脑损伤
-
Figure 1 | ADSC culture and identity verification, and the effect of VEGF on ADSCs.
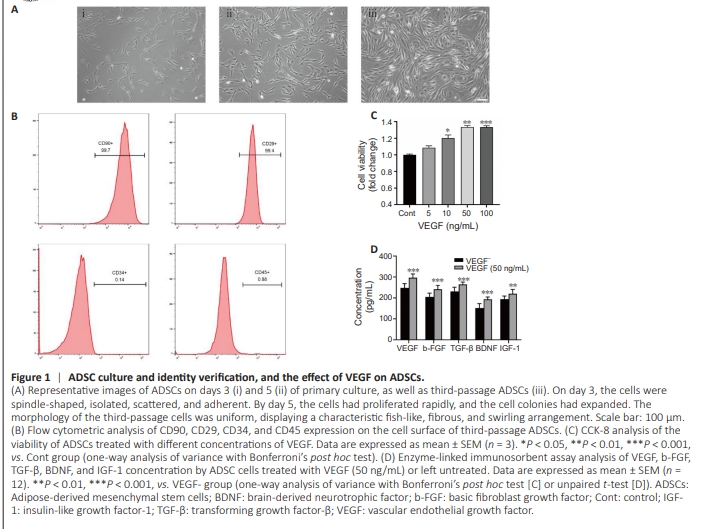
After 72 hours of primary culture, the cells appeared spindleshaped, isolated, scattered, and adherent (Figure 1Ai). By day 5 of culture, the cells had proliferated rapidly, and the cell colonies gradually enlarged (Figure 1Aii). The morphology of third-passage cells was uniform, and they displayed a typical fish-like, fibrous, and swirling arrangement (Figure 1Aiii). Flow cytometry analysis of the adherent cells revealed high levels of CD90 and CD29 expression and low levels of CD34 and CD45 expression (Figure 1B). These results are consistent with previous findings reported by Leu et al. (2010). To investigate the effects of VEGF on stem cell activity, different concentrations of VEGF were introduced to the cell growth medium. The results showed that cellular viability increased in parallel with the VEGF concentration within the range of 0 to 50 ng/mL. However, no significant difference in cellular activity was observed within the range of 50 to 100 ng/mL (Figure 1C). Therefore, 50 ng/mL of VEGF was used for all subsequent experiments. After 1 day of culture, VEGF, b-FGF, TGF-β, BDNF, and IGF-1 levels in the supernatant were assayed by enzyme-linked immunosorbent assay to detect paracrine activity of the stem cells. There was a clear increase in the expression of these cytokines by stem cells treated with 50 ng/mL VEGF (Figure 1D). Based on these findings, the optimal VEGF concentration for stem cell growth was determined to be 50 ng/mL.
Figure 4 | Effect of co-transplantation of ADSCs with the VEFG-loaded CS-HA hydrogel scaffold on cytokine expression and angiogenesis after tMCAO.
