脑损伤
-
Figure 2 | Characterization of hUC-MSCs.
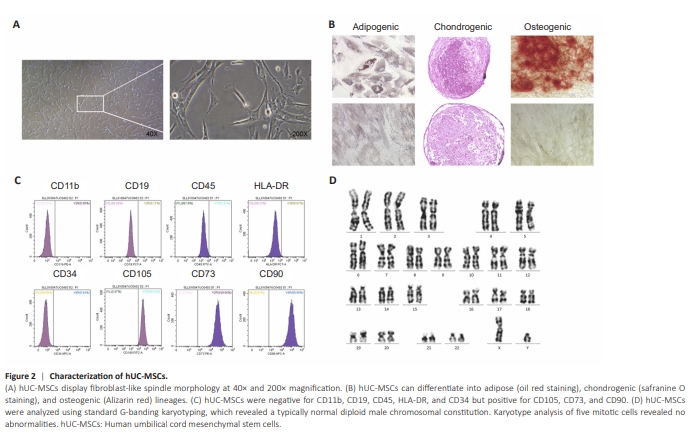
Cell surface markers of hUC-MSCs were detected by flow cytometry, and the results indicated low expression of CD19, CD34, CD45, HLA-DR, and CD11b, with an expression rate of ≤ 2%, while the cells had high expression of CD73, CD90, and CD105, with an expression rate of ≥ 95%. To further confirm the identity of the hUC-MSCs, differentiation tests were performed. The results showed that hUC-MSCs could successfully transdifferentiate into adipocytes, osteoblasts, and chondrocytes (Figure 2). In summary, these results indicate that hUC-MSCs are characterized by high purity and multidirectional differentiation capabilities. Additionally, the tests for bacteria, fungi, mycoplasma, hepatitis virus, and endotoxins were all negative (data not shown).
Figure 3 | Safety of the intrathecal transplantation of hUC-MSCs in pMCAO rats.
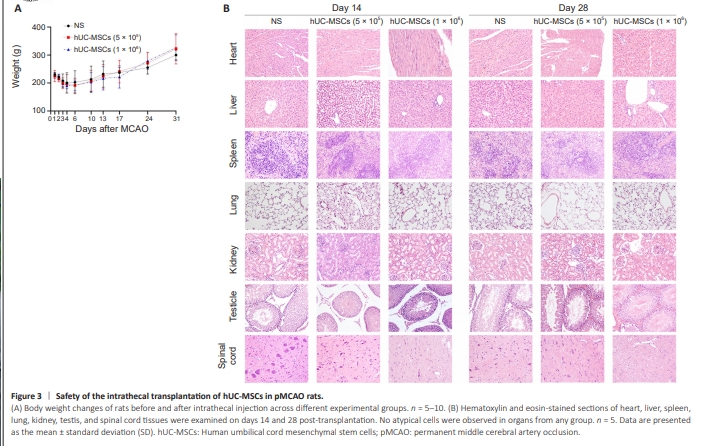
We assessed the safety of intrathecally transplanting hUC-MSCs for treatment of pMCAO rats by monitoring mortality rates, physical appearance or behavioral changes, body weight alterations, and pathological changes. Among the 50 rats, 16 died within 72 hours after pMCAO surgery because of complications such as anesthesia accidents, subarachnoid hemorrhage, and severe brain edema. After intrathecal injection, among surviving rats, the mortality rates of the low-dose hUC-MSC group, high-dose hUC-MSC group, and NS group were 16.67% (2/12 rats), 9.09% (1/11 rats), and 9.09% (1/11 rats), respectively. The results indicated that intrathecal administration of hUC-MSCs did not increase the risk of death in pMCAO rats. Compared with those in the NS group, intrathecal hUC-MSC-treated rats showed no notable deterioration in terms of physical appearance or behavioral activity, suggesting no occurrence of meningitis or major adverse events. No significant differences in body weight were observed among the different groups. All experimental rats experienced an initial period of weight loss post-MCAO surgery, which was subsequently followed by a gradual recovery in body weight over time (Figure 3A). At 14 and 28 days after intrathecal injection, H&E-stained images of the heart, liver, spleen, lungs, kidneys, testes, and spinal cord showed no evidence of atypical cells (Figure 3B).
Figure 5 | Migration of hUC-MSCs in the ischemic brain after intrathecal administration of hUC-MSCs in pMCAO rats.
