脊髓损伤
-
Figure 2 | Successful labeling of the monkey lumbosacral spinal cord via injection of CTB into the sciatic nerve.

The T12 to S2 segments of the spinal cord were serially cut into coronal sections and immunostained with an anti-CTB antibody. CTB+ signals could be clearly identified mainly in the lumbar (L)4–S1 spinal cord, in both the ventral and dorsal horns of gray matter but not in white matter (Figure 2A). In the ventral horn of the L6– S1 segments, CTB+ signals were more like cell somas, mainly in the L6 and L7 segments (Figure 2B). Quantification of the number of CTB-labeled cells confirmed that the L6 and L7 segments had the most CTB-labeled cells (Figure 2D). In the dorsal horn, CTB+ signals were densely intermingled, mainly in the L5–L7 segments and less so in the L4 and S1 segments (Figure 2C). Quantification of the area of CTB-labeled structures confirmed that the L5– L7 segments had the most CTB-labeled structures (Figure 2E). Taken together, these results suggested that the injection of CTB into the sciatic nerve labeled cells and structures of the lumbosacral spinal cord in a rhesus monkey.
Figure 3 | The CTB-labeled cells in the ventral horn of the monkey lumbosacral spinal cord were motor neurons.

To identify the CTB-labeled cells in the ventral horn, we first costained CTB with neuron-specific nuclear protein (NeuN), a neuronal marker. In the ventral horns of the L6–S1 segments, CTB-labeled somas were positive for NeuN (Figure 3A and D), suggesting that the CTB-labeled cells were neurons. Moreover, CTB-labeled neurons in the ventral horn of the L6–S1 segments were positive for choline acetyltransferase (ChAT) (Figure 3B, D, and E) andvesicular acetylcholine transporter (VAChT) (Figure 3C and F), indicating that the CTB-labeled cells were cholinergic neurons. CTB+ neurons were also positive for insulin gene enhanced protein 1 (ISLET1) (Figure 3G), a transcription factor of motor neurons. Quantification of the percentages of CTB+ NeuN+ , CTB+ ChAT+ and CTB+ VAChT+ cells among the NeuN+ , ChAT+ and VAChT+ cells (Figure 3H) indicated that intrasciatic nerve injection of CTB did not label all of the neurons, or even motor neurons, in the ventral horn. However, quantification of the percentages of CTB+ NeuN+ , CTB+ ChAT+ and CTB+ VAChT+ cells among the CTB+ cells (Figure 3I) indicated that the CTBlabeled cells in the ventral horn were exclusively motor neurons.
Figure 4 | CTB-labeled cells in the monkey spinal cord were not glial cells.
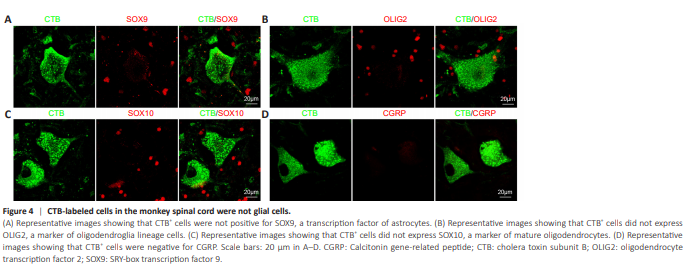
CTB-labeled somas were negative for astrocyte marker SOX9 (Figure 4A) and oligodendrocyte markers OLIG2 (Figure 4B) and SOX10 (Figure 4C), suggesting that no astrocytes or oligodendroglia were labeled with CTB. Although CTB-labeled rat spinal cord motor neurons have been reported to express calcitonin gene-related peptide (CGRP) (Cui et al., 2022), we did not observe CGRP expression in CTB-labeled monkey motor neurons (Figure 4D). Taken together, these results indicated that CTB injection into the sciatic nerve labeled motor neurons in the lumbosacral spinal cord in monkeys.
Figure 5 | The CTB-labeled structures in the dorsal horn of the monkey lumbosacral spinal cord were primary sensory afferents.
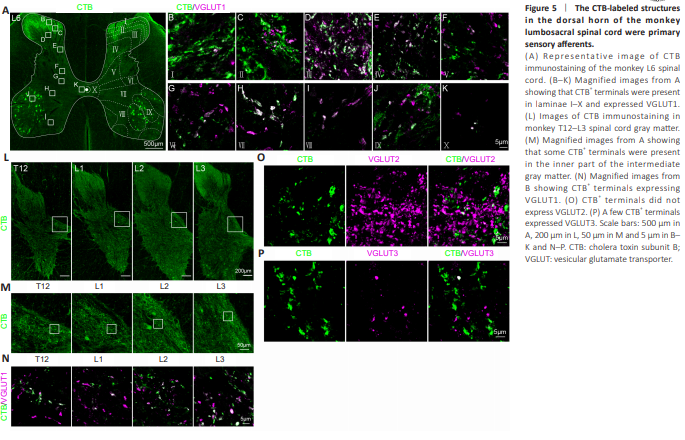
We next determined the identity of CTB-labeled structures in the dorsal horn. Although CTB-labeled structures were mostly located in the dorsal horn (Figure 5A), at higher magnification, CTB-labeled axon terminals and punctate-like structures were also observed in all laminae of gray matter, including laminae I to X (Figure 5B–K). Moreover, these CTB+ terminals expressed vesicular glutamate transporter 1 (VGLUT1) (Figure 5B–K). Although CTB+ terminals were mostly present in the L4–S1 segments (Figure 2E), higher magnification of the inner part of the intermediate gray matter of the T12–L3 segments (Figure 5L) showed some CTB+ terminals (Figure 5M) that expressed VGLUT1 (Figure 5N). CTB+ terminals did not express VGLUT2 (Figure 5O), and a few CTB+ terminals expressed VGLUT3 (Figure 5P).
Figure 6 | Presence of CTB-labeled primary sensory afferents in the monkey T12–L3 spinal cord.

