronal survival after SCI (Figure 6A). Coimmunostaining analysis of CC-3 and NeuN showed that LTtreated BV2 cell-derived conditional medium (LT-CM) inhibited the apoptosis of N2a cells, compared with that in the LD-CM (LD-treated BV2 cell-derived CM) group (P = 0.0057; Figure 6B and C). As expected, LT-CM increased Bcl-2 expression and downregulated Bax expression in N2a cells (Figure 6D– F) compared with the LD-CM group. To further investigate the potential mechanisms of the neuroprotective effect of Tryptreated BV2 cells on N2a cells, we evaluated the activation of ER stress, which has been demonstrated to cause apoptosis, especially in neurons (Penas et al., 2007). Western blot analysis of p-eIF2α (P = 0.0013), eIF2α, ATF4 (P = 0.0004), and CHOP (P = 0.0110) showed that LT-CM treatment alleviated ER stress in N2a cells compared with LD-CM treatment (Figure 6G–K). Double immunostaining analysis of CHOP and NeuN also showed that LTCM treatment inhibited ER stress in N2a cells (Figure 6L and M). Taken together, these results suggested that Tryp pretreatment of microglia reduced the ER stress-related apoptosis of neurons.