脊髓损伤
-
Figure 1 | Identification of cell types in the human spinal cord.

To achieve single-cell resolution, we conducted snRNA-seq on 64,021 nuclei isolated from lumbar enlargements of the spinal cord in nine adult humans. From these data, we identified eight major cell types (Figure 1A) with distinctive molecular markers. Astrocytes (5.2% of total nuclei) were identified by expression of the representative markers AQP4, ATP1A2, and SLC1A2, microglia (13.1% of total nuclei) by PTPRC, CTSS, and ITGAM, and oligodendrocytes (58.8% of total nuclei) by MBP, MOBP, MOG, and PLP1 (Figure 1A–C). We further explored the spatial locations of these three glial cell types by integrating the snRNA-seq data with the spatial transcriptomics data (Zhang et al., 2024b). The spot locations of the spatial transcriptomics data revealed that astrocytes were widely present in the spinal cord, but with higher expression in the white matter than in the gray matter (Figure 1D). This finding was validated by immunofluorescence staining (Figure 1E). Microglia and oligodendrocytes were mainly found in the white matter (Figure 1F–I). To determine the heterogeneity within each glial cell type in the human spinal cord, we classified astrocytes (Figure 2A–C and Additional Figure 1A), microglia (Figure 2D– F and Additional Figure 1B), and oligodendrocytes (Figure 2G–I and Additional Figure 1C) into six subclusters each, based on their transcriptional characteristics. In the mouse spinal cord, astrocytes (Figure 3A–C and Additional Figure 1D), microglia (Figure 3D–F and Additional Figure 1E), and oligodendrocytes (Figure 3G–I and Additional Figure 1F) were classified into five, four, and five subclusters, respectively, based on the results of a previous study (Sathyamurthy et al., 2018). We further analyzed the representative marker genes with uniquely high expression levels in each subcluster, ultimately identifying several marker genes associated with somatosensory system disorders. These included the following: CACNB2 (calcium voltage-gated channel auxiliary subunit beta 2) (Maselli et al., 2003) in Cluster 1 (C1) of human astrocytes; SCN7A (sodium voltage-gated channel alpha subunit 7) (Dolivo et al., 2021) in Cluster 3 (C3) of human astrocytes and Cluster 5 (C5) of mouse oligodendrocytes; GRID2 (glutamate ionotropic receptor delta type subunit 2) (Khan et al., 2023) in C1 of human microglia; Ctss (cathepsin S) (Clark et al., 2007) in Cluster 2 (C2) of mouse microglia; and Grm3 (glutamate receptor, metabotropic 3) (Sheahan et al., 2018) in C1 of mouse oligodendrocytes.
Figure 5 | Spatial distribution of various glial cell subtypes in the human spinal cord.
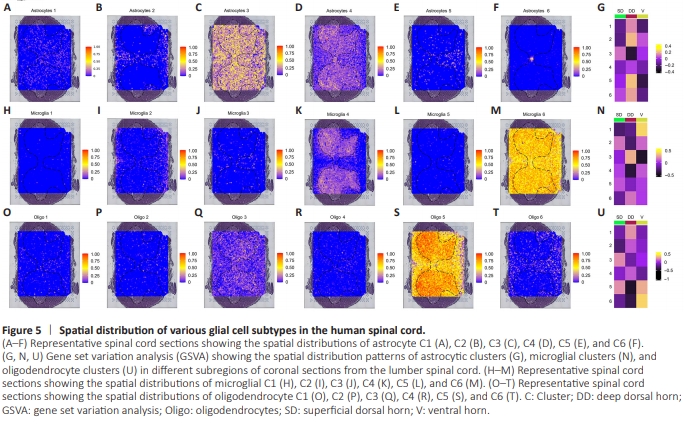
Next, we examined the spatial location of glial subtypes using the spatial transcriptomes, as described in our previous work (Zhang et al., 2024b). We selected spatial transcriptomes from one of the most typical lumbar spine enlargement sections and used the equivalent segment of each sample for analysis, ultimately identifying a total of five major cell subtypes. Using the SpaCET software (Ru et al., 2023), we characterized the distribution patterns of glial subtypes by integrating the snRNA-seq data of each with the corresponding spatial sections. The typical distributions of astrocyte Clusters 1–6 in the spinal cord are shown in Figure 5A–F. To analyze the spatial distribution of different glial subtypes in greater detail, we subsequently partitioned the human spinal gray matter into three regions: the superficial dorsal horn (Lamina I–II), deep dorsal horn (Lamina III–VI), and ventral horn (Lamina VII–IX) (Todd, 2010). Figure 5G shows the distribution characteristics of various astrocytic clusters in the three regions. Similarly, the typical distributions of different microglia and oligodendrocyte subtypes are shown in Figure 5H–N and Figure 5O–U, respectively. Microglia C2 and C5, and oligodendrocyte C3 and C5, were predominantly present in the superficial dorsal horn; astrocyte C2, C5, and C6, microglia C3 and C6, and oligodendrocyte C1, C2, and C4 were found in the deep dorsal horn; and microglial C1 and oligodendrocyte C6 were in the ventral horn.