脑损伤
-
Figure 1 | Generation of chemical induced MNs from hiPSC (ciMNs).
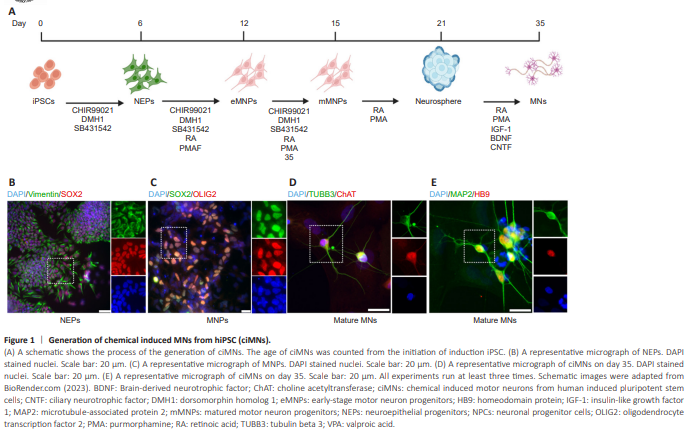
The process of generating ciMNs replicates the developmental stages during MN differentiation. It spans approximately 5 weeks and comprises several distinct stages, including the expansion of hiPSCs, the formation of NEPs, the transition to eMNPs, the maturation into mMNPs, the formation of neuron spheres, and ultimately the differentiation into mature MNs (Figure 1A). At each stage, cell identity can be confirmed by the expression of specific markers. For instance, cytoplasmic vimentin and nuclear SOX2 are highly expressed in NEPs (Figure 1B), and the co-expression of nuclear SOX2 and OLIG2 defines the MNP identity (Figure 1C). In addition to the generic neuron markers of TUBB3 and MAP2, the identity of MNs can be validated by the nuclear expression of MNX1 (HB9) in early development stages and ChAT in late maturation (Figure 1D and E). These neurons exhibited typical MN morphology with long axons. Based on the typical MN morphology and the expression of MN-specific markers HB9 and ChAT, we concluded that we have successfully generated hiPSC-MNs using chemical induction methods.
Figure 2 | Characterization of ciMNs.
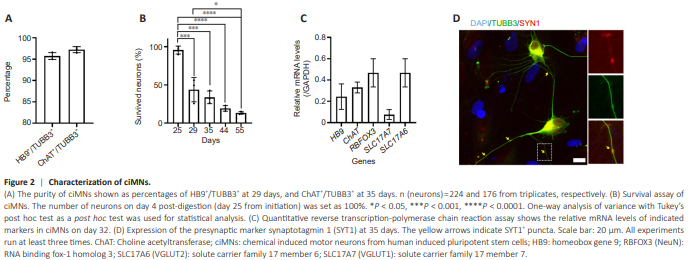
To assess the purity of ciMNs, we quantified the ratios of HB9-positive cells or CHAT-positive to TUBB3-positive cells (all neurons). Both ratios (HB9+ / TUBB3+ and ChAT+ /TUBB3+ ) exceed 95% (Figure 2A), indicating almost all neurons exhibit MN identity. Achieving survival poses a significant challenge when culturing induced MNs, particularly in achieving late maturation stages. To further characterize the survival of ciMNs, we conducted a survival assay, following an established protocol (Ding et al., 2021). CiMNs were replated onto coverslips with a monolayer of astrocytes, and the number of neurons on day 25 (4 days after replating) was set as 100%. The survival of neurons was calculated at various time points based on TUBB3 staining. Although the newly generated ciMNs were co-cultured with astrocytes, which are known to significantly support neuronal survival, the number of surviving neurons decreased noticeably over time. Approximately 50% of neurons died within the first few days after replating (day 8 vs. day 4 after replating) (Figure 2B). On day 55, only about 15% of the originally seeded neurons remained viable (Figure 2B), suggesting the significant challenge of maintaining long-term neuron cultures. To further characterize the maturation of ciMNs, RNA was collected from pure cultured ciMNs at day 32 and subjected to quantitative PCR. Constantly, ciMNs robustly expressed the neuronal marker RNA binding fox-1 homolog 3 (RBFOX3) and MN markers of HB9 and CHAT (Figure 2C). Interestingly, at this stage, ciMNs exhibited significant expression of genes encoding subunits of vesicular glutamate transporters solute carrier family 17 member 7 (SLC17A7) and SLC17A6 (Figure 2C), indicating their progression towards functional maturation. On day 35, ciMNs highly expressed the presynaptic marker SYT1 and exhibited the synaptic puncta within neuronal processes (Figure 2D), suggesting that ciMNs had achieved functional maturation. These results indicate that ciMNs exhibited typical MN morphology, expressed specific MN markers, and gradually achieved functional maturation around day 35, although the survival rate and total yield were moderate.
