脑损伤
-
Figure 2 | FTO regulates the microglial activation during the inflammatory response.
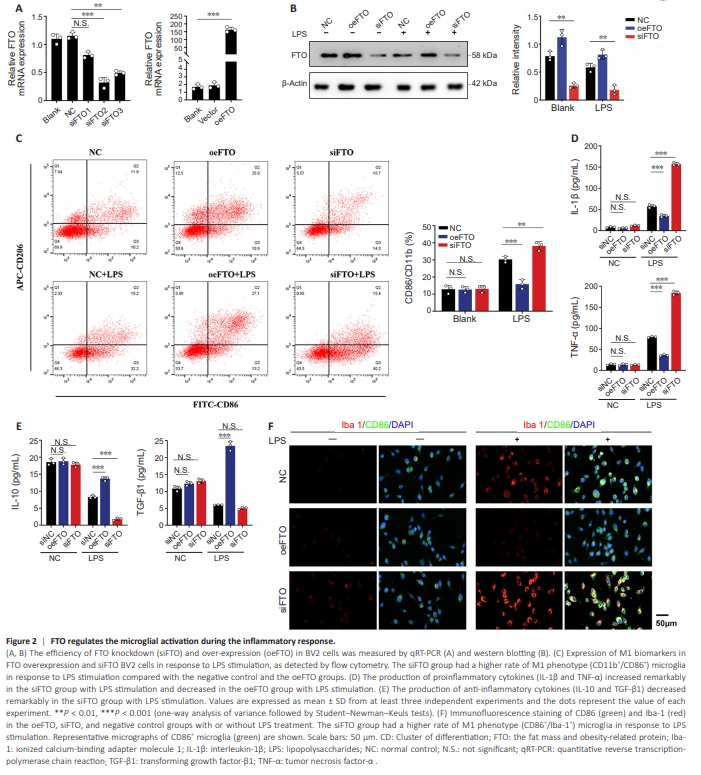
Our results showed downregulation of FTO in M1-like microglia. We thus further verified the role of FTO in the polarization and activation of microglia using FTO siRNAs and plasmids to knock down and overexpress FTO in BV2 cells, respectively. FTO mRNA expression was reduced after siRNA transfection (siFTO group) and upregulated after pcDNA3.1-Flag-FTO transfection (oeFTO group) (Figure 2A and B). As shown in Figure 2C, under LPS stimulation, the proportion of M1-like phenotype cells (CD86+ ) in the siFTO group was higher compared with the negative control and oeFTO groups. To assess changes in microglial inflammatory phenotype, immunofluorescence and ELISA were used to examine the levels of inflammatory cytokines following FTO knockdown and overexpression. The expression of antiinflammatory factors (IL-10 and TGF-β1) were elevated in the presence of overexpressed FTO and decreased in the absence of FTO in LPS-stimulated BV2 cells (Figure 2D and E). Immunofluorescence co-staining showed that under LPS stimulation, the M1-like phenotype population (Iba1+ /CD86+ ) increased in the siFTO group and decreased in the oeFTO group (Figure 2F). These data suggested that the m6 A demethylase FTO inhibited M1-like microglial polarization during the inflammatory response.
Figure 5 | Inhibition of ADAM17 in vitro blocks microglial activation after FTO-m6A modification.

To explore the interaction between FTO protein and ADAM17 transcripts, we performed co-localization assay by FISH and immunofluorescence in BV2 cells. FTO proteins (labeled with Cy3) co-localized with ADAM17 mRNA (indicated by FITC) in the cytoplasm; and green fluorescent signal (ADAM17 mRNA) was reduced upon overexpression of FTO or LPS treatment, which indicated a negative regulatory relationship between FTO proteins and ADAM17 transcripts (Figure 5A and B). These findings were consistent with the luciferase activity and ADAM17 mRNA stability results. Given that silencing of FTO promoted the expression of ADAM17 in activated BV2 cells, we next inhibited ADAM17 expression to examine whether ADAM17 was involved in M1 polarization. To further examine ADAM17-mediated effects on the activation of microglia, we examined M1-like microglia and M2-like microglia. Upon LPS stimulation, down-regulation of FTO expression promoted M1 polarization (CD86+ ) in BV2 cells; however, inhibition of ADAM17 (TAPI-1) reversed this phenomenon (Figure 5C). The results of WB and ELISA similarly supported the above conclusions. Inhibition of ADAM17 reversed FTO-mediated upregulation of ADAM17, CD86, and iNOS (Figure 5D). Downregulation of FTO further worsened pro-inflammatory upregulation (TNF-a, IL-1β and IL-6) and anti-inflammatory downregulation (TGF-β), both of which were blocked by TAPI (Figure 5E). Taken together, we concluded that ADAM17 was required for FTO-mediated M1 polarization.
