神经损伤与修复
-
Figure 2|Cardinal features of inflammation in CPIP mice.
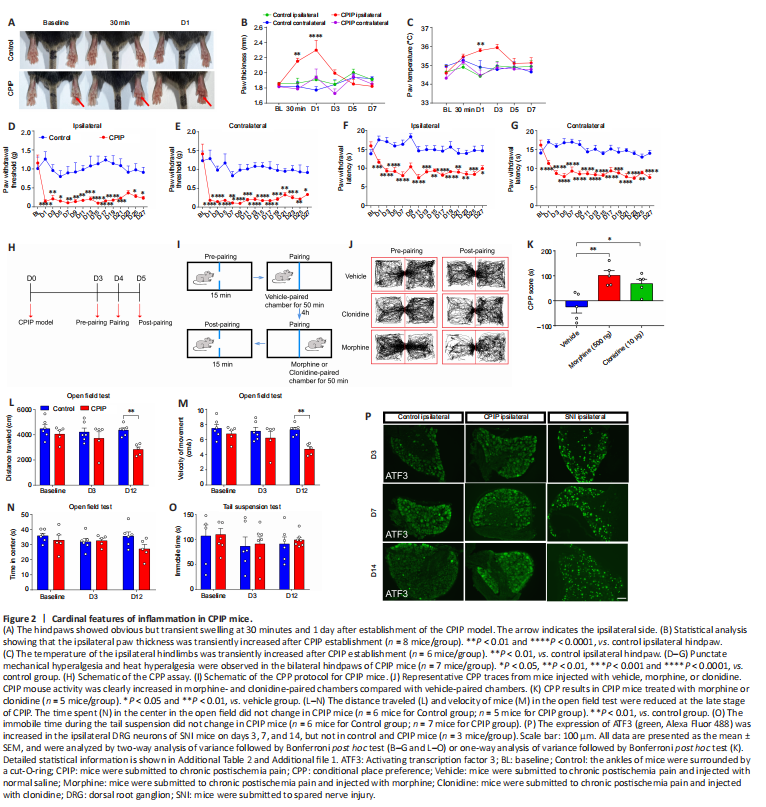
We first established the CPIP mouse model by inducing prolonged hindpaw ischemia-reperfusion to mimic clinical CRPS-1 (Chen et al., 2020a; De Logu et al., 2020). The CPIP mice exhibited cardinal features of inflammation, including redness, swelling, heat, and nociceptive behavior. The ipsilateral hindpaws of CPIP mice showed transient but obvious redness, edema, and an increased temperature 30 minutes after reperfusion, and these effects lasted for 1 day (Figure 2A–C). Furthermore, the CPIP mice showed clear, persistent heat hyperalgesia and punctate mechanical hyperalgesia in the bilateral hindpaws that was maintained throughout the 4-week experimental period (Figure 2D–G). We also performed a CPP assay, which is widely used to measure ongoing or spontaneous pain (Tajerian et al., 2015; Donnelly et al., 2021), to detect nonreflexive nociception in the CPIP mice. A single injection of morphine or clonidine induced obvious CPP in the CPIP mice (Figure 2H–K), suggesting the existence of both reflexive and nonreflexive pain in CPIP mice.
Next, an open field test and tail suspension test were performed to observe motor function and mood, respectively. We found that the distance traveled and velocity of movement in the open field test were slightly decreased at the late stage (day 12) but not at the early stage (day 3) in CPIP mice compared with sham control mice (Figure 2L and M). However, no differences between CPIP and control mice in terms of the time spent in the center of the open field or in the immobility time during the tail suspension test were observed, suggesting that CPIP mice did not experience anxiety or depression (Figure 2N and O). Furthermore, expression of ATF3, an indicator of peripheral neuron injury (Wang et al., 2021), did not differ between CPIP and control mice (Figure 2P). In contrast, ATF3 expression was increased in the DRG of a mouse model of spared nerve injury (SNI) (Figure 2P). Thus, CPIP mice exhibited obvious peripheral inflammation but no nerve injury, consistent with patients with CRPS-1 (Shim et al., 2019).
Figure 3|Neuroinflammation and central sensitization in the spinal cord of CPIP mice.
