脑损伤
-
Figure 2 | hNSCs characterization in vitro.
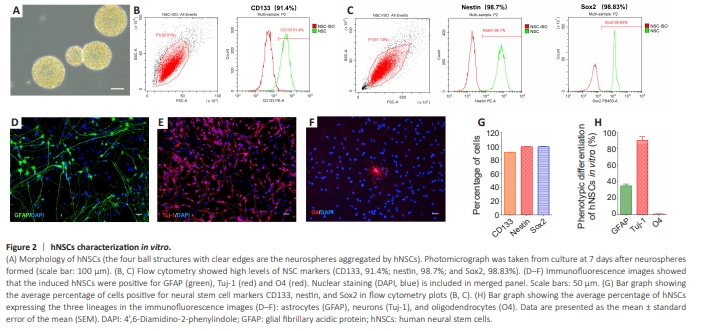
Human fetal NSCs formed clusters of small, round cells that grew into floating spheres (Figure 2A). Flow cytometry analysis of the single cells dissociated from the spheres showed the cells were positive for CD133, nestin, and Sox2 (Figure 2B, C, and G). Immunofluorescence staining that was performed after 10 days of induced differentiation showed that the hNSCs generated cells of all three lineages: astrocytes (GFAP: 34.9% ± 2.1%), neurons (Tuj-1: 89.8% ± 4.6%), and oligodendrocytes (O4: 0.9% ± 0.5%; Figure 2D–F and 2H). Thus, human fetal NSCs showed multilineage differentiation potential in our culture system.
Figure 5 | Cell survival and differentiation of hNSCs in the ischemic brain in rats.
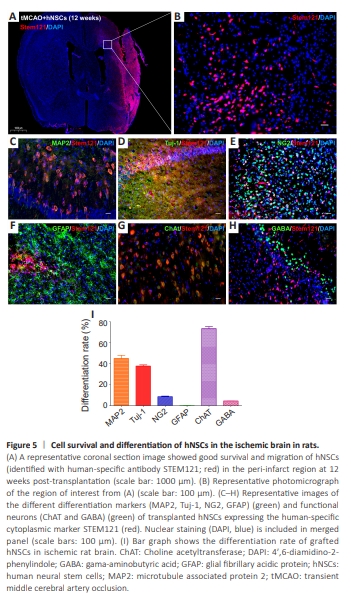
Immunohistochemical analysis was performed in the boundary zone of the ischemic core. Human NSCs transplanted into the rat striatum and cortex regions 7 days post-stroke showed robust survival (labeled with humanspecific antibody Stem121, red) and extensive migration toward the ischemic lesions at 12 weeks post-transplantation (Figure 5A and B). The abundant hNSCs survival within the striatum and cortex lesions reflected a large graft volume (Figure 5A). Thus, the results indicated that many hNSCs were able to survive close to the lesion as long as they were transplanted into nonischemic tissue. To assess the differentiation pattern of transplanted hNSCs, we used lineagespecific phenotypic markers and Stem121 double staining. The results showed that grafted NSCs (recognized with the human-specific marker Stem121) in the peri-infarct area were differentiated into MAP2-positive mature neurons, Tuj-1-positive neurons (including both immature and mature neurons), and NG2-positive oligodendrocytes, and residual cells were GFAP-positive glial cells (Figure 5C–F and I). To examine the specificity of the functional neurotransmitter, double immunohistochemistry of the grafted cells with either ChAT or GABA was performed. The vast majority of grafted neurons were excitatory, expressing ChAT, and only a few individual cells exhibited the GABAergic inhibitory phenotype at the 12-week time point (Figure 5G–I).
Figure 6 | hNSCs treatment changes the ratio of M1/M2 phenotype of microglia in injured brain.
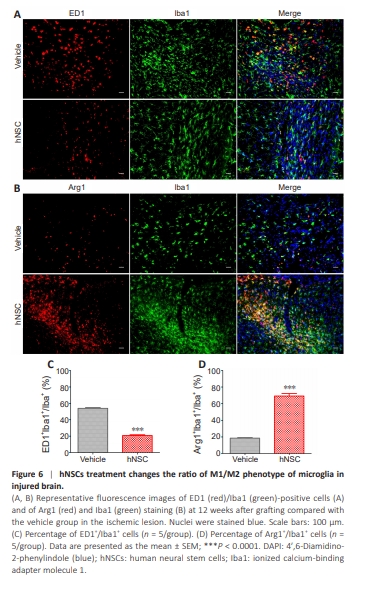
Double staining of CD68 (a classical proinflammatory marker for M1 microglia) or Arg1 (an alternative anti-inflammatory marker for M2 microglia) and Iba1 was performed 12 weeks after transplantation to examine the possible effects of hNSC transplantation on the phenotypic transition of M1/M2 microglia. The results indicated that the expression of Arg1+ /Iba1+ was significantly increased (P < 0.0001; Figure 6B and D), and the expression of ED1+ / Iba1+ was significantly decreased (P < 0.0001) in the hNSCs-treated group compared with the vehicle group (Figure 6A and C). This suggested that NSC transplantation shifted the M1/M2 phenotype of microglia and enhanced neuroprotective function.
