脑损伤
-
Figure 1 | Experimental design, transplantation procedure, and evaluation of neurogenesis in the SVZ following ES.
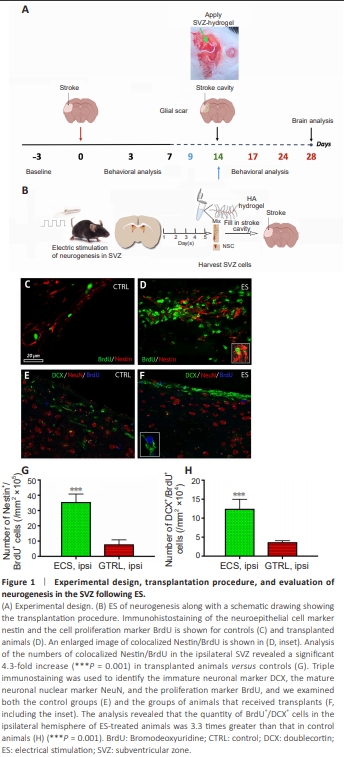
The experimental setup is illustrated in Figure 1A (top panel), while Figure 1B presents a schematic representation of the transplantation process. For the first 3 days after stroke, the animals in all the groups were fed soft pellets to facilitate nutrition. Thereafter, they were allowed to eat ad libitum. The mortality rate was nearly identical, at 8% for each group. All the animals that died did so within the first 7 days after stroke. In the figures showing poststroke behavioral recovery, we have provided the number of animals that survived the 28-day testing period. Electrical stimulation leads to increased neurogenesis in the subventricular zone To assess whether ES led to an increased number of newborn cells in the SVZ, we performed double immunostaining with the neuroepithelial cell marker nestin and the cell proliferation marker BrdU (Balseanu et al., 2020) in both control (Figure 1C) and transplanted animals (Figure 1D, and inset). Analysis of the number of colocalized Nestin/BrdU in the ipsilateral SVZ revealed a significant 4.3-fold increase (P = 0.001) in transplanted animals compared with controls (Figure 1G). By performing triple immunostaining to detect the immature neuronal marker DCX, the mature neuronal nuclear marker NeuN, and the proliferation marker BrdU in control (Figure 1E) and transplanted animals (Figure 1F, and inset), we found that the number of BrdU+ /DCX+ cells in the ipsilateral hemisphere of ES animals outnumbered that in control animals by a factor of 3.3 (P = 0.001; Figure 1H).
Figure 3 | Nestin colocalized with BrdU in the infarcted area of treated animals.
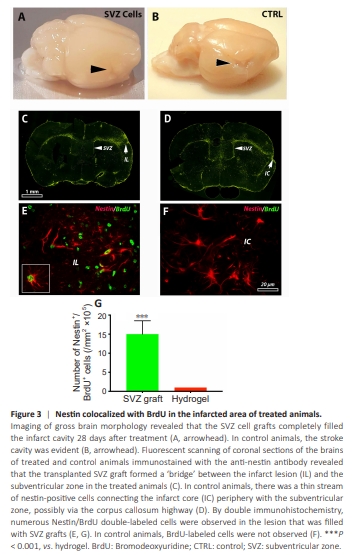
Examination of brain morphology revealed that the SVZ cell grafts completely filled the infarct cavity 28 days after hydrogel administration at 14 days poststroke (Figure 3A). Histologically, we were unable to observe the infarct border in the cortex of animals treated with SVZ. However, in the control animals, the stroke cavity was clearly evident (Figure 3B). Scanning coronal sections of the brains of treated and control animals immunostained with anti-nestin antibodies clearly revealed that the transplanted SVZ graft formed a “bridge” between the infarct lesion and the SVZ zone in the treated animals (Figure 3C). In control animals, there was a thin stream of nestin-positive cells connecting the infarct core (IC) periphery with the SVZ zone, possibly via the corpus callosum highway (Figure 3D). Finally, examination of the lesioned hemisphere via double immunohistochemistry revealed numerous Nestin/ BrdU double-labeled cells in the lesion filled with the SVZ grafts (Figure 3E and G). In control animals, BrdU-labeled cells were not observed (Figure 3F).
