视神经损伤
-
Figure 2 | The diameter and concentration of particles present in EV preparation were determined by nanoparticle analysis and immunoblotting with EV-specific markers.
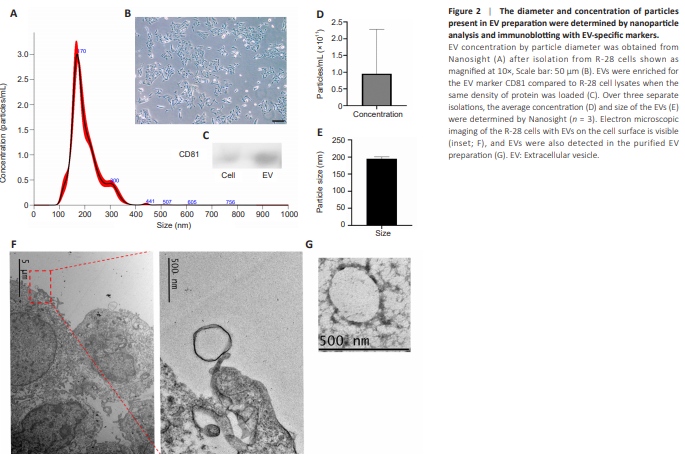
EVs were isolated from R-28 cell line (Seigel, 2014) which showed a classic elongated spindle shape (Figure 2B). Nanoparticle tracking analysis revealed that the nanoparticle size in R-28 cell-derived EV preparation was relatively homogenous with sizes ranging from 100 and 400 nm. The mean particle size was determined as 200.8 ± 1.1 nm and the mode was 171.2 ± 3.3 nm (Figure 2E). The average concentration/yield was 1 × 1011 particles in 1:100 diluted EV solution, a typical isolation from 180 ml media (assuming 15 mL of media per 6 × 106 cells in a T75 flask; Figure 2A and D). Immunoblotting displayed higher expression of EV phenotypic marker CD81 in EVs, with relatively less protein expression in cell lysate. Cell lysates serve as a useful negative control as within the same quantity of protein, they will have significantly less CD81, which is expected to be enriched in samples with purified EVs (Abello et al., 2019; Arteaga-Blanco et al., 2020; Figure 2C). Transmission electron microscopy imaging of the R28 cells (Figure 2F) and of the purified EV preparation (Figure 2G) revealed the presence of nanoparticles of the expected size. These results suggest that EVs were secreted from R-28 cell line with proper characteristics.
Figure 3 | In vitro testing of R-28 cell-derived EVs on RGC survival and regeneration.
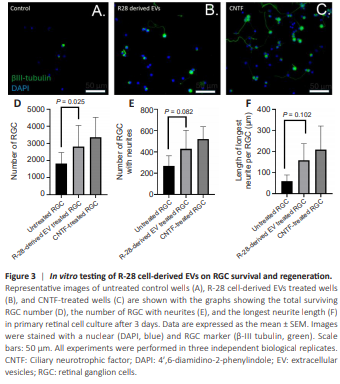
We investigated the neuroprotective effect of R-28-derived EVs (2.5 × 109 particles) in primary retinal cell culture (Figure 3A–C). The treatment showed significantly improved RGC survival (2814.33 ± 712.59 RGCs/well) compared to untreated control wells (1827 ± 368.38 RGCs/well, P = 0.025), in which 35.08% more RGC were detected in EV-treated wells (Figure 3D). We also investigated the neuritogenesis effect of R-28 cell-derived EVs, which proved to be similarly regenerative to that of CNTF treatment, with the number of RGCs with regenerating neurites in R-28 cell-derived EVs treated wells approaching significance compared to untreated wells (P = 0.082; Figure 3E). Moreover, we also compared the length of the neurites, which showed a similar pattern to the number of neurons with neurites. The neurite length of R-28 cell-derived EVs treated RGCs was on average, longer than the control group (58.6 9 ± 17.36 μm and 157.8 ± 45.86 μm, P = 0.102 respectively; Figure 3F) and was similar to CNTF treated RGCs, again approaching significance. Overall, these results indicate that R-28 cell-derived EVs protected RGCs from degeneration.
Figure 4 | R-28 cell-derived EVs promote human ESC-derived RGC survival in vitro.