Figure 7 | Grafted hNSCs receive direct monosynaptic inputs from host neurons in injured host tissue.
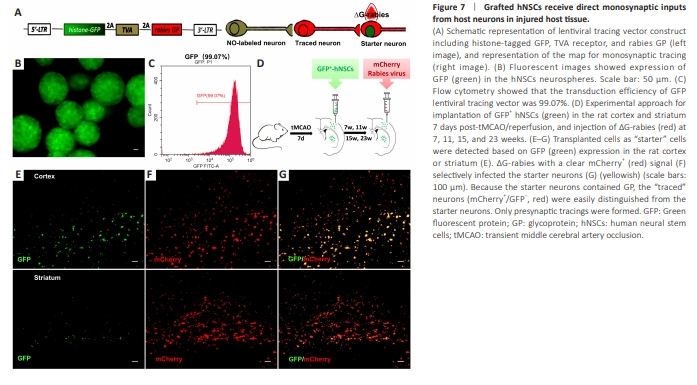
To analyze whether grafted neurons form connections with host neurons, a retrograde transsynaptic tracing strategy with ?G-rabies was used (Callaway and Luo, 2015). The hNSCs were stably transduced with the lentiviral tracing vector, which expressed a histone-tagged GFP, the TVA receptor necessary for selective infection with ?G-rabies, and rabies GP to allow for transsynaptic spread. The ?G-rabies with the foreign coat protein EnvA, in which the gene mCherry replaced the gene for GP in the viral genome, infected only the cells with the TVA receptor (Figure 7A). Fluorescence imaging (Figure 7B) showed that GFP containing lentiviral tracing vector transduced hNSCs with a transduction efficiency of 99.07% (Figure 7C). After 7 days post-tMCAO/R, rats were implanted with GFP-transduced hNSCs adjacent to the injury in thecortex or striatum (Figure 7D). Grafted hNSCs expressing nuclear GFP were identified as starter neurons (Figure 7E). After infection by ?G-rabies (Figure 7D), the targeted neurons were visualized by expression of GFP and mCherry (Figure 7F and G). In contrast, presynaptic traced neurons were identified by mCherry expression without GFP (Figure 7G).
Figure 9 | Electrophysiological properties of functional neurons in acute brain slices of rat cortex and striatum.
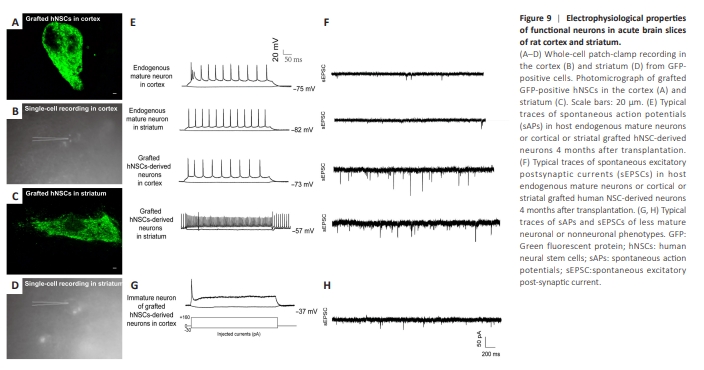
Finally, to determine whether cells generated from GFP-transfected hNSCs exhibited neuronal function, we examined the electrophysiological activity of cells using whole-cell patch-clamp recordings in acute brain slices at 16 weeks after transplantation into cortex and striatum adjacent to the lesion areas of tMCAO mice. We found that human GFP-positive cells in the cortex (Figure 9A and B) and striatum (Figure 9C and D) grafts generated current-induced action potentials and spontaneous action potentials (Figure 9E). Out of 113 GFP-positive cells identified from seven mice, approximately 72% of patched cells were functionally mature neurons with resting membrane potentials of approximately ?57 mV and ?73 mV in the cortex and striatum, respectively (Figure 9E and F), and produced spontaneous excitatory postsynaptic currents, suggesting functional maturation of grafted human neurons. Other cells with unstable, abnormal spiking patterns or resting membrane potentials (Figure 9G and H) in current-clamp mode were excluded from the analysis. Interestingly, no spontaneous inhibitory postsynaptic currents were observed.