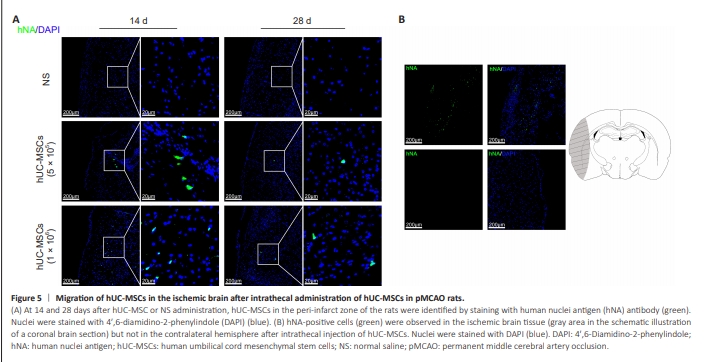
To determine whether intrathecally delivered hUC-MSCs can migrate into the ischemic brain, we treated hUC-MSCs with an hNA antibody. The migration of hUC-MSCs was observed at 14 and 28 days postintrathecal transplantation. The hNA-positive cells were found in the peri-infarct zone of the ischemic hemisphere in rats, and the number of hNA-positive cells significantly decreased over time (Figure 5A). Moreover, hNA-positive cells were not observed in the intact contralateral hemisphere after intrathecal delivery (Figure 5B).
Figure 6 | Immunofluorescence staining of neural cells after intrathecal transplantation of hUC-MSCs in pMCAO rats.
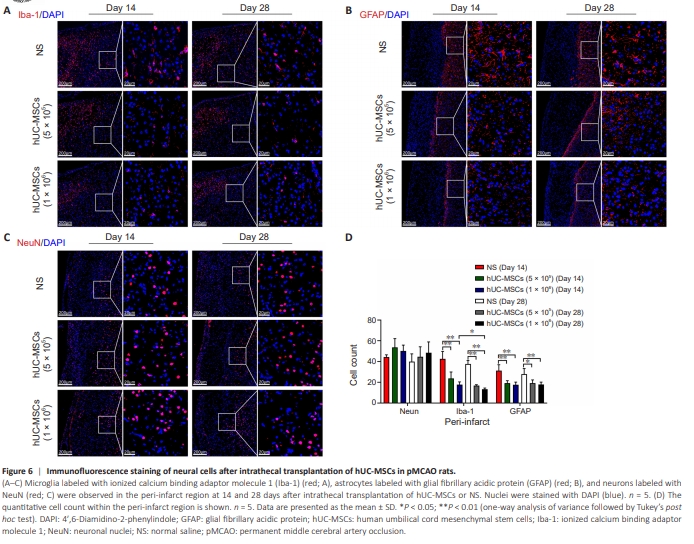
Neuron death is a cardinal pathological change in cerebral infarction, and inflammation aggravates brain damage after ischemic stroke. Immunofluorescence was used to evaluate the protein expression of Iba1, GFAP, and NeuN in the brain tissue of MCAO rats at 14 and 28 days after transplantation. Fourteen days after intrathecal administration, compared with that in the NS group, the numbers of Iba-1-positive cells in the periinfarct areas of the 5 × 105 hUC-MSC and 1 × 106 hUC-MSC groups showed a significant decrease (P < 0.05). The significant reduction in Iba-1-positive cells persisted until the 28th day, at which point the counts in both the low-dose and high-dose hUC-MSC groups were notably lower than that in the NS group (Figure 6A and D). Similarly, administration of 5 × 105 hUC-MSCs and 1 × 106 hUC-MSCs significantly reduced the number of GFAP-positive cells compared with that in the saline controls at 14 days after transplantation (P < 0.05). Similar results were observed at 28 days in the numbers of GFAP-positive cells in the low-dose hUC-MSC, high-dose hUC-MSC, and NS groups (P < 0.05) (Figure 6B and D). Although not significantly different, the number of NeuNpositive cells was greater in the low-dose hUC-MSC and high-dose hUC-MSC groups than in the NS group at 14 days (P > 0.05). Consistent with the findings at 14 days post-transplantation, a similar trend was shown among the groups at 28 days. The number of NeuN-positive cells was not significantly different among the low-dose hUC-MSC, high-dose hUC-MSC, and NS groups (P > 0.05) (Figure 6C and D). These data demonstrated that intrathecal administration of hUC-MSCs might improve ischemic brain repair by alleviating the local inflammatory response in the peri-infarct region. Moreover, although the differences observed did not reach statistical significance, hUC-MSC transplantation appears to promote neuronal survival.
Figure 7 | Effects of intrathecal hUC-MSC administration on cerebral microvessels, angiogenesis, and neurogenesis in pMCAO rats.