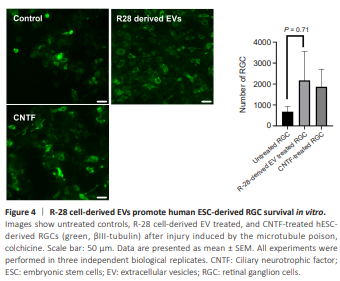
R-28 cell line is derived from rat cells (Seigel, 2014), and their efficacy on rat retinal cells may not be recapitulated when used on human RGCs or may even show negative effects. Therefore, we investigated EVs on human RGCs differentiated from human ESCs to detect possible differences. R-28 cell-derived EVs are neuroprotective on hESC-derived RGCs (injured with colchicine) compared to untreated hESC-derived RGCs injured with colchicine (2178.67 ± 795.43 RGCs/well, 682.33 ± 145.89 RGCs/well, respectively, n = 3, P = 0.071) (Figure 4). Interestingly, these EVs trended to be more neuroprotective than CNTF with 16.65% more surviving RGCs. In summary, these results indicate that R-28 cell-derived EVs are neuroprotective even in human-derived RGCs and did not show any apparent neurotoxic effects.
Figure 5 | R-28 cell-derived EVs show protective trend for RGCs in a chronic glaucoma model.
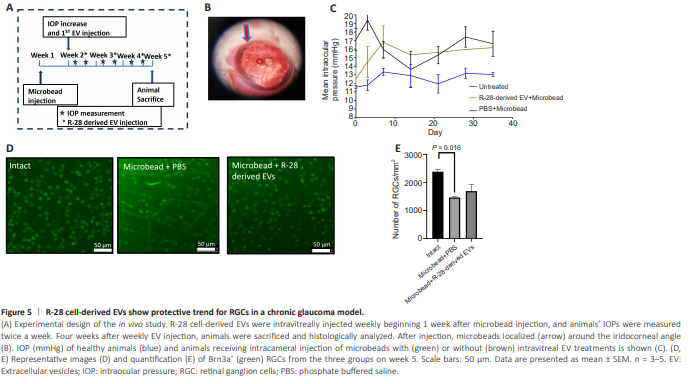
We investigated the effects of R-28 cell-derived EVs after weekly intravitreal injection into an in vivo rat model of glaucoma (Figure 5A). Microbead intracameral injection (Figure 5B) increased IOP to 17.1 ± 1.00 mmHg on day 7 (Figure 5C) whereas IOP remained low in the control group (11.57 ± 0.16 mmHg). The increased IOP was sustained until the end of the experiment (day 36: 16.17 ± 2.20 mmHg). The number of Brn3a+ RGCs after microbead injection was significantly decreased (1463 ± 58.52 RGCs/mm2 ) compared to the intact group (2383 ± 80.725 RGCs/mm2 , P = 0.002; Figure 5D and E). Weekly injection of 2.5 × 109 R-28 cell-derived EV elicited some level of neuroprotection on Brna3a+ RGCs (1690.25 ± 239.4 RGCs/mm2 , P = 0.3991). Together these results provide insights into the neuroprotective trend of R-28 cell-derived EVs in vivo.
Figure 6 | Optic nerve damage in glaucomatous eyes after EV treatment.
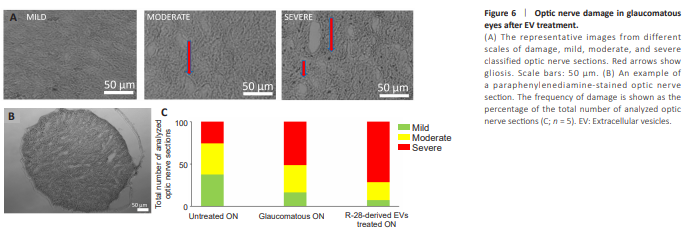
To investigate if R-28 cell-derived EVs preserve RGC axons in the optic nerve, we stained optic nerve sections with PPD staining, and scored damage on the optic nerve as mild, moderate, or severe (Figure 6A and B). The percentage of axons that received a severe grading was higher in the glaucomatous optic nerve group (16.21%, 32.43%, and 51.35%, respectively), compared to the uninjured group (37.59%, 36.84%, and 25.56% respectively; Figure 6C) but no difference was observed for the R-28 cell-derived EV treated group. In summary, these results suggest that R-28 cell-derived EVs failed to protect the ON.