脊髓损伤
-
Figure 1 | Microglia are essential for recovery from spinal cord injury, and their removal is detrimental to recovery from spinal cord injury.
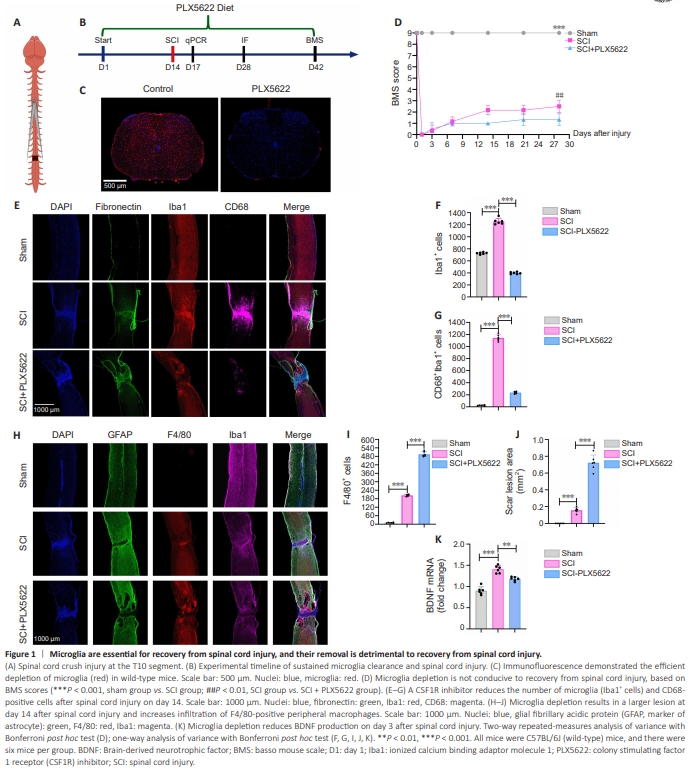
To investigate the effects and mechanisms of microglia depletion on SCI, we subjected mice to spinal cord crush injury (Figure 1A) and provided them with a PLX5622-containing diet starting 14 days before injury and continuing until the 28th day after injury (Figure 1B). To assess the depletion efficiency of PLX5622, we administered the PLX5622-containing diet to WT uninjured mice for 14 days and observed a marked reduction in the number of microglia in the spinal cord after 2 weeks of consuming the PLX5622-containing diet compared with the normal diet group (Figure 1C and Additional Figure 3A). For all subsequent experiments, we used the established mouse model of SCI in which microglia had been cleared via PLX5622 administration. We evaluated hindlimb motor function on day 1 before SCI, as well as on days 1, 3, 7, 14, 21, and 28 after injury, using the BMS scale. Our findings revealed that continuous removal of microglia was detrimental to the recovery of hindlimb motor function after SCI (Figure 1D). Previous studies have indicated that, in response to SCI, microglia are heavily activated and secrete large amounts of inflammatory factors that hinder recovery (Liu et al., 2020; Li et al., 2022b; Gong et al., 2023). Although microglia removal reduced the number of activated CD68+ microglia on day 14 after SCI (Figure 1E–G), unexpectedly, the average lesion size in the microglia removal (SCI + PLX5622) group was larger than that in the control group on day 14 after SCI. Additionally, the removal of microglia affected astrocyte proliferation (Figure 1H and Additional Figure 4), the formation of glial cell scars was reduced, lesion area increased, and infiltration of F4/80-positive peripheral macrophages increased (Figure 1H– J). RT-qPCR analysis demonstrated downregulation of BDNF mRNA levels after SCI following the removal of microglia (Figure 1K). This finding was particularly unexpected because, in the CNS, the main sources of BDNF are neurons and astrocytes, suggesting that the removal of microglia may reduce the expression of microglia-derived BDNF after SCI or affect the secretion of BDNF from other cellular sources. This indicates that microglia play a crucial role in lesion improvement and motor function recovery after SCI by modulating astrocyte proliferation and BDNF secretion.