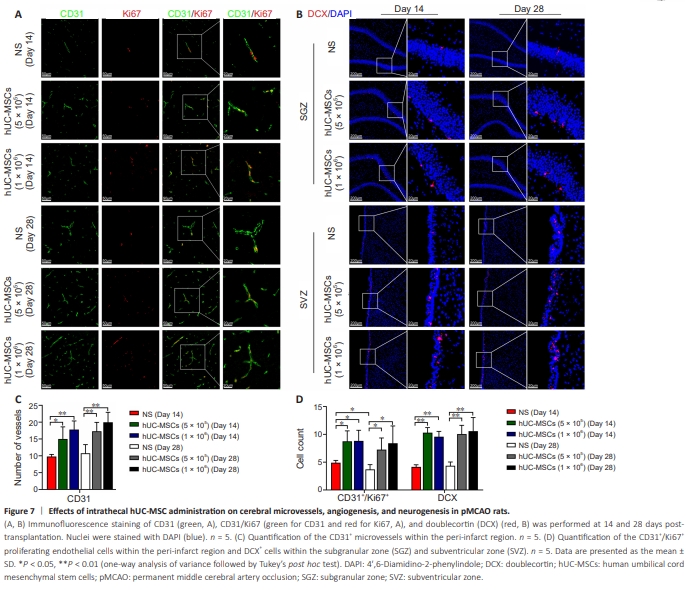
To examine whether hUC-MSC therapy increased vascular volume in the pMCAO model, we examined CD31-positive vessels in the peri-infarct region. Fourteen days after transplantation, the numbers of CD31-positive vessels in the low-dose hUC-MSC group and high-dose hUC-MSC group were significantly greater than that in the NS group (P < 0.05), suggesting an increase in vascular density after the intrathecal administration of hUC-MSCs. The significant differences in the numbers of CD31-positive vessels persisted until 28 days in the low-dose hUC-MSC, high-dose hUC-MSC, and NS groups(P < 0.01; Figure 7A and C). Double immunostaining with CD31 and Ki67 was used to investigate angiogenesis in the peri-infarct zone after ischemia. At 14 and 28 days post-transplantation, compared with NS, low-dose and high-dose hUC-MSCs significantly increased the number of CD31+ /Ki67+ proliferating endothelial cells (P < 0.05). Furthermore, the number of CD31+ /Ki67+ cells at 28 days was decreased compared with that at 14 days post-transplantation in all groups. However, only the NS group exhibited a statistically significant difference (P < 0.05; Figure 7A and D). Additionally, we investigated whether hUC-MSC therapy enhances neurogenesis in ischemic brains by detecting DCX, a marker of immature neurons. As expected, the numbers of DCXpositive cells in the SVZ and SGZ of the ipsilateral ischemic brain hemisphere were significantly greater in the 5 × 105 and 1 × 106 hUC-MSC groups at 14 days post-transplantation than that in the NS group (P < 0.01). Similarly, at 28 days post-transplantation, the numbers of DCX-positive cells in the 5 × 105 and 1 × 106 hUC-MSC groups were significantly higher than that in the NS group (P < 0.01; Figure 7B and D).
Figure 8 | Effects of intrathecal hUC-MSC administration on brain-derived neurotrophic factor (BDNF) secretion and expression in pMCAO rats.
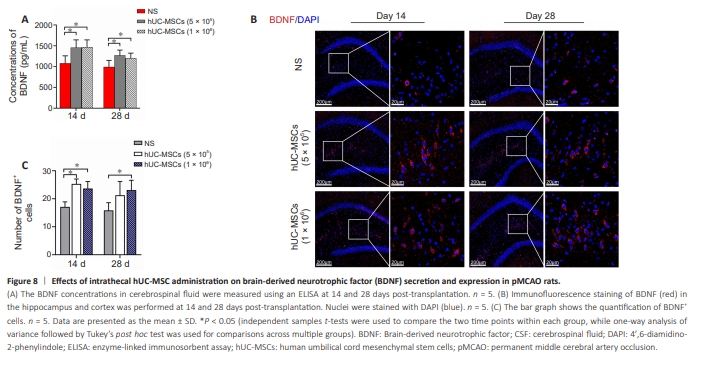
Fourteen days after intrathecal administration, the CSF BDNF concentrations in both the 5 × 105 hUC-MSC and 1 × 106 hUC-MSC groups were significantly greater than that in the NS group (P < 0.05). The significant difference persisted until the 28th day (P < 0.05; Figure 8A). Moreover, we quantified the distribution of BDNF in the peri-infarct cortex and hippocampus. There were significantly more BDNF-positive cells in both the 5 × 105 hUC-MSC group and the 1 × 106 hUC-MSC group than in the NS group at 14 days posttransplantation (P < 0.05). However, at 28 days, significantly greater BDNF expression was detected only in the 1 × 106 hUC-MSC compared with that in the NS group (P < 0.05; Figure 8B and C).