脑损伤
-
Figure 1 | AAV-PHP.eB mainly targets neurons in healthy mice.
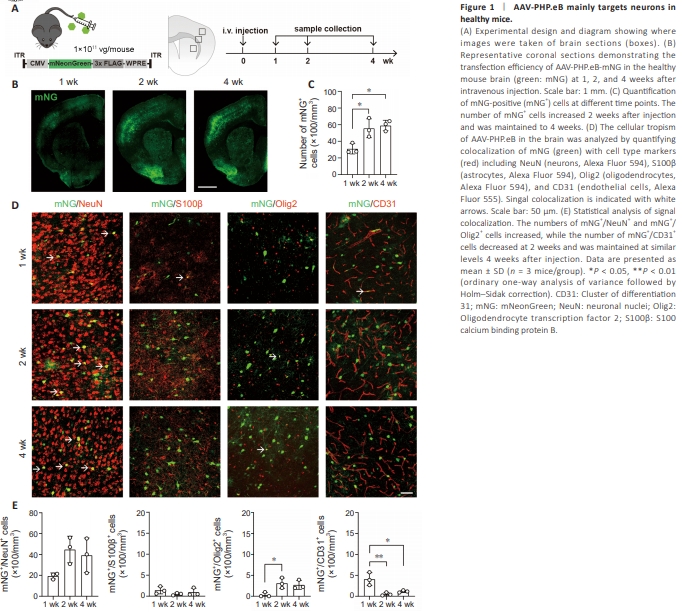
To validate previous observations (Chan et al., 2017) on the tropism and transfection efficiency of AAV-PHP.eB, we intravenously injected adult C57BL6/J mice with AAV-PHP.eB-mNG at a dose of 1 × 1011 vg/mouse. Brain samples were collected 1, 2, and 4 weeks after injection (Figure 1A), and mNG fluorescence was detected to confirm transfection (Figure 1B and C). We then performed co-immunostaining for mNG and NeuN, S100β, Olig2, or CD31 to determine whether the transfected cells were neurons, astrocytes, oligodendrocytes, or endothelial cells, respectively (Figure 1D). We found most of the mNG signals colocalized with NeuN+ cells, which is consistent with a previous report (Chan et al., 2017). Limited colocalization of mNG with S100β, CD31, and Olig2 was also detected, indicating a small degree of tropism for other cell types (Figure 1E). Statistically, there was no significant difference in transfected cell number or cell tropism between 2 and 4 weeks, indicating that AAV-PHP.eB transfection induced stable gene expression 2 weeks after injection.
Figure 2 | The cellular tropism of AAV-PHP.eB in the mouse brain shifted after ischemic stroke.
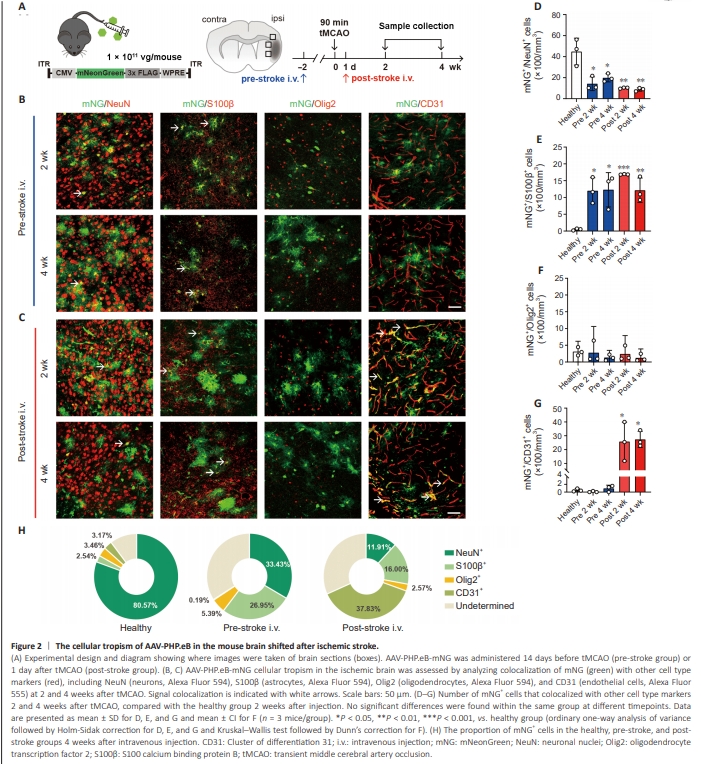
Since AAV-PHP.eB was administered systemically, we asked whether cerebrovascular diseases would alter its tropism. To investigate this, we established the tMCAO mouse model to mimic ischemic stroke. Similar to AAV-mediated gene delivery timelines used in preclinical and clinical studies, we injected AAV-PHP.eB-mNG at two different time points: 14 days before (pre-stroke group, similar to preclinical studies testing genetic modulation) or 1 day after (post-stroke group, more relevant for clinical application) ischemic stroke onset (Figure 2A). Immunostaining of the peri-infarct area showed that neuronal tropism decreased in both the pre-stroke and post-stroke groups (Figure 2B–D) compared with the healthy group at 2 weeks after intravenous injection, with a more significant decline in the post-stroke group (Figure 2D). Altered morphology of mNG+ cells in the peri-infarct area was also observed (Figure 2B and C), indicating a shift in cellular tropism towards non-neuronal cells. We found that the number of mNG+ /S100β+ cells increased in both stroke groups, with a significant increase at 2 weeks followed by a plateau in the number of double-positive cells (Figure 2E), with no significant difference in mNG+ /Olig2+ cells (Figure 2F). We also observed a sharp increase in the number of mNG+ /CD31+ cells in the post-stroke group, compared to the healthy group (Figure 2G), indicating that viral administration after ischemic stroke induced a shift to endothelial cell tropism. To rule out any variation owing