Figure 3 | Generation and characterization of Lenti-MNs through lentiviral delivery transcription factors.
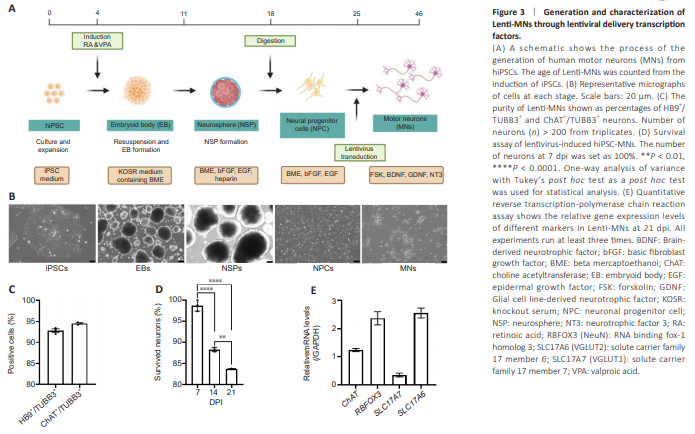
In contrast, the lentiviral method for generating hiPSC-MNs (Lenti-MNs) showed a different trajectory. Initially, hiPSCs are induced to form embryoid bodies (EBs), which then differentiate into NSPs, and subsequently into NPCs (Figure 3A). At each stage, the cells show distinct morphological characteristics under different defined culture conditions (Figure 3B) (Sepehrimanesh and Ding, 2020; Akter et al., 2022; Akter and Ding, 2022). Subsequently (on around day 25), NPCs will be transduced with lentivirus expressing three transcription factors ISL-1, Neurog-2, and LHX3. These three factors are necessary and sufficient to induce the differentiation of NPCs into MNs (Sepehrimanesh and Ding, 2020). As early as 3 days post-viral infection (dpi), the infected NPCs exhibit an MN-like morphology with pronounced exons (Figure 3B). These Lenti-MNs robustly express MN markers HB9 and ChAT within 14 dpi and reach full maturation around 21 dpi (around 46 days from hiPSC culture). The purity of Lenti-MNs is over 90%, based on the ratios of HB9+ /TUBB3+ and ChAT+ /TUBB3+ (Figure 3C). We quantified the surviving neurons at 1, 2, and 3 weeks post-viral infection (wpi), with the numbers of neurons at 1 wpi set at 100%. We found that approximately 50% of Lenti-MNs survived at 2 wpi and about 20% survived at 3 wpi (Figure 3D). In addition to MN markers (ChAT), Lenti-MNs also robustly express maturation markers (RBFOX3, SLC17A7, and SLC17A6) at 21 dpi (Figure 3E), validating the MN identity and maturation.
Figure 4 | Comparison of two protocols in the generation of human induced pluripotent stem cell (hiPSC)–derived motor neurons (MNs).
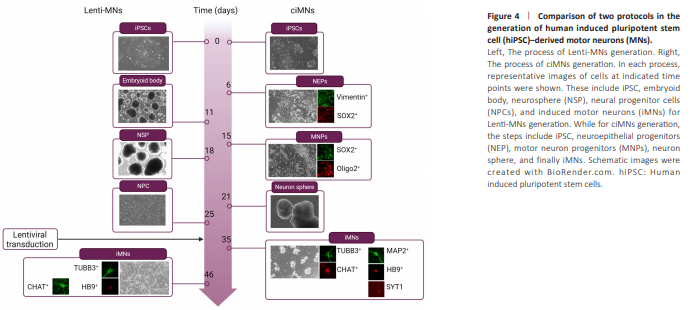
The comparative analysis of the timelines for the two protocols revealed that both methods took more than one month from hiPSCs to mature MNs. Compared to Lenti-MNs, ciMNs achieved maturation relatively faster (35 days vs. 46 days) (Figure 4). However, the lentivirus-based method possesses a unique advantage that cells at the NPC stage can be prepared as frozen stocks. Depending on the desired experiments, these NPC frozen stocks can be thawed and expanded, rendering this protocol more feasible and manageable. In our attempts with the ciMNs method, we tried freezing and subsequently culturing them again from various intermediate stages, but the results did not meet our expectations.