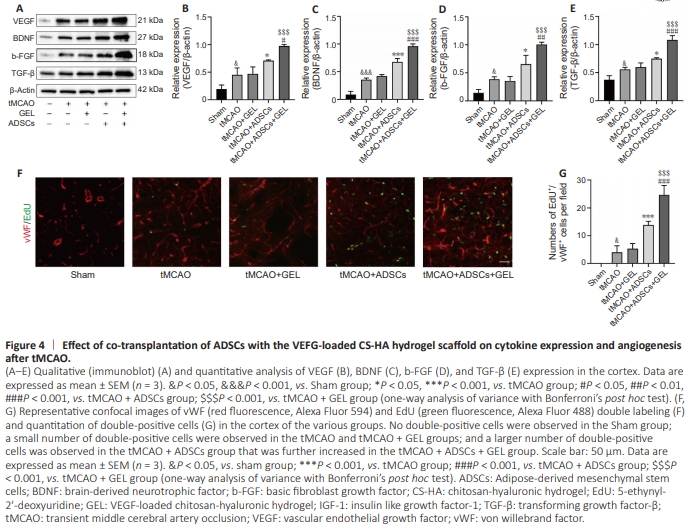
Next, we measured the levels of several representative c y to k i n e s k n o w n to b e u p re g u l ate d b y ste m c e l l transplantation (Bajek et al., 2016; González-Nieto et al., 2018; Figure 4A), including VEGF, BDNF, b-FGF, and TGF-β, produced by ADSCs injected alone or combined with VEGF-loaded CSHA hydrogel scaffold. Western blotting analysis revealed higher expression levels of these cytokines in the ADSCtreated rats compared with the ischemic rats. Furthermore, rats treated with both ADSCs and GEL exhibited even higher cytokine expression levels than those treated with ADSCs alone or GEL alone (Figure 4B–D). Furthermore, angiogenesis was assessed at 7 days post-tMCAO by quantifying vWF and EdU double-positive cells (Figure 4F). Compared with the ischemic rats, both the ADSC-treated rats and the ADSCs + GEL-treated rats exhibited a higher number of vWF and EdU double-positive cells. Notably, the ADSCs + GEL-treated rats showed a significantly greater number of double-positive cells than the ADSC-treated rats alone (Figure 4G). These findings clearly indicate that administration of ADSCs alone can effectively enhance the concentration of cytokines and promote vascular regeneration in the peri-ischemic area, and that co-transplantation with the VEGF-loaded CS-HA hydrogel scaffold markedly enhances these beneficial effects.
Figure 5 | Co-transplantation of ADSCs with the VEGF-loaded CS-HA hydrogel scaffold decreased apoptosis after tMCAO in vivo, and coculture of neurons with ADSCs pretreated with VEGF decreased apoptosis after OGD/R in vitro.
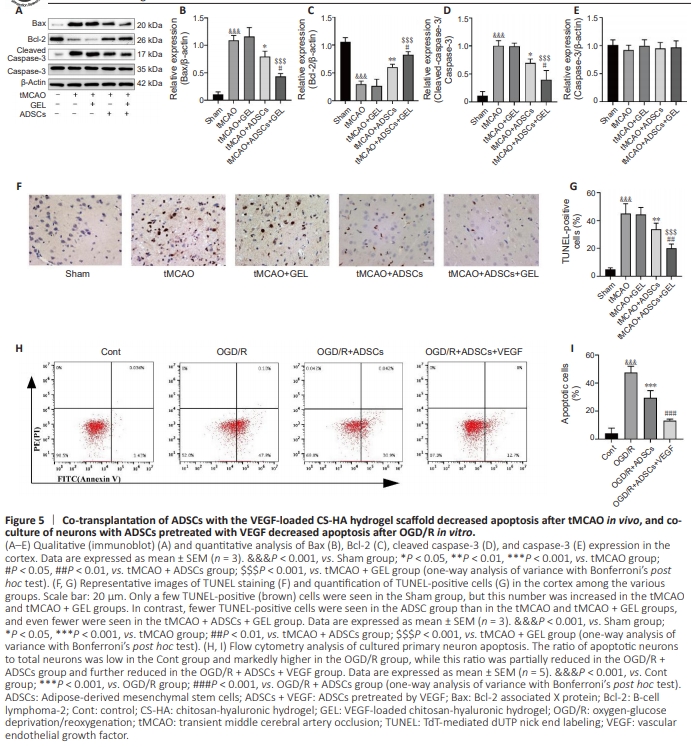
ubsequently, we investigated the effects of transplantation of ADSCs alone and co-transplantation of ADSCs with the VEGF-loaded CS-HA hydrogel scaffold on neuronal apoptosis. Our analyses revealed that, 7 days after surgery, there was an obvious decrease in Bcl-2 expression and activation of Bax and cleaved caspase-3 in tMCAO rats compared with their Sham counterparts. However, no significant difference in Bcl2, cleaved caspase-3, and Bax levels was observed between GEL-treated rats and ischemic rats. In contrast, both ADSCtreated and ADSCs + GEL–treated rats showed upregulation of Bcl-2 expression and downregulation of cleaved caspase-3 and Bax expression compared with the tMCAO group. Notably, the ADSCs + GEL–treated rats displayed higher levels of Bcl2 expression and much lower levels of cleaved caspase-3 and Bax expression than their GEL-treated and ADSC-treated counterparts. No significant difference in pro-caspase-3 levels was observed across all groups (Fguire 5A–E). Consistent with our earlier findings, 7 days post-operation the percentage of TUNEL-positive cells was increased in both tMCAO rats and GEL-treated