to differences in transfection efficiency, the proportion of doublepositive cells in the mNG+ cells was analyzed, with similar results to the total number of positive cells (Figure 2H). Next, we examined the contralateral hemisphere to determine whether AAVPHP.eB cellular tropism was only altered within the ipsilateral hemisphere or changed in both hemispheres. Both the cell number and the proportion of cells indicated similar tropism overall in the contralateral brain and the healthy brain, with strong neuronal targeting evident in both the pre-stroke and post-stroke groups (Additional Figure 2).
Figure 3 | AAV-PHP.eB targets astrocytes that do not arise by proliferation after tMCAO.
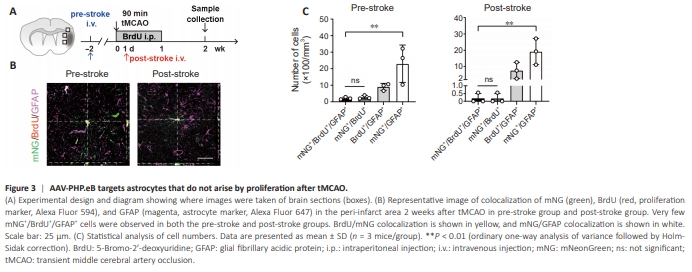
Since astrocytes proliferate after ischemic stroke, we next asked whether the apparent increase in astrocytic targeting was due to astrocyte proliferation. To test this, we injected mice that had received AAV-PHP.eB pre-stroke or post-stroke with BrdU (50 mg/kg/day) from day 1 to day 7 after tMCAO and performed immunostaining for GFAP (Figure 3A and B). Only a few mNG+ / BrdU+ /GFAP+ cells were observed, while a significantly larger number of mNG+ /GFAP+ cells were seen (Figure 3C), indicating that the majority of the targeted astrocytes did not arise by proliferation after tMCAO.
Figure 4 | Altered AAV-PHP.eB endothelial cell tropism correlates with Ly6A expression after ischemic stroke in mice.
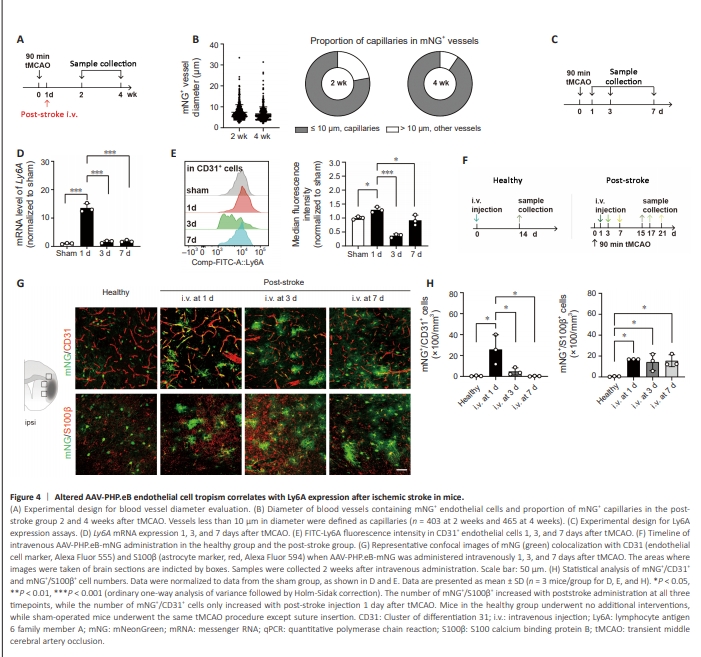
We also found that the targeted endothelial cells were mostly located in vessels less than 10 μm in diameter (Figure 4A and B), indicating preferential targeting to capillary endothelial cells. Given the sharp increase in AAV-PHP. eB targeting to endothelial cells after ischemic stroke and the fact that AAVPHP.eB transcytosis in endothelial cells relies on its specific receptor Ly6A (Hordeaux et al., 2019; Huang et al., 2019), we asked whether the shift in cellular tropism related to a change in Ly6A expression after ischemic stroke. To test this, we collected samples from the peri-infarct area and examined Ly6A expression at 1, 3, and 7 days after tMCAO (Figure 4C). The results showed that Ly6A messenger RNA (mRNA) expression in the peri-infarct region (Figure 4D) and Ly6A protein expression in CD31+ endothelial cells (Figure 4E) were significantly increased at 1 day after tMCAO compared with the sham group and other time points. These data indicate an increase in Ly6A expression at 1 day post-stroke, which was when we administered AAVPHP.eB to mice in the post-stroke group. To determine whether AAV-PHP.eB administered at later timepoints exhibits a similar shift in cellular tropism, we injected the virus at 1, 3, or 7 days after tMCAO and collected samples 14 days after viral administration (Figure 4F). We found that the number