脑损伤
-
Figure 4 | Abolishing ALK5-mediated TGF-β signaling in neonatal mice in the mGFAPcre-ALK5fl/fl-Ai9 cKO mice alters the migration of postnatal-born neurons.

The TGF-β signaling pathway is a dynamic system that needs to be studied in a precise time and context-dependent manner due to its multi-functional roles in many different cell types, especially with the correct cellular context in vivo. To this end, we developed two novel inducible and cell type-specific mouse models to specifically silence TGF-β signaling in NSCs (mGFAPcre-ALK5fl/fl-Ai9) or immature neuroblasts (DCXcreERT2-ALK5fl/fl-Ai9) to investigate how the lack of TGF-β signaling affects NSCs or adult-born immature neuroblasts migration in vivo. The Cre-inducible tandem dimer tomato (tdTomato) fluorescent protein (Ai9 allele) enables us to genetically label NSCs in vivo and fate map their survival and morphology at different stages of the neurogenic cascade. In mGFAPcre-ALK5fl/fl-Ai9 mice (Figure 4A), Cre recombinase becomes active around early postnatal stages (Ganat et al., 2006; Chow et al., 2008) in NSCs (and astrocytes) and has previously been used by us and many other groups to study postnatal neurogenesis (Garcia et al., 2010; Wang et al., 2022a). Given the previous study showing the potential role of ALK5 in hippocampal adult neurogenesis (He et al., 2014), we focused on the hippocampal niche in our study. In the mGFAPcre-ALK5fl/fl-Ai9 mice cKO mouse line, at 8 weeks of age (a time point that allows cKO aNSCs to mature into new neurons from the onset of Cre activity), we did not observe any differences in the total number of doublecortin (DCX)+ immature neuroblasts suggesting that loss of TGF-β signaling in NSCs does not affect the net generation of immature neuroblasts (Figure 4B and C). There is a significant decrease in the migration of neurons throughout the granule cell layer (Figure 4D and F) without changes in the total number of tdTomato+ cells (Figure 4D and E), indicating that the increase in cells at the inner layer (SGZ) and decrease in cells at the outer layer is not due to change of total tdTomato+ cells but due to the aberrant migration of KO cells. This data supports the role of TGF-β signaling in the migration of newly born neurons in this early post-natal stage (from P0–P60 of age).
Figure 5| DCXcreERT2 driver line effectively recombines floxed alleles in immature neurons in the dentate gyrus of the hippocampus.
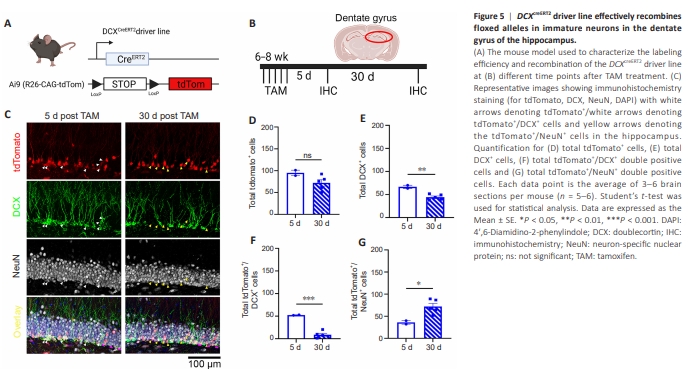
Figure 6 | Late-stage ablation of TGF-β signaling in a DCXcreERT2-ALK5fl/fl-Ai9 iKO mice does not affect the migration of adult-born neurons in the dentate gyrus of the hippocampus.

The mGFAPcre driver line deletes floxed genes around postnatal day 0 (P0) (Garcia et al., 2010) in mice and targets NSCs and leads to Alk5 gene deletion throughout the whole neurogenic cascade (including RGLs-radial glial cells, IPCs-intermediate progenitor cells, immature neurons, and mature neurons). Therefore, to further investigate the role of TGF-β signaling specifically in adult newborn immature neuroblasts (DCX+ ) in the hippocampus, we generated inducible DCXcreERT2-ALK5fl/fl-Ai9 iKO mice to specifically target TGF-β signaling in immature neuroblasts (Figure 5A). To validate the specificity of this Cre driver line (Zhang et al., 2010), 6-week-old DCXCreERT2-Ai9 mice were treated with 180 mg/kg of TAM once a day for 5 days and harvested the mice at 5- or 30 days post-TAM (Figure 5B) to examine the recombination of the Ai9 reporter allele in a cohort of DCX+ immature neurons longitudinally. Our data show that at 5 days post-TAM, most tdTomato+ cells were DCX+ (Figure 5C–G) while at 30 days most tdTomato+ cells were NeuN+ (Figure 5C–G), indicating accurate labeling of immature neurons and successful tracking of these cells through maturation into NeuN+ neurons. While we did not observe a change in the total amount of tdTomato+ cells, there was a significant difference in total DCX+ cells between post-TAM-5 day and post-TAM-30 day brains (Figure 5E). This difference in total DCX+ cells in DG could be attributed to the differences in the age of the mice at the time of harvest, suggesting that neurogenesis is more active in the 8-week-old brain compared with the 11-week-old brain. Nevertheless, after the specificity and efficiency of the DCXCreERT2 line were validated, we next investigated how the lack of TGF-β signaling in immature newly born neuroblasts affects the migration of these cells. To this end, we generated DCXCreERT2-ALK5fl/fl-Ai9 iKO and control (DCXCreERT2 -ALK5wt/wt- Ai9) mice (Figure 6A). Control and iKO mice were treated between 5–6 weeks of age with the same 5XTAM dosage, and the hippocampus was harvested and analyzed for the number and location of tdTomato+ cells 30days later. Interestingly, there are no significant differences in the migration of adult-born neurons in the DCXcreERT2-ALK5fl/fl-Ai9 iKO mice when compared with WT mice (Figure 6B–H) despite previous studies reporting TGF-β signaling to be important for later stages of adult neurogenesis (He et al., 2014; Gradari et al., 2021). In line with our data, we observed no changes in total tdTomato+ or DCX+ cells (Figure 6D and E) between control and DCXcreERT2-ALK5fl/fl-Ai9 iKO mice. Additionally, there were no significant differences in tdTomato+ / DCX+ double positive cells or tdTomato+ /NeuN+ double positive cells when comparing DCXcreERT2-ALK5fl/fl-Ai9 iKO to control mice (Figure 6F and G). Taken together, our data suggests that TGF-β signaling is not essential to the migration of adult-born neurons when ablated during the later stages of the neurogenic cascade in young adult mice.
