视神经损伤
-
Figure 1 | LIRD induced an increase in photoreceptor death in mice.
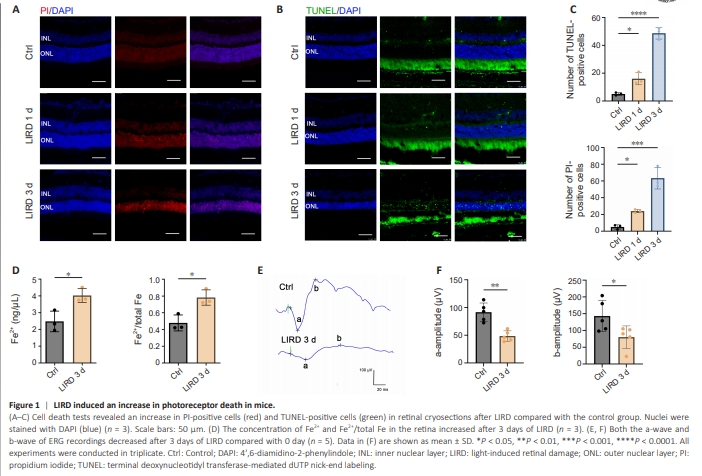
To evaluate photoreceptor cell death in the LIRD mouse model, PI and TUNEL staining was performed on retinal sections. Compared with the control group, the LIRD group had a higher number of PI-positive (P = 0.0002) and TUNELpositive (P < 0.0001) cells in the retina in a time-dependent manner (Figure 1A–C). Furthermore, the numbers of nuclei rows in the ONL were significantly reduced compared with those in controls, as a result of photoreceptor mortality and loss during LIRD (P < 0.0001; Additional Figure 2). Excess Fe2+, which is highly cytotoxic through the Fenton reaction, facilitates ferroptotic cell death (Zhao et al., 2021b). The Fe2+ concentration (P = 0.0166) and proportion (P = 0.0226) in the retina significantly increased after 3 days of light exposure (Figure 1D), suggesting the involvement of Fe2+ in light-induced photoreceptor cell death. ERG was performed to further assess the changes in retinal function after LIRD. The results showed a significant decrease of a-wave (P = 0.0010) and b-wave (P = 0.0398) amplitudes 3 days after LIRD (Figure 1E and F), which indicated light damage to the retina. The pathological changes in retinal structure matched the functional changes suggested by ERG, demonstrating retinal damage in the LIRD model.
Figure 2 | The expression of SLC7A11 increases in the retina of LIRD mice.
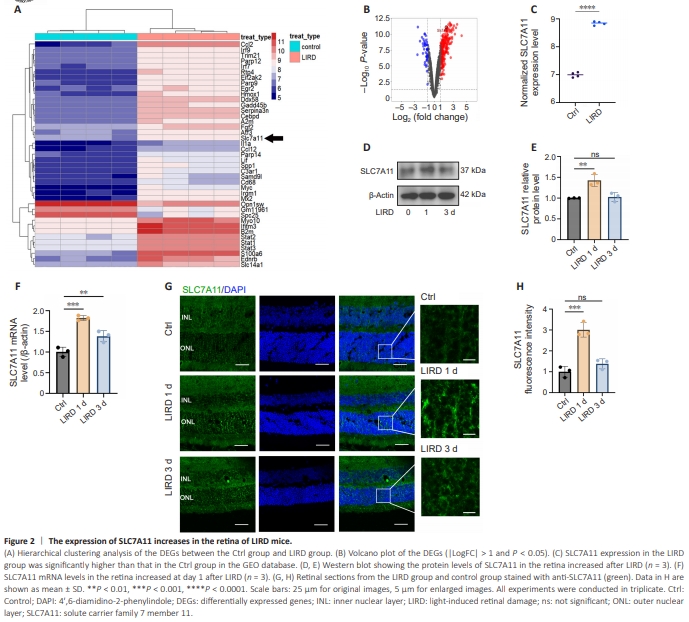
To explore the mechanism of photoreceptor death, we performed differential expression analysis using the GSE37773 dataset from the GEO database revealed that the albino mice in LIRD group displayed upregulation of 232 genes and downregulation of 45 genes compared with the control group (log fold-change (|LogFC|) > 2 or < 0.5 and P? <? 0.05). A heatmap of gene expression patterns after light damage is shown in Figure 2A. The results revealed upregulation of SLC7A11 and iron metabolic–related proteins such as heme oxygenase (decycling) 1 (Homx1), lipocalin 2 (Lcn2), and transferrin (Trf) in the LIRD group. SLC7A11 showed upregulation with an FC of 3.60 (Figure 2B). SLC7A11 expression was also higher in the LIRD group in the database (P? <? 