神经损伤与修复
-
Figure 2 | Association of the CNKSR2 interactome with the centrosome.
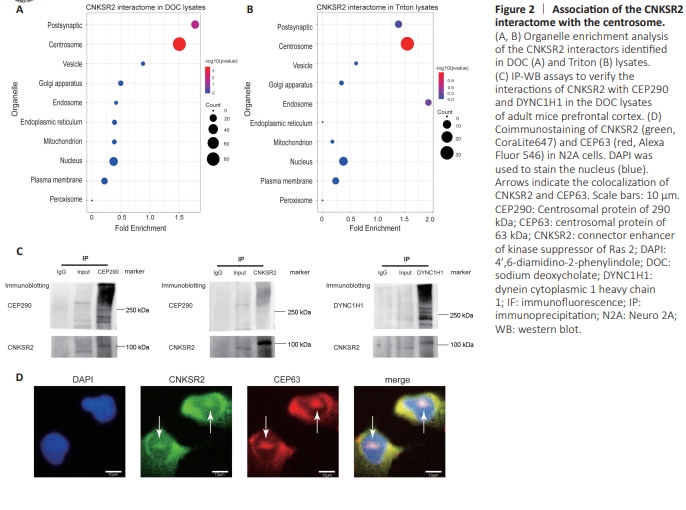
Organelle enrichment analysis was performed to better understand how CNKSR2 is localized within cells. The CNKSR2 interactors showed significant enrichment in the centrosome proteome in both the DOC and Triton groups. In the DOC group, the CNKSR2 interactors were also significantly enriched in the PSD proteome (Figure 2A and B). We used reciprocal IP-WB assays to verify the association between CNKSR2 and the microtubule and centrosome. We identified the microtubule protein dynein cytoplasmic 1 heavy chain 1 (DYNC1H1) and the centrosome marker CEP290 as CNKSR2 interactors in the DOC lysates. We precipitated the complexes of these two proteins and found CNKSR2 in both coprecipitated samples, confirming the interactions between CNKSR2 and each of these two proteins and further supporting the link of CNKSR2 with the microtubule and centrosome structures (Figure 2C). Using CEP63 as a centrosome marker, we performed coimmunostaining of CNKSR2 and CEP63. The confocal images showed colocalization of these two proteins, confirming the centrosomal localization of CNKSR2 (Figure 2D).
Figure 4 | CNKSR2 knockdown disturbs centrosome-related functions.
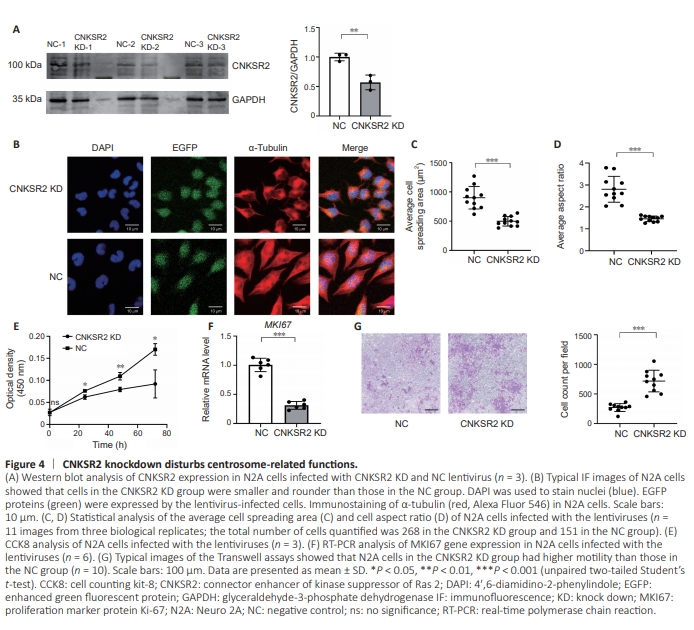
The centrosome plays crucial roles in a variety of cellular functions associated with the microtubule cytoskeleton, affecting cell size and shape, cell cycle, cell migration, and cilia formation (Nigg and Raff, 2009; Conduit et al., 2015; O’Neill et al., 2022). Thus, we down-regulated CNKSR2 expression in N2A cells (Figure 4A) and examined a range of cellular processes related to the microtubule cytoskeleton and centrosome. The centrosome regulates the distribution and arrangement of microtubules, which are composed of alternating α-tubulin and β-tubulin subunits (Murphy and Stearns, 1996; Conduit et al., 2015; Petry and Vale, 2015; Akhmanova and Kapitein, 2022) Through fluorescent immunoimaging of α-tubulin, we examined the morphology of N2A cells in the CNKSR2 KD and NC groups (Figure 4B). Statistical analysis showed that the cells in the KD group were smaller than those in the NC group, and had a more rounded morphology with a lower length/ width ratio of the cell body than that of the NC group (Figure 4C and D).Differentiation culture conditions promote neurite outgrowth in N2A cells (Mao et al., 2000; Zeng and Zhou, 2008) visible with α-tubulin immunoimaging. After being seeded and cultured in the differentiation medium for 48 hours, approximately 20%–60% of the cells in each group had neurite outgrowth. There were no statistically significant differences in the percentages of neurite-growing cells, the average neurite number per cell, and mean neurite length (Additional Figure 1), indicating that CNKSR2 did not have a notable impact on the formation of cell projections. The centrosome also plays a role in cell proliferation. Thus, we used CCK8 assay to examine the effect of CNKSR2 downregulation on cell proliferation. OD450 values of the CNKSR2 KD group were markedly lower than those of the NC group, suggesting that CNKSR2 plays a role in cell proliferation (Figure 4E). Consistent with this, real-time polymerase chain reaction showed that CNKSR2 knockdown led to the reduced gene expression of the proliferation marker MKI67 (Figure 4F). Centrosomes influence cell migration by orienting the microtubule cytoskeleton and regulating microtubule dynamics, providing directionality and stability during cell movement (Conduit et al., 2015; Petry and Vale, 2015). We performed Transwell experiments to assess the effect of CNKSR2 knockdown on cell motility. Down-regulating CNKSR2 expression enhanced the motility of N2A cells. Compared with that in the NC group, more cells passed through the pores to the other side of the membrane in the CNKSR2 KD group (Figure 4G), indicating that CNKSR2 was involved in regulating cell migration.