Figure 7 | Late-stage ablation of TGF-β signaling in the DCXcreERT2-iSuRe-Alk5fl/fl iKO mice does not affect the migration of adult-born neurons in the dentate gyrus of the hippocampus.
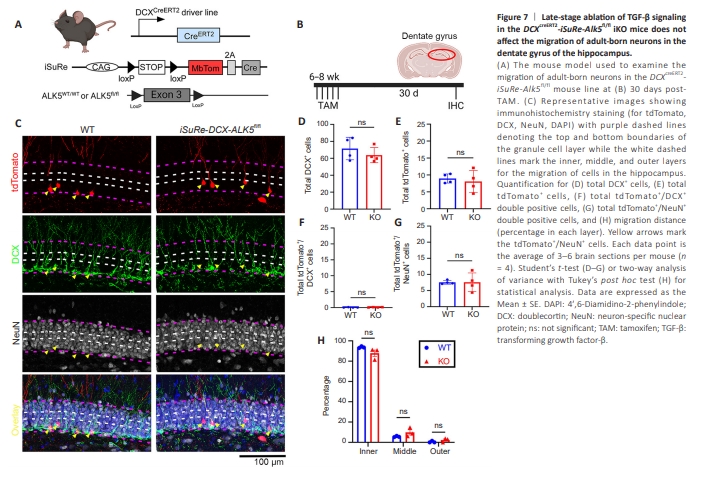
Substantial efforts have been made in recent years to enhance the accuracy and efficiency of Cre-mediated recombination and tracking of KO cells by fluorescent proteins. This is especially important to ensure that reporterpositive cells are truly tracking KO cells when antibodies are not available. Recent reports by others and us show that recombination of a single floxed reporter allele does not always demonstrate successful recombination of both alleles of the targeted genes, depending on the size of the floxed region (Faust et al., 2023; Bedolla et al., 2024b). To further enhance the rigor of our research and to increase the accuracy of the tracking of KO cells, we turned to a recently developed new tool, iSuRe mice developed by the Molecular Genetics of Angiogenesis from the Benedito lab (Fernández-Chacón et al., 2019). This mouse model contains an inducible dual reporter-cre that, upon recombination, produces a constitutively active CRE protein, with high recombination efficiency. Additionally, this novel iSuRe-Cre construct contains a Cre-dependent membranous tomato reporter that is expressed along with the constitutively active Cre, via P2A, enabling more accurate genetic labeling and tracking of the target cell population (Figure 7A). Recent work characterizing this model to ensure proper recombination efficiency and specificity was done when crossing the DCXcreERT2 driver line to generate the DCXcreERT2-iSuRe mouse line (Bedolla et al., 2024b). We therefore further generated the DCXcreERT2-iSuRe-ALK5fl/fl iKO mouse line and subjected these mice to the same treatment paradigm as the DCXcreERT2-ALK5fl/fl-Ai9 iKO mouse line and harvested mice 30 days post-TAM (Figure 7B). Similarly, our results show no differences in the migration of adult-born neurons in the DCXcreERT2- iSuRe-ALK5fl/fl iKO mice (Figure 7C and H). We also report no significant differences between total tdTomato+ or DCX+ cells as well as no change in tdTomato+ /DCX+ and tdTomato+ /NeuN+ cells (Figure 7C–G), suggesting that TGF-β signaling in DCX+ immature neurons are not critical for their survival or maturation. Taken together, our data supports that loss of TGF-β signaling in adult-born immature neuroblasts during later stages of adult neurogenesis is not required for the survival or proper migration of these newly born neurons, highlighting the need for precise cell-specific models to study TGF-β signaling in highly dynamic and complex systems.