Figure 4 | Regional distribution of the juvenile neuroepithelial marker Mash1 on the ipsilateral side.
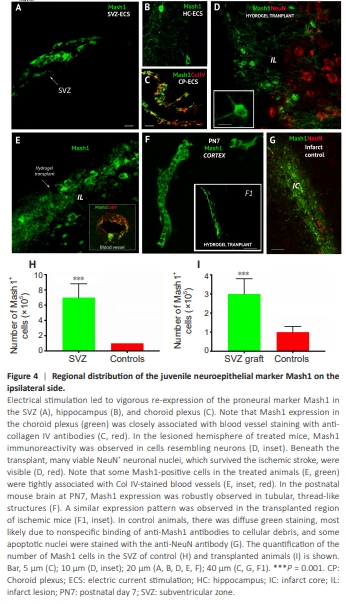
The neuroepithelial marker Mash1 is expressed during brain development but is downregulated in the adult brain (Popa-Wagner et al., 2007). However, ES led to vigorous re-expression of proneural Mash1 in the SVZ (Figure 4A and H), hippocampus (Figure 4B), and choroid plexus (Figure 4C, green). Mash1 expression in the choroid plexus was closely associated with blood vessel staining with anti-collagen IV antibodies (Figure 4C). Mash1 immunostaining in the lesioned hemisphere of treated mice revealed that in the transplanted region, Mash1 immunoreactivity was observed in neuronal-like cells (Figure 4D and I). In the hydrogel containing Mash1+ cells, many NeuN+ neuronal nuclei that survived ischemic stroke were clearly visible (Figure 4D). Like those in the choroid plexus, some Mash1-positive cells (Figure 4E, green) in the treated animals were tightly associated with blood vessels, as visualized with an anti-Col IV antibody (Figure 4E, inset, red). Indeed, in the postnatal mouse brain PN7, Mash1 was robustly expressed in tubular, thread-like structures (Figure 4F). Similar expression was observed in the transplanted region of ischemic mice (Figure 4F1, inset). Finally, in control animals, there was diffuse green staining, most likely resulting from nonspecific binding of anti-Mash1 antibodies to cellular debris caused by tissue necrosis after ischemia, and some apoptotic nuclei were stained with the anti-NeuN antibody (Figure 4G).
Figure 5 | The microvascular density is greater in the area transplanted with SVZ cells.

VCAM-1, also known as CD106, is abundantly present in the brain endothelium following stroke. Its presence has been associated with an augmented process of angiogenesis (Kaur et al., 2023). As a result, the microvasculature within the SVZ-transplanted region showed robust upregulation of VCAM-1. This was evident by a 2.2-fold increase at 28 days, in contrast to the animals in the control group that were administered hydrogel only (Figure 5A, B, and G). However, the expression of the other two angiogenesis markers, laminin and CD31, was not significantly different between the control group and the transplanted animals (Figure 5A, B, and G). Microglial/macrophages play a pivotal role in angiogenesis following ischemic stroke. An increase in microvessel density post-stroke has been observed exclusively in regions containing macrophages. This finding implies a potential association between angiogenesis and the proliferation of macrophages (Mcnay et al., 2006). In previous research conducted by our team, ANXA3 was identified as a marker for both resting and activated microglia (Junker et al., 2007). In the present study, we observed lower levels of ANXA3 in the cortical region of animals that were transplanted with SVZ cells than in those that were administered only hydrogel (Figure 5C, D, and H). Doublecortin expression is increased in the area transplanted with subventricular zone cells Next, we investigated the presence of the immature neuronal marker doublecortin by immunofluorescence in the peri-infarct area of animals after stroke. We observed scattered DCX-positive cells against a background of inflammatory cells expressing Iba1 in the perilesional area of the control animals (Figure 5E). The infarct cavity filled with the hydrogel containing SVZ cells contained many more (2-fold) neurons expressing DCX (Figure 5F and I). The microglia marker Iba1 did not significantly differ between the control and treated animals (Figure 5E and F).