脑损伤
-
Figure 2 | rtPA attenuates neurological behavior impairment and apoptosis after ICH.
