神经损伤与修复
-
Figure 2 | Indicaxanthin prevents HFD-induced neuronal apoptosis.
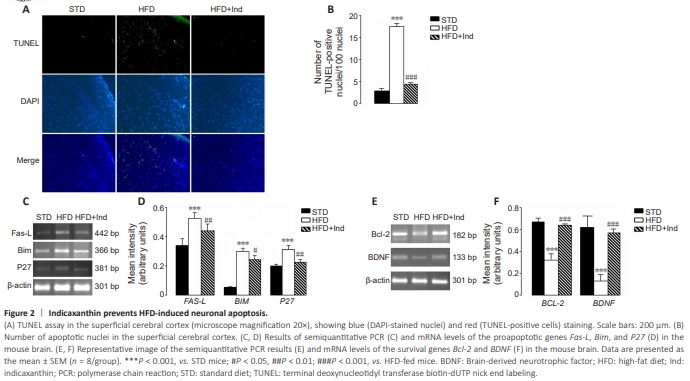
The presence of neuroapoptosis was evaluated in superficial cortex sections via a TUNEL assay, which can reveal fragmented DNA, indices of cell death, and comparative analyses of the gene expression of proapoptotic and antiapoptotic factors. Compared with STD animals, HFD-fed brains presented a significant increase in the number of apoptotic neurons; strong gene overexpression of the proapoptotic proteins Fas-L, Bim, and P27; and a significant reduction in the expression of the antiapoptotic proteins Bcl-2 and BDNF (Figure 2A–F). Moreover, we observed significantly fewer apoptotic cells in Ind-treated mice than in HFD-fed mice (P < 0.001; Figure 2A and B). Furthermore, the HFD-induced gene upregulation of the proapoptotic proteins Fas-L (P < 0.01), Bim (P < 0.05), and P27 (P < 0.01) was significantly attenuated by Ind treatment compared with that of the HFD-fed group (Figure 2C and D). Accordingly, the gene expression of the antiapoptotic proteins Bcl2 and BDNF was similar to that of STD mice in the HFD + Ind group (Figure 2E and F), suggesting that Ind was able to prevent HFD-induced neuroapoptosis.
Figure 4 | Indicaxanthin ameliorates brain inflammation induced by HFD.
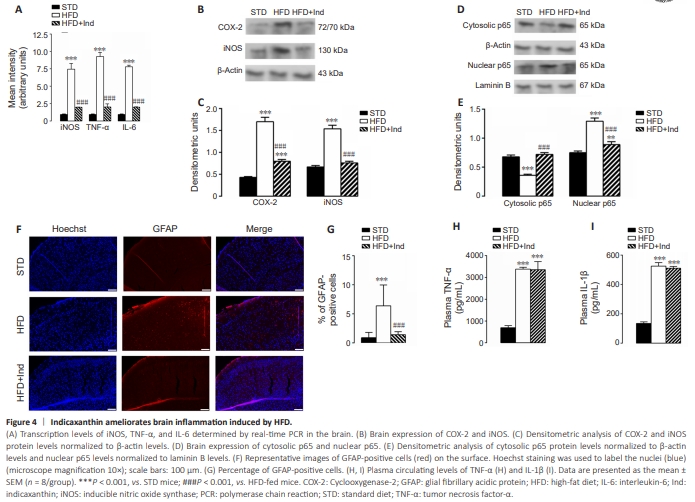
Oxidative stress and inflammation are mutually linked and are closely associated with neurodegeneration (Kempuraj et al., 2016). Consequently, we evaluated whether and how Ind treatment could alleviate HFD-induced neuroinflammation. Molecular analyses of proinflammatory parameters revealed that the gene upregulation of iNOS, TNF-α, and IL-6 observed in HFD-fed brains was significantly decreased in HFD + Ind-fed brains (P < 0.001), as was the protein overexpression of iNOS and cyclooxygenase-2 (COX-2) (P < 0.001; Figure 4A?C). Moreover, the nuclear translocation of NF-kB, a key transcription factor involved in the elaboration of proinflammatory mediators, was increased in HFD-fed brains. Notably, these effects were mitigated by Ind treatment (Figure 4D and E).Furthermore, to confirm the anti-inflammatory activity of Ind in the brain, the presence of GFAP, an index of astrocyte activation and gliosis in the brain, was evaluated in brain sections from different animal groups via immunofluorescence. The analysis revealed increased GFAP immunoreactivity in the superficial cerebral cortex of HFD-fed animals compared with that in STD-fed animals (P < 0.001). This increase was not observed in HFD + Ind mice. In fact, the number of GFAP-positive cells in the cerebral sections from HFD + Ind mice was similar to that in the STD-mice (Figure 4F and G). However, the plasma concentrations of the inflammatory cytokines TNF-α and IL-1β, which were significantly greater in HFD-fed mice than in STD-fed mice (P < 0.001), were not significantly modified by Ind treatment (Figure 4H and I). These results suggest that Ind is able to mitigate HFD-induced neuroinflammation.