We next investigated whether CTB could label other primary sensory afferents. Many CGRP+ nonmyelinated peptidergic C fibers were present in the dorsal horn of the L4–S1 segments (Figure 6A), and a few of these fibers were labeled by CTB (Figure 6B). Isolectin B4 (IB4)+ nonmyelinated nonpeptidergic C fibers were present only in the L4 and S1 segments and were not observed in the L5–L7 segments (Figure 6A). In the L4 segments, IB4+ fibers were not labeled by CTB (Figure 6C). Although there are reports of CTB labeling in galanin+ and neuropeptide Y (NPY)+ peptidergic fibers in the rat spinal cord (Bao et al., 2002; Shehab et al., 2003), we observed that no monkey galanin+ (Figure 6D) and few NPY+ (Figure 6E) fibers were labeled with CTB. Taken together, these results suggested that CTB injection into the sciatic nerve labeled myelinated primary sensory afferents in the lumbosacral spinal cord in monkeys.
Figure 7 | CTB-labeled primary sensory afferents form synapses with multiple types of monkey spinal neurons.

Primary sensory afferents form synapses with multiple sensory and motorrelated neurons in the rodent spinal cord. Thus, we examined the postsynaptic targets of monkey primary sensory afferents labeled with CTB. Monkey CTB+ terminals expressed the presynaptic marker synaptophysin (Syn) (Figure 7A) and were in close proximity to the postsynaptic marker Homer1 (Figure 7B and C), suggesting that CTB+ terminals indeed formed synapses. Then, we checked the postsynaptic neurons of monkey primary sensory afferents along the dorsal ventral axis. Monkey CTB+ terminals formed synapses with both LMX1B+ excitatory (Figure 7D) and LHX1+ inhibitory (Figure 7E) interneurons. In the dorsal gray matter, CTB+ terminals, including calretinin (CR)+ (Figure 7F) and calbindin (CB)+ (Figure 7G) neurons, formed synapses with sensoryassociated interneurons. In the ventral gray matter, CTB+ terminals formedsynapses with motor-related interneurons, specifically FOXP2+ (Figure 7H) and SATB1+ (Figure 7I) neurons. Moreover, in the motor neuron pool, CTB+ terminals appeared to contact motor neurons (Figure 7J) and form synapses with motor neurons (Figure 7K). Finally, monkey CTB+ terminals were innervated by glutamate decarboxylase 65 (GAD65)+ inhibitory synapses (Figure 7L). Taken together, these results suggested that CTB-labeled monkey primary sensory afferents formed synapses with multiple types of spinal neurons.
Figure 8 | Injection of CTB into the sciatic nerve did not cause glial activation or apoptosis of motor neurons in the monkey spinal cord.

Because CTB is a protein produced by cholera toxin that can cause inflammation and lead to cell death, we investigated the safety of injecting CTB into monkey sciatic nerves. In both the dorsal and ventral horns where CTB-labeled primary sensory afferents and motor neurons were present, ionized calcium binding adaptor molecule 1 (IBA1)+ microglia had small cell somas and thin ramified processes, with evident expression of the homeostatic marker P2RY12 (Figure 8A). However, around the SCI lesion site, IBA1+ microglia were round and had larger cell bodies without processes, and the expression of P2RY12 was almost completely absent (Figure 8A). Quantification of fluorescence intensity showed an increasing trend in IBA1 expression and a decreasing trend in P2RY12 in the spinal lesion site, compared to the dorsal and ventral horns with dense CTB signals (Figure 8E and F). However, these findings are descriptive in nature and lack statistical analysis. Thus, microglia around CTB+ terminals and motor neurons appeared to be inactive. Similarly, in the dorsal and ventral horns where CTB+ terminals and motor neurons were present, glial fibrillary acidic protein (GFAP)+ astrocytes exhibited many thin and ramified processes (Figure 8B). However, around the SCI lesion site, the GFAP+ astrocytes had thick and parallel processes (Figure 8B), suggesting that they were activated and formed astroglial scars. Fluorescence analysis suggested a trend of elevated GFAP expression in the spinal lesion site, compared with the dorsal and ventral horns with dense CTB signals (Figure 8G). Thus, these findings suggest that astrocytes around CTB+ terminals and motor neurons were also quiescent. Finally, although cleaved caspase-3 (c-Caspas-3)+ cells that were undergoing apoptosis were present (Figure 8C), CTB-labeled motor neurons did not express c-Caspase-3 (Figure 8D), indicating that CTB+ motor neurons were not undergoing apoptosis. Taken together, these results suggested that CTB injection into the monkey sciatic nerve did not cause glial activation or apoptosis of motor neurons in the spinal cord.