rats compared with the Sham rats, with no significant difference between tMCAO rats and GEL-treated rats. However, the proportion of TUNEL-positive cells was significantly reduced in both ADSC-treated and ADSCs + GEL– treated rats compared with tMCAO rats. Furthermore, the reduction was more pronounced in the ADSCs + GEL–treated rats compared with the rats treated with either ADSCs alone or GEL alone (Figure 5F and G). Additionally, flow cytometry analysis revealed excessive apoptosis in cultured neurons 24 hours after OGD/R that was significantly attenuated by coculturing the neurons with ADSCs. Notably, neuronal apoptosis was further reduced when the neurons were co-cultured with ADSCs that were pretreated with CS-HA-VEGF-EM (Figure 5H and I).
Figure 6 | Co-transplantation of ADSCs with the VEFG-loaded CS-HA hydrogel scaffold increased mitochondrial integrity after tMCAO in vivo, and co-culture of neurons with ADSCs pretreated with VEGF increased mitochondrial integrity after OGD/R.
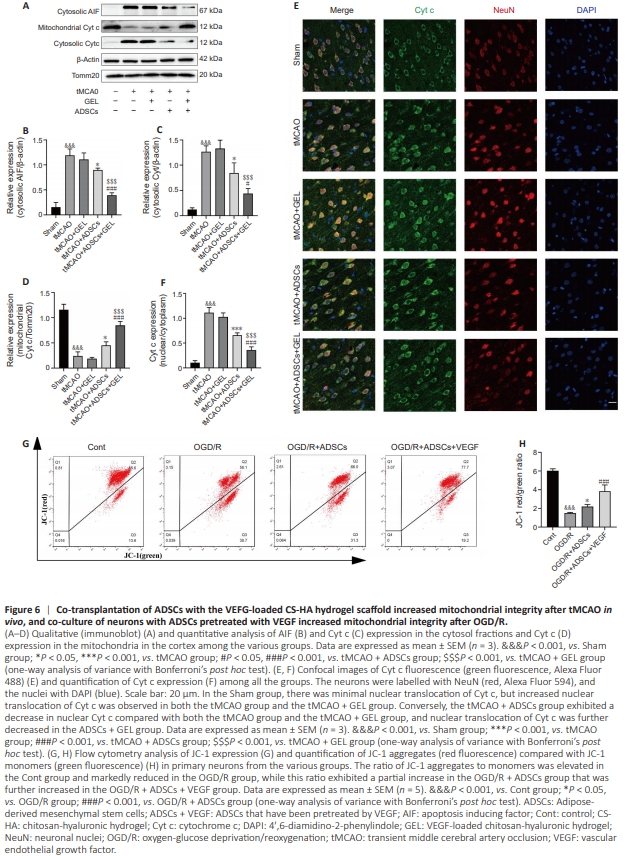
Mitochondrial breakdown is a critical early event following I/R injury (Guo et al., 2023). Therefore, we subsequently investigated the effects of transplanting ADSCs alone or ADSCs and the VEGF-loaded CS-HA hydrogel scaffold on mitochondrial integrity. Seven days after tMCAO, the level of Cyt c increased in the cytosol but decreased in the mitochondria of cortical cells in tMCAO-treated rats. However, no statistical difference was observed between GEL-treated rats and ischemic rats. Compared with the tMCAO group, both ADSC treatment and ADSCs + GEL treatment significantly reduced cytosolic Cyt c levels and increased mitochondrial Cyt c levels. Notably, the mitochondrial Cyt c level was significantly higher in ADSCs + GEL–treated rats than rats treated with either ADSCs alone or GEL alone (Figure 6A–D). Consistent with our earlier findings, immunofluorescence analysis showed Cyt c localization to the nucleus 7 days after tMCAO. This effect was reduced by transplantation of ADSCs or ADSCs and the VEGF-loaded CS-HA hydrogel scaffold. Notably, the ADSCs + GEL–treated rats exhibited less Cyt c diffusion to the nucleus compared with the ADSCtreated group (Figure 6E and F). In cultured neurons, OGD/R treatment caused extensive depolarization of the mitochondrial membrane potential (ΔΨm), which manifested as a significant increase in JC-1 monomer formation (green fluorescence) and a notable decrease in JC-1 aggregation (red fluorescence). This membrane depolarization was significantly alleviated by co-culturing neurons with ADSCs or with ADSCs pretreated with CS-HA-VEGF-EM. Notably, co-culture with CSHA-VEGF-EM–pretreated ADSCs was more effective than CSHA-VEGF-EM–pretreated with ADSCs alone in attenuating membrane depolarization (Figure 6G and H). These results suggest that ADSC transplantation preserves mitochondrial integrity, thereby exerting neuroprotection after cerebral infarction, and that this protective effect is enhanced by ADSC co-transplantation with a VEGF-loaded CS-HA hydrogel scaffold.
