脑损伤
-
Figure 5 | OPC ferroptosis and demyelination in the peri-infarct area of the corpus callosum after MCAO/R is rescued by Fer-1.

First, we examined immunostaining changes of MBP and the OL marker, OLIG2, in the peri-infarct region of the corpus callosum at various time points following MCAO/R to assess myelin sheath damage and OLs. Compared with the sham group, MBP mean fluorescence intensity decreased from day 1 to day 7, with the lowest point on day 3 (Figure 5A and B). Quantifying OLIG2 revealed a decrease in the total amount of OLIG2+ cells after MCAO/R (Figure 5A and C). Lipid peroxidation is a pivotal contributor to ferroptosis (Costa et al., 2023), therefore we assessed MDA levels, a lipid hydroperoxide end-product (Wang et al., 2023a), in the corpus callosum. The findings showed a marked increase in MDA content after MCAO/R. Intraperitoneal administration of Fer-1 effectively mitigated this MDA upsurge (Figure 5D). Subsequently, we conducted immunofluorescence staining targeting the OPC marker, NG2, and found a significant reduction in OPCs after MCAO/R. Fer1 treatment partially rescued the reduced number of NG2+ cells (Figure 5E and F). Further, we injected PI into the lateral ventricles of mice. This showed that the distribution of PI+ cells increased in the MCAO/R group, with apparent co-localization of these cells and OPCs. This effect was significantly inhibited by Fer-1 (Figure 5E and G). Lastly, LFB staining of brain sections showed that Fer-1 significantly restored myelin coverage in the corpus callosum (Figure 5H and I).
Figure 6 | Increased PLIN2 levels induce OPC ferroptosis after MCAO/R.

We combined the data of Module 1 from PPI (Figure 1F), the MEturquoise Module from WGCNA (Figure 2C), and the leading-edge subset from scRNA-seq analysis (Figure 4G), resulting in the identification of four key genes: Plin2; hypoxia inducible factor 1, alpha subunit; cytochrome b-245, beta polypeptide; and lysosomal-associated membrane protein 2 (Figure 6A). Plin2 emerged as a core gene within the MEturquoise Module (MM > 0.8 and GS > 0.2; Figure 2C) and was ranked highly in the leading-edge subset (Figure 4G and Additional Table 4), indicating its potential significance in OPC ferroptosis following cerebral ischemia. Consequently, we established correlation between Plin2 and functional gene sets. Notably, Plin2 expression showed a significant positive correlation with lipid peroxidation (r = 0.548, P = 0.019; Figure 6B) and a substantial negative correlation with OL development (r = –0.664, P = 0.003) and myelin sheath formation (r = –0.748, P < 0.001; Figure 6C and D). This suggests that Plin2 is critical in initiating OPC ferroptosis and impairing OPC functionality. Next, we examined changes in PLIN2 levels and its distribution across the brain after MCAO/R. Immunofluorescence analyses indicated a significant increase in PLIN2 levels in the periinfarct area, beginning on day 3 after MCAO/R and persisting until day 7. This was accompanied by considerable colocalization with OPCs (Figure 6E). Temporally and spatially consistent with myelin injury and OPC cell death, this trend suggests that increased levels of PLIN2 after MCAO/R might contribute to OPC ferroptosis and the ensuing myelin damage.
Figure 7 | Plin2 knockdown rescues demyelination and improves neurological deficits after MCAO/R.
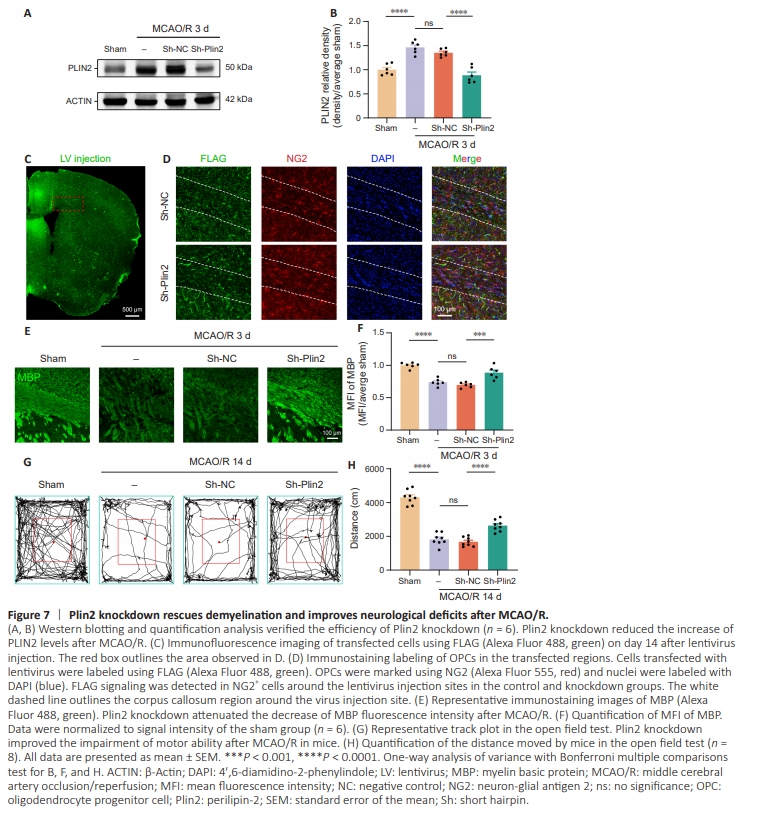
Based on the previous analysis, we sought to determine whether inhibition of Plin2 can rescue the myelin damage and improve neurological deficits. We therefore performed a stereotaxic injection of lentivirus to knockdown Plin2 in the corpus callosum of mice. Western blotting showed significantly higher PLIN2 levels in the corpus callosum on day 3 after MCAO/R compared with the sham group, a finding consistent with immunofluorescence results. Lentivirusmediated knockdown of Plin2 significantly decreased PLIN2levels on day 3 post-ischemia (Figure 7A and B). Additionally, FLAG immunofluorescence staining was used to examine lentiviral transfection efficiency. This showed the presence of FLAG signal in OPCs surrounding the injection site (Figure 7C and D). We performed immunofluorescence staining of brain slices using MBP to determine whether Plin2 knockdown can improve myelin integrity. The results showed that Plin2 knockdown significantly increased MBP levels after MCAO/R (Figure 7E and F). This was further supported by the open field test, which showed that all mice in the MCAO/R and lentiviral Sh-NC MCAO/R groups traveled a shorter distance compared with the sham group on day 14 post-ischemia. Plin2 knockdown significantly improved these deficits in motor behavior at the corresponding time points (Figure 7G and H).