0.0001; Figure 2C).To examine the expression levels of SLC7A11 in the LIRD model, we collected retinas from the LIRD group at different time points and performed western blot analysis. SLC7A11 levels increased significantly on day 1 and then decreased on day 3 (P = 0.0038; Figure 2D and E). RT-PCR analysis also showed upregulated SLC7A11 mRNA levels on day 1, followed by a decline (P = 0.0003; Figure 2F). Immunofluorescence showed increased SLC7A11 expression in the ONL of the retina and SLC7A11 localization on the cones and rods on day 1 after LIRD (Figure 2G and H and Additional Figure 3).
Figure 3 | Ferroptosis and oxidative stress is induced in response to light damage in vivo.
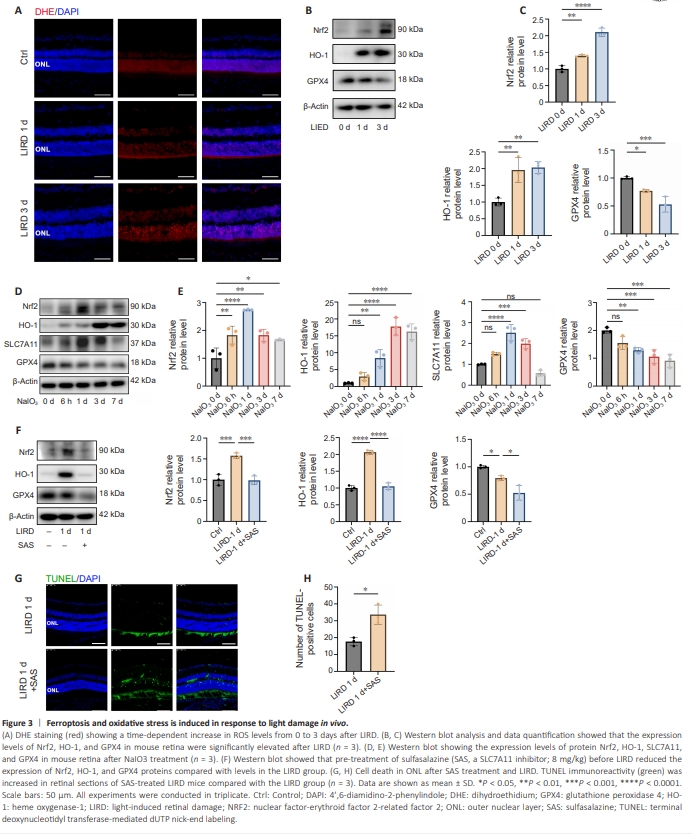
ROS accumulation in response to adverse external stimuli exceeds the scavenging capacity of the peroxidase system, leading to oxidative stress, triggering lipid peroxidation and, eventually, ferroptosis. We next used DHE staining to measure the ROS levels in mouse retinas with or without light exposure. The ROS levels increased on days 1 and 3 after light exposure, consistent with the change in nuclei rows in the ONL (Figure 3A). We measured the levels of several molecules involved in oxidative stress, including nuclear erythroid factor 2-related factor 2 (Nrf2) (P < 0.0001) and heme oxygenase-1 (HO-1) (P = 0.0037) and the levels of these molecules increased after LIRD, peaking on day 3 (P < 0.01; Figure 3B and C), suggesting the involvement of oxidative stress in light-induced photoreceptor damage. As a negative regulator of ferroptosis, glutathione peroxidase 4 (GPX4) plays a role in reducing lipid hydroperoxide levels. We also observed downregulation of GPX4 after light exposure in a time-dependent manner (P = 0.0012; Figure 3B and C). Intraperitoneal injection of NaIO3 is widely used to establish a model of oxidative stress damage (Chan et al., 2019). We next investigated the association of oxidative stress with ferroptosis using this model. Nrf2 and SLC7A11 expression increased 6 hours after NaIO3 treatment, plateaued on day 1 (P < 0.0001), and then decreased, whereas HO-1 expression plateaued on day 3 and then declined (P < 0.0001; Figure 3D and E). GPX4 expression decreased continuously from days 6 to 7 after treatment (P = 0.0003). To further investigate the role of SLC7A11 in photoreceptor degeneration, SAS, a pharmacological inhibitor of SLC7A11, was injected intraperitoneally into mice for 3 consecutive days before light exposure. SAS treatment resulted in downregulated expressions of Nrf1, GPX4, and HO-1 (Figure 3F) and increased numbers of TUNEL-positive cells in the photoreceptors (Figure 3G and H).