Figure 7 | Co-transplantation of ADSCs with the VEFG-loaded CS-HA hydrogel scaffold protected axons after tMCAO in vivo, and coculture of neurons with ADSCs pretreated with VEGF protected axons after OGD/R in vitro.

To determine the protective effects of transplantation of ADSCs alone or ADSCs with that VEGF-loaded hydrogel scaffold on axons after cerebral I/R injury, axonal density was assessed by detecting neurofilament-200 (NF-200) and growth-associated protein-43 (GAP-43) expression. Seven days after surgery, rats in the tMCAO group exhibited decreased NF-200 levels and increased GAP-43 levels compared with the Sham group. No significant difference was observed between the tMCAO and GEL groups. However, NF-200 and GAP-43 expression levels were notably higher in the ADSC-treated group than in the tMCAO group. Furthermore, both NF-200 and GAP-43 expression levels were even higher in the ADSCs + GEL–treated rats compared with rats treated with either ADSC alone or GEL alone (Figure 7A–C). Consistent with the results described above, NF-200–positive fibers were uniformly distributed and arranged in an orderly manner in the Sham group but were sparsely distributed and disordered in the tMCAO group. Similar to the western blot results, 7 days after surgery there were fewer NF-200-positive fibers in the tMCAO group compared with the Sham group,with no statistical difference seen between the tMCAO and GEL groups. However, the number of NF-200-positive fibers was markedly increased in ADSC-treated rats and ADSCs + GEL–treated rats compared with tMCAO rats. In addition, the ADSCs + GEL–treated rats exhibited markedly more NF-200– positive fibers than either the ADSC-treated or GEL-treated rats (Figure 7D and E). Next, we assessed the effect of CS-HA-VEGF-EM-pretreated ADSCs on neuronal morphology in vitro. Exposure to OGD/R notably decreased both the length of the longest neurite and the number of primary neurites compared with the Control group. Co-culturing neurons with ADSCs pretreated with CS-HA-EM or CS-HA-VEGF-EM mitigated these effects. Additionally, neurons co-cultured with CS-HA-VEGF-EMpretreated ADSCs exhibited a significantly greater number of primary neurites, as well as a longer length of the longest neurite, compared with those co-cultured with CS-HA-EMpretreated ADSCs (Figure 7F–H). Overall, these findings indicated that co-transplanting ADSCs with the VEGF-loaded CS-HA hydrogel scaffold significantly enhanced the ability of ADSCs to promote axonal regeneration.