Figure 6 | FTO is closely related to brain injury after TBI.
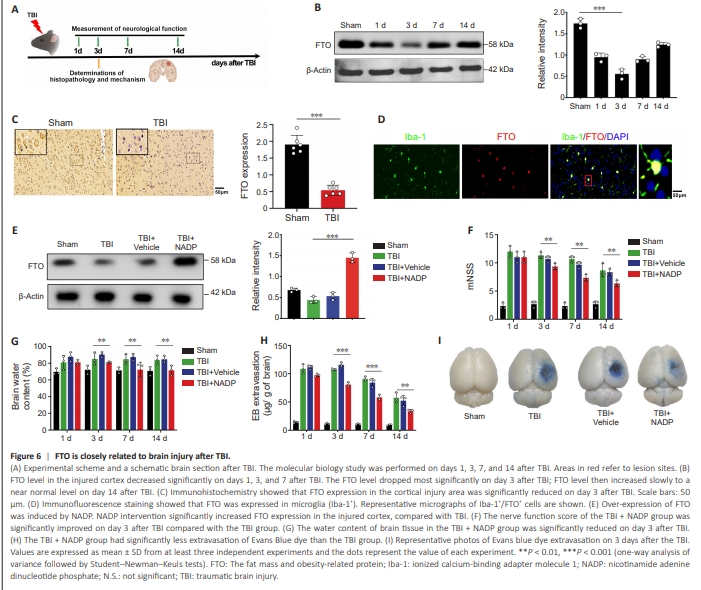
To examine whether FTO exerts an effect on microglial polarization during the initial inflammatory process caused by brain injury in vivo, a TBI mouse model was established (Figure 6A). Western blot results showed that FTO levels decreased on days 1, 3, and 7 after TBI, with the most significant decline on day 3, and gradually increased near baseline levels on day 14 after injury (Figure 6B). Immunohistochemistry showed that FTO in the cortical injury area was reduced on day 3 in the TBI group (Figure 6C). Immunofluorescence staining showed that FTO was expressed in microglia on day 3 in the TBI group (Figure 6D).NADP induces the over-expression of FTO (Meng et al., 2014). FTO in brain tissue decreased after TBI, and NADP reversed this phenomenon (Figure 6E). mNSS, brain water content, and Evans blue dye content were used to evaluate post-TBI neurological function. The mNSS, brain water content, and penetration rate of Evans blue in the TBI + NADP group were reduced on day 3 after TBI compared with that in the TBI and TBI + Vehicle group (Figure 6F–I).
Figure 7 | Enhancement of FTO reduces neuronal apoptosis and inhibits neuroinflammation after TBI in vivo.