Figure 2 | Cre-dependent manipulation of BDNF expression by microglia

Previous studies have demonstrated the crucial role of microglia-derived BDNF in the regulation of neurogenesis and neuronal synaptic plasticity (Parkhurst et al., 2013; Zhang et al., 2021; Wang et al., 2023b). Our results also indicate that the removal of microglia is detrimental to recovery from SCI and affects BDNF production after SCI, suggesting the potentially crucial role of microglia-derived BDNF. Therefore, we generated mice whose microglia conditionally overexpress BDNF (Figure 2A and B). The labelling rate of tdTomato positive microglia in CX3:BDNF mice reached nearly 98% after tamoxifen administration via gavage (Figure 2C and D). Initially, we examined BDNF expression after activation in CX3:BDNF mice and found that the BDNF mRNA content in the spinal cord of mice on day 14 after completion of tamoxifen gavage was higher than that in mice that received tamoxifen gavage after 7 days and also higher than that in the WT group (Activation here refers to the conditional overexpression of the BDNF gene in microglia of CX3:BDNF mice achieved by administering tamoxifen. The metabolites of tamoxifen bind to estrogen receptors (ER), leading to the release of Cre into the nucleus and subsequent recombination at LoxP sites) (P < 0.001; Figure 2H and I). Subsequently, we assessed BDNF mRNA expression in the spinal cord of mice on the third day after SCI and observed that the CX3:BDNF group exhibited higher levels compared with the WT group and the WT-SCI group (P < 0.001; Figure 2G). Furthermore, we detected BDNF expression in the spinal cord adjacent to the injury site through immunofluorescence on the 28th day after SCI and found that the CX3:BDNF group maintained stable overexpression of BDNF on the 28th day after SCI (P < 0.001; Figure 2Eand F). However, we found that, although BDNF protein and mRNA levels in CX3:BDNF in transgenic mice were markedly increased, colocalization of BDNF and ionized calcium binding adaptor molecule 1 (Iba1) was relatively low. This is most likely due to the fact that BDNF is a secreted protein, and thus BDNF overexpressed by microglia is secreted outside of the cells. Moreover, we found that colocalization of BDNF and Iba1 was markedly increased in the CX3:BDNF group compared with WT group, and there was almost no colocalization of BDNF and Iba1 in the WT group.
Figure 4 | BDNF overexpression reduces neuronal apoptosis and axonal necrosis.

To elucidate the mechanism by which microglial BDNF overexpression promotes the recovery of motor function after SCI, we initially assessed axon and neuron preservation, as well as the expression of pro-apoptotic factors, in mice following SCI using immunofluorescence and quantitative reverse transcription-polymerase chain reaction (Figure 4A–H). 5-Hydroxytryptamine (5-HT, serotonin)-positive axons are an important axon type in the spinal cord. The descending 5-HT positive axons in the spinal cord regulate muscle tone, movement and rhythm, as well as movement coordination through a central pattern generator. SCI interrupts the projections of 5-HT positive axons where the central pattern generator is located, resulting in severe motor impairment. To investigate the effect of BDNF overexpression on axons, we counted the number of 5-HT-positive axons on day 28 after SCI. BDNF overexpression by mouse microglia preserved 5-HT-positive axons on day 28 post-SCI (Figure 4A) and reduced the number of apoptotic neurons on day 1 post-SCI (Figure 4B, C, F, G, Additional Figure 3B). Furthermore, BDNF expression by microglia promoted production of the anti-apoptotic factor Bcl2 (Figure 4H) and inhibited production of the pro-apoptotic factors Bax and Caspase-3 (Figure 4D and E). These results indicate that BDNF overexpression by microglia inhibits apoptosis.
Figure 5 | BDNF overexpression promotes activation of microglia to the Arg1-positive phenotype and reduces inflammation after spinal cord injury.

Numerous previous studies have indicated that M2-type microglia promote anti-inflammatory factor secretion and BDNF expression (Van den Bossche et al., 2016; Ge et al., 2021; Zhang et al., 2021). The classification of microglia into M1 and M2 subtypes is not sufficiently comprehensive (Paolicelli et al., 2022), but M1/M2 classification reveals that different types of microglia may play distinct roles in the pathology of SCI (Stratoulias et al., 2023). To investigate the impact of BDNF overexpression on microglial phenotype and the secretion of inflammatory factors following SCI, we used two classical microglial markers: the anti-inflammatory marker Arg1 and the pro-inflammatory marker IL-1β. Our findings demonstrated that BDNF overexpression by microglia promoted increased the proportion of Arg1- positive microglia and decreased the proportion of IL-1β-positive microglia after SCI (Figure 5A, B, E, and F). Additionally, BDNF overexpression by microglia attenuated the secretion of inflammatory factors and enhanced the production of anti-inflammatory factors on day 3 after SCI (Figure 5C, D, G, and H). Furthermore, BDNF overexpression by microglia stimulated microglial activation up to day 28 post-SCI (Additional Figures 3D and 5). These results suggest that BDNF overexpression by microglia may promote SCI repair by regulating microglial phenotype. In addition, we found that, under normal physiological conditions, BDNF overexpression did not affect the number and activation state of microglia, and that microglia overexpressing BDNF expressed the homeostatic marker P2RY12 and maintained a ramified morphology (Figure 2C). However, in the early stage after SCI, BNDF overexpression by microglia reduced the total number of activated microglia (Figure 5, 3 days post-SCI). In the late stage of SCI, BDNF overexpression increased the number of activated amoeboid microglia compared with the SCI group (Figure 2E and Additional Figure 5, 28 days post-SCI). Moreover, BDNF overexpression by microglia after SCI promotes microglial transition to an activated amoeboid Arg1-positive type. Our results indicate that BDNF overexpression by microglia reduces neuroinflammation and induces microglial phenotype transition.
Figure 6 | BDNF overexpression by microglia promotes vascular regeneration.