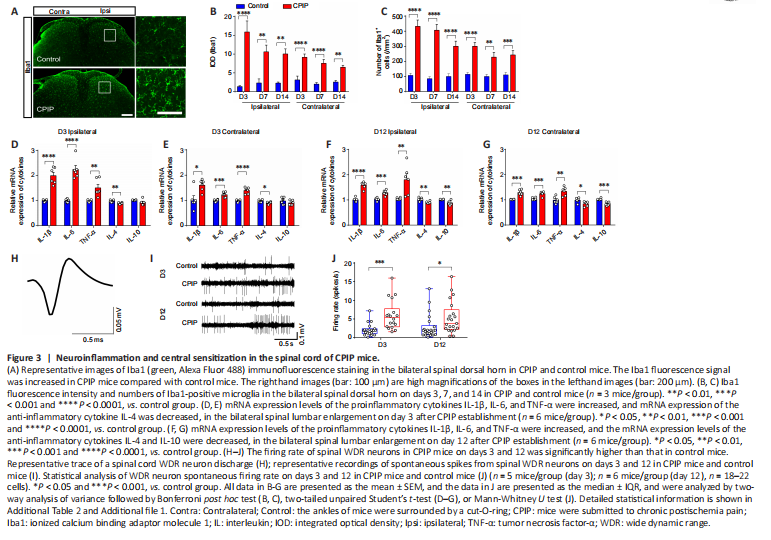
Next, we examined microglia activation in the spinal cord using the microglial marker Iba1 (Chen et al., 2020b) to determine whether CPIP causes neuroinflammation. Iba1 fluorescence intensity and the number of Iba1-positive cells were significantly increased in the spinal cord dorsal horn of CPIP mice compared with control mice on days 3, 7, and 14 (Figure 3A–C). Moreover, the mRNA levels of the proinflammatory cytokines TNF-α, IL-1β, and IL-6 were higher in the spinal cord dorsal horn of CPIP mice at both the early stage (day 3) and late stage (day 12) compared with control mice (Figure 3D–G). In contrast, the mRNA level of IL-4, an anti-inflammatory cytokine (Kedong et al., 2020), was lower at both the early and late stages of CPIP, while the expression of another anti-inflammatory cytokine, IL-10 (Han et al., 2022), was lower at only the later stage of CPIP, compared with control mice (Figure 3D–G). Moreover, the spontaneous firing rate of spinal wide dynamic range (WDR) neurons in CPIP mice was higher than that in control mice (Figure 3H–J). These results suggest that CPIP mice exhibit neuroinflammation and central sensitization in the spinal cord, consistent with previous studies (Tang et al., 2018; Shim et al., 2019).
Figure 5|HIS abolishes neuroinflammation and central sensitization in the spinal cord of CPIP mice.
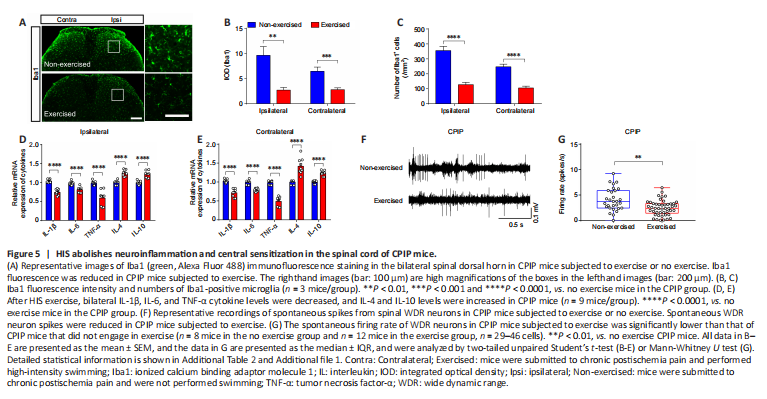
Next, we asked whether HIS attenuated CPIP-induced neuroinflammation and central sensitization. To assess this, we examined spinal Iba1 immunofluorescence staining in CPIP mice that engaged in HIS or no exercise. Iba1 fluorescence intensity and the number of Iba1-positive cells were reduced in the HIS group compared with the no exercise group (Figure 5A–C). In addition, TNF-α, IL-1β, and IL-6 levels were lower, and IL-4 and IL-10 levels were higher, in the bilateral spinal cord in the HIS CPIP group compared with the no exercise CPIP group (Figure 5D and E). We also observed lower spontaneous firing of WDR neurons in the HIS CPIP group compared with the no exercise CPIP group (Figure 5F and G). Based on these results, HIS exercise attenuates nociception by ameliorating neuroinflammation and decreasing the hyperactivity of spinal dorsal horn neurons.
Figure 7|The RvE1-ChemR23 axis is involved in HIS-induced analgesia.