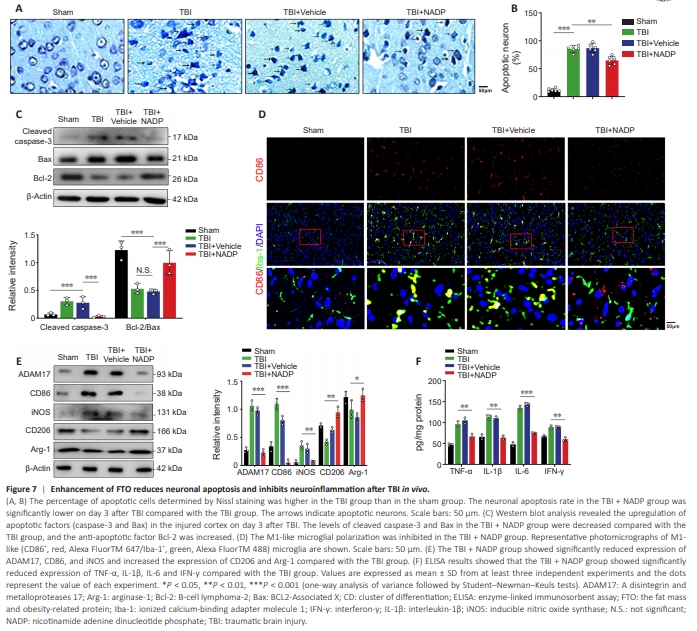
The neuronal apoptosis rate in the TBI + NADP group was lower on day 3 after TBI compared with the TBI group (Figure 7A–C). The number of M1- like microglia (CD86+ /Iba+ ) in the TBI + NADP group was lower compared with that in the TBI group (Figure 7D and E). The production of proinflammatory cytokines decreased remarkably in the TBI + NADP group (Figure 7F). These data demonstrate that over-expression of FTO inhibited microglia-induced inflammation and improved neurological function after TBI.
Figure 8 | FTO regulates neuroinflammation in vivo by targeting ADAM17 in microglia after TBI.
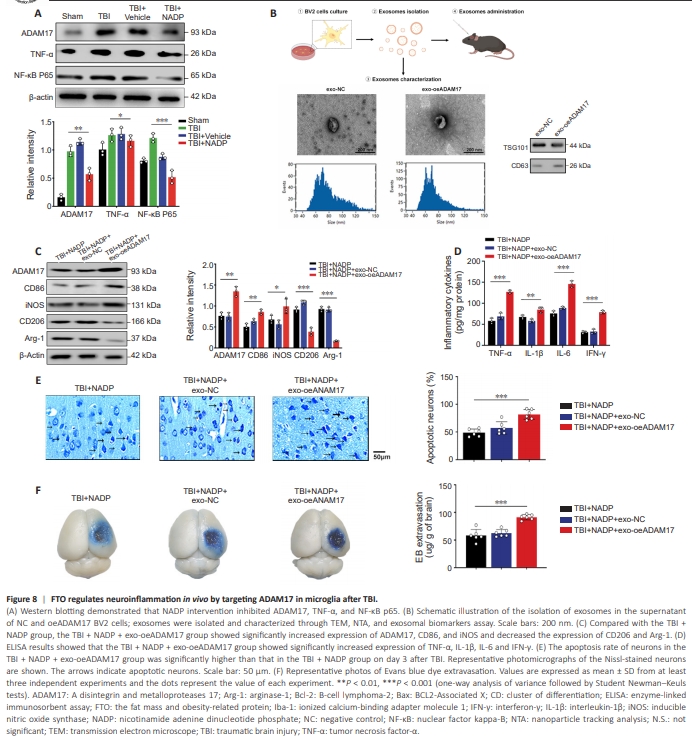
Western blot results showed that NADP intervention inhibited ADAM17, TNF-α, and NF-κB p65 protein expression (Figure 8A). Given that exosomes are important mediators of cell-to-cell communication and that exosome therapy holds promise for clinical translation, we next isolated exosomes in the supernatant of the ADAM17-BV2 overexpression group, followed by identification, concentration, and quantification of exosomes (Figure 8B). Compared with the TBI + NADP group, the TBI + NADP + exo-oeADAM17group had a higher level of the M1 microglial biomarkers CD86 and iNOS (Figure 8C), and the production of pro-inflammatory cytokines was remarkably increased (Figure 8D). Consistent with these results, the results of Nissl staining and Evans blue dye confirmed that exo-oeADAM17 treatment offset the neuroprotective effect of FTO overexpression (Figure 8E and F). These in vivo data demonstrated that the over-expression of FTO contributed to the inhibition of the inflammation and improved nerve function by targeting ADAM17 after TBI.