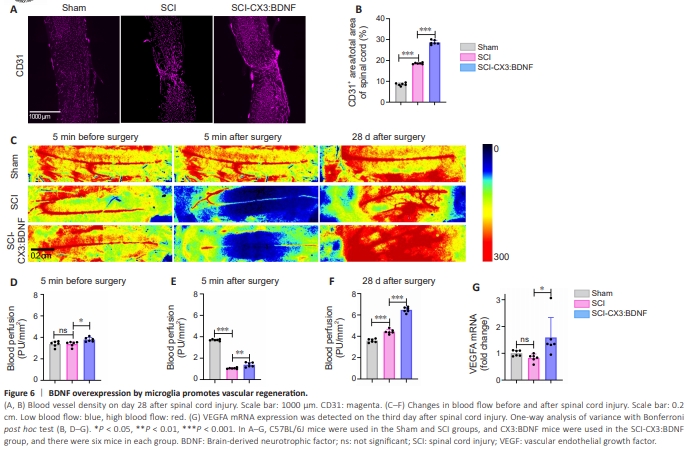
SCI often disrupts the blood–spinal cord barrier, resulting in extensive hemorrhage, vascular rupture, and hematoma in the affected area, leading to destruction of the neurovascular network. Vascular cells play a crucial role in promoting the proliferation of neural precursor cells, and dividing cells are closely associated with blood vessels in the SGZ region of adult mice. Therefore, promoting reconstruction of the neurovascular network following SCI may be an effective strategy for treating this condition (Leventhal et al., 1999; Kojima et al., 2010; Tsai et al., 2016). Previous in vitro experiments have demonstrated that BDNF can promote blood vessel formation in vascular endothelial cells in vitro (Lin et al., 2014), and in this study, we assessed vascularization on day 28 after SCI through immunofluorescence staining. In the sham-operated group, opening the vertebral plates of mice did not have a large effect on blood flow to the mouse spinal cord, whereas a clamp injury resulted in a rapid disruption of blood flow to the mouse spinal cord (Figure 6C–E). Our findings revealed that BDNF overexpression by microglia effectively promoted blood vessel formation and increased blood flow compared with the SCI group following SCI (Figure 6A–F). Vessel density and blood flow were also increased, possibly due to the hypoxic environment that develops after SCI, which promotes hypoxiainducible factor 1 secretion, subsequently stimulating blood vessel formation.It has also been demonstrated in the previous study that the removal of microglia affects vascular regeneration after SCI (Li et al., 2020). Furthermore, we analyzed the association of BDNF with angiogenesis-related factors using Genemania and found strong associations and interactions between BDNF, VEGFA, Angpt1 (Additional Figure 3E). Subsequently, we examined VEGFA expression on the third day after SCI using qPCR and observed that BDNF overexpression by microglia promoted VEGFA expression following SCI (Figure 6G). These findings suggest that BDNF overexpression by microglia may promote angiogenesis and increase blood flow after SCI by enhancing VEGFA expression.
Figure 7 | BDNF overexpression primarily by microglia but not by macrophages promotes functional recovery after spinal cord injury.

Both microglia and macrophages are labeled in the transgenic mice used in this experiment (CX3CR1 creER). It is worth noting that macrophages, which overexpress BDNF in the periphery following SCI, infiltrate toward the injury site (Bruttger et al., 2015). This raises the question of whether it is microglia or macrophages overexpressing BDNF that contribute to recovery after SCI. To address this, we employed the transgenic mouse line TMEM119 creER, in which microglia are labeled, as TMEM119 is expressed exclusively in microglia and not in macrophages (Bennett et al., 2016; Masuda et al., 2020). By crossing TMEM119 creER+/+ mice with LSL-BDNF-tdTomato+/+ mice, we obtained transgenic mice that specifically overexpress BDNF only in microglia (Figure 7A). Initially, we assessed the labeling efficiency after tamoxifen gavage in the resulting transgenic mice and observed that the labeling efficiency in TMEM119 creER–/+:LSL-BDNF–/+- tdtomato mice was considerably lower than that in CX3CR1 creER mice (Figure 7C, F and Additional Figure 3C). Despite the low labelling efficiency of TMEM119 transgenic mice, overexpression of BDNF specifically in this fraction of labelled microglia was still able to facilitated the restoration of motor function in mice subjected to SCI (Figure 7I). To exclude the potential impact of peripheral macrophages, which also overexpress BDNF and infiltrate into the peripheral injury zone after SCI on SCI recovery. We administered dietary PLX73086 to the CX3:BDNF mice from 19 days prior to SCI until the 28th day after injury (Figure 7B). PLX73086 is a CSF1R inhibitor that have a low blood–brain barrier penetration. Our findings revealed that PLX73086 cleared macrophages from the peripheral liver, but did not clear microglia from the spinal cord (Figure 7D, E, G, and H), and that motor function was restored in CX3:BDNF mice treated with PLX73086 (Figure 7J). In summary, our findings suggest that it is primarily microglia overexpressing BDNF that play a therapeutic role in SCI.