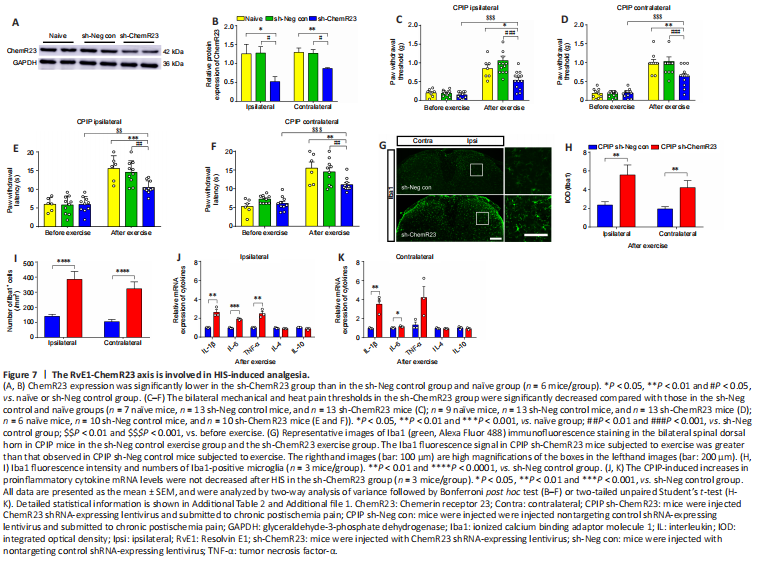
To explore the role of the RvE1-ChemR23 axis in HIS-induced analgesia, we next injected mice with an RNA interference virus to knock down ChemR23 expression in the spinal cord. As expected, ChemR23 protein expression was lower in the sh-ChemR23 group than in the control virus group, verifying that ChemR23 was downregulated in the spinal cord (Figure 7A and B). After successfully knocking down ChemR23 expression in the spinal cord, we found that the analgesic effect of HIS was substantially inhibited in the sh-ChemR23 group compared with the na?ve and sh-Neg control groups (Figure 7C–F). In addition, the HIS-induced inhibition of microglial activation in CPIP mice and the decreased levels of IL-1β, IL-6, and TNF-α in the HIS CPIP group were all blocked in the sh-ChemR23 group compared with the sh-Neg control group (Figure 7G–K). Thus, the RvE1-ChemR23 axis may mediate HIS-induced analgesia and inhibit neuroinflammation in CPIP mice.
Figure 8|Exogenous RvE1 inhibits CPIP-induced nociception, neuroinflammation, and central sensitization.
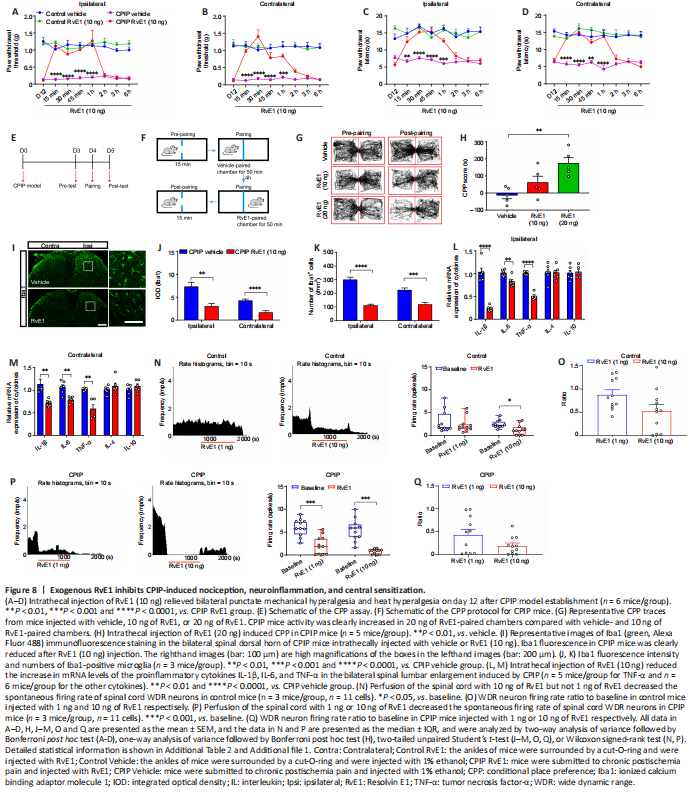
RvE1 was intrathecally injected to test the analgesic effect of exogenous RvE1 on nociception in CPIP mice. Intrathecal injection of RvE1 (10 ng) reduced the response to mechanical and heat-induced pain in the bilateral hindpaws of CPIP mice (Figure 8A–D). In addition, RvE1 (20 ng) induced CPP in CPIP mice, indicating that RvE1 suppresses ongoing pain (Figure 8E–H). RvE1 also exerted marked anti-neuroinflammatory effects, as indicated by reduced microglial activation and lower levels of the proinflammatory cytokines TNF-α, IL-1β, and IL-6 in the bilateral spinal cord (Figure 8I–M). However, RvE1 had no effect on the expression of the anti-inflammatory cytokines IL-4 and IL-10 (Figure 8L and M). Compared with control mice, the inhibitory effect of RvE1 on WDR neuron spontaneous firing was more robust in CPIP mice (Figure 8N–Q). These data suggested that spinal cord supplementation with exogenous RvE1 simulates the effect of HIS on decreasing nociceptive behavior, neuroinflammation, and central sensitization in CPIP mice.