of mNG+ /CD31+ cells significantly increased when AAV-PHP.eB injected was 1 day after tMCAO, while no significant difference was found when the injection was administered 3 or 7 days after tMCAO, suggesting a correlation between Ly6A expression and endothelial cell targeting (Figure 4G and H). We also examined the number of mNG+ /S100β+ cells after AAV-PHP.eB administration at different timepoints and observed a similar increase in S100β+ cell transfection in all three postinjection groups compared with the healthy group, indicating that astrocytic tropism was not impacted by the injection time, nor by differential expression of Ly6A at different time points.
Figure 6 | AAV-PHP.eB-mediated bFGF gene therapy promotes neurogenesis after ischemic stroke in mice.
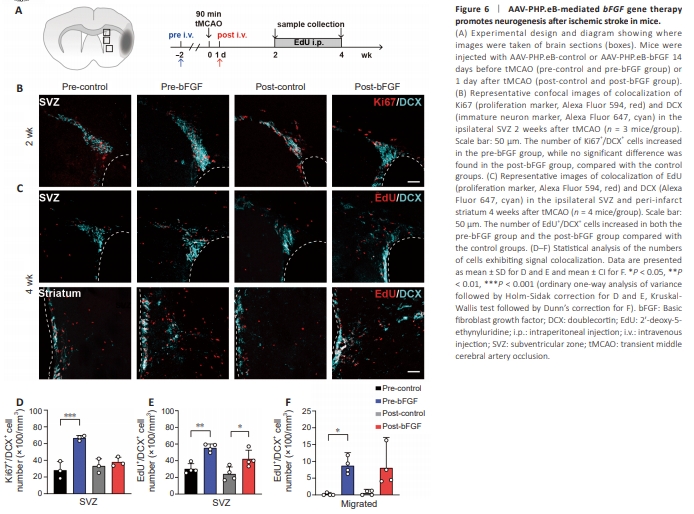
Next we examined neurogenesis in the ipsilateral SVZ and striatum (Figure 6A). The SVZ is enriched in neural stem cells, and after stroke these cells proliferate and migrate towards the peri-infarct area, where they differentiate into functional neurons. This remodeling process ultimately achieves functional repair of the peri-infarct area, including the affected motor cortex (Williamson et al., 2019). The striatum also plays an important role in motor function recovery because it receives synaptic input from the motor cortex to generate certain movements. Motor outcomes are related to the extent of coordinated activity between the cortex and the striatum after ischemic stroke (Guo et al., 2021). First, we examined neurogenesis by co-staining brain sections collected 2 weeks after tMCAO for Ki67 (cell proliferation marker) and DCX (immature neuron marker) (Figure 6B). To explore longer-term neurogenesis, we injected mice with EdU for 2 to 4 weeks after tMCAO, then examined co-localization of EdU and DCX in both the SVZ and the striatum to detect proliferated and migrated neurons (Figure 6C). Two weeks after tMCAO, only the pre-bFGF group showed a significant increase in the number of Ki67+ /DCX+ cells in SVZ (Figure 6D), while both the pre-bFGF and post-bFGF groups exhibited an increase in the number of EdU+ /DCX+ cells in the SVZ and striatum (Figure 6E and F), indicating that AAV-PHP.eB-bFGF strongly promoted neurogenesis.
Figure 7 | AAV-PHP.eB-mediated bFGF gene therapy promotes angiogenesis after ischemic stroke in mice.

We further examined endothelial cell proliferation in the peri-infarct area (Figure 7A) in brain samples from mice injected with EdU from 2 to 4 weeks after tMCAO. The number of Ki67+ /CD31+ cells at 2 weeks (Figure 7B) and EdU+ /CD31+ cells at 4 weeks (Figure 7C) after tMCAO increased in the postbFGF group (Figure 7D and E) but not in the pre-bFGF group, indicating that only post-stroke AAV-PHP.eB-bFGF administration improved angiogenesis after ischemic stroke.