Figure 4 | SLC7A11 expression was upregulated in 661W cells with H2O2-treatment and the SLC7A11 inhibitor SAS promoted 661W ferroptosis.
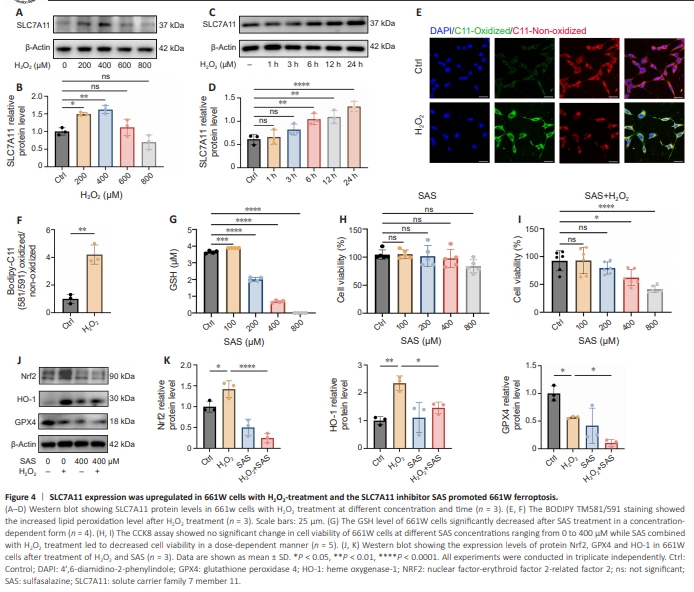
To explore the mechanism of SLC7A11 in photoreceptors, we used 661W cells. After treatment with H2O2, BODIPY-C11 581/591 staining showed that lipid peroxidation in 661W cells significantly increased, which was observed as an increase in the ratio of the immunofluorescence intensity of the oxidized/ non-oxidized state (P = 0.0019; Figure 4A and B). To examine the activation of ferroptosis and the increase of lipid peroxidation level, malondialdehyde (MDA) levels were measured in 661W cells with and without H2O2 (400 μM) stimulation. The level of MDA in cells of H2O2 group was significantly increased compared with the control group (P < 0.01; Additional Figure 4). Subsequently, to investigate the relationship between oxidative stress and SLC7A11 in vitro, we treated 661W cells with H2O2 at different concentrations and times and analyzed SLC7A11 expression by western blot. We observed significantly up-regulated SLC7A11 expression after H2O2 treatment (P = 0.0002; Figure 4C–F). The expression of SLC7A11 reached the maximum at the H2O2 concentration of 400 μM. Figure 4E shows results after different time points of H2O2 stimulation, that is, SLC7A11 protein expression increased in a time-dependent manner. We next explored the potential role of SLC7A11 in photoreceptor degeneration by treating 661W cells with SAS. A previous study showed that SLC7A11 reduces lipid peroxidation in cells to inhibit ferroptosis by transporting cysteine into the cytosol to promote GSH production (Fang et al., 2020). We observed a decrease in GSH level after treatment with 200 μM SAS and GSH level was nearly undetectable following treatment with 400 μM SAS (P < 0.0001; Figure 4G). To examine the mechanism of GSH reduction, we examined the levels of enzymes related to GSH synthesis in661W cells treated with SAS (Additional Figure 5A and B). Both glutathione reductase (GR) and glutathione synthase (GS) protein levels showed a SAS concentration–dependent decreasing trend, which was consistent with the change trend of GSH levels after SAS treatment. CCK-8 assay showed that 0–400 μM SAS alone had no effects on 661W cell viability (Figure 4H), while SAS combined with H2O2 treatment led to decreased cell viability in a dosedependent manner (P < 0.0001; Figure 4I). We next examined ferroptosis-related molecules in the oxidative stress model induced by NaIO3. As a result, the levels of SLC7A11, NRF2 and HO-1 increased and the expression of GPX4 decreased, indicating the induction of ferroptosis during oxidative stress-induced photoreceptor death (Additional Figure 6). SAS treatment significantly inhibited the expression levels of GPX4 (P = 0.0017), Nrf2 (P = 0.0001), and HO-1 (P = 0.0038) protein (Figure 4J and K), which indicated that SLC7A11 inhibition enhanced lipid peroxidation and promoted ferroptosis in 661W cells.
Figure 7 | SLC7A11 overexpression in photoreceptors suppresses retinal degeneration of LIRD mice.
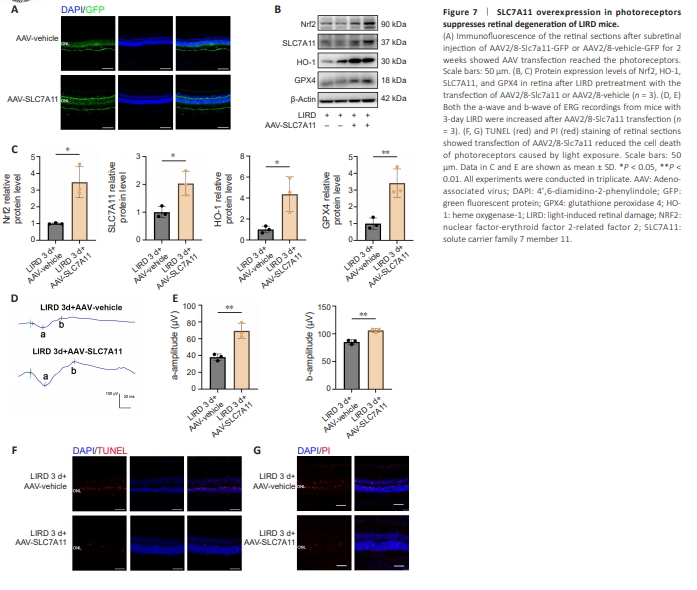
To explore the therapeutic potential of SLC7A11 overexpression, we injected a recombinant AAV–mediated Slc7a11 vector (rAAV2/8-Slc7a11-GFP) into LIRD mice to deliver an SLC7A11 expression cassette into the photoreceptors. Two weeks later, the mice underwent light exposure. Cryosections revealed efficient transfection of rAAV2/8-Slc7a11-GFP into the retinal pigment epithelium (RPE) and photoreceptors in the whole range of retina (Figure 7A and Additional Figure 8). We also confirmed the expression of exogenous SLC7A11 and other related proteins including Nrf2, GPX4, HO-1 in the retina by western blotting (Figure 7B and C). ERG assay before and 3 days after LIRD showed that SLC7A11 overexpression in photoreceptors increased the a-wave (P = 0.0013) and b-wave (P = 0.0039) amplitudes 3 days after LIRD (Figure 7D and E). We investigated the impact of rAAV2/8-Slc7a11 on photoreceptor cell death and found that SLC7A11 overexpression decreased the numbers of both TUNEL-positive cells (Figure 7F) and PI-positive cells (Figure 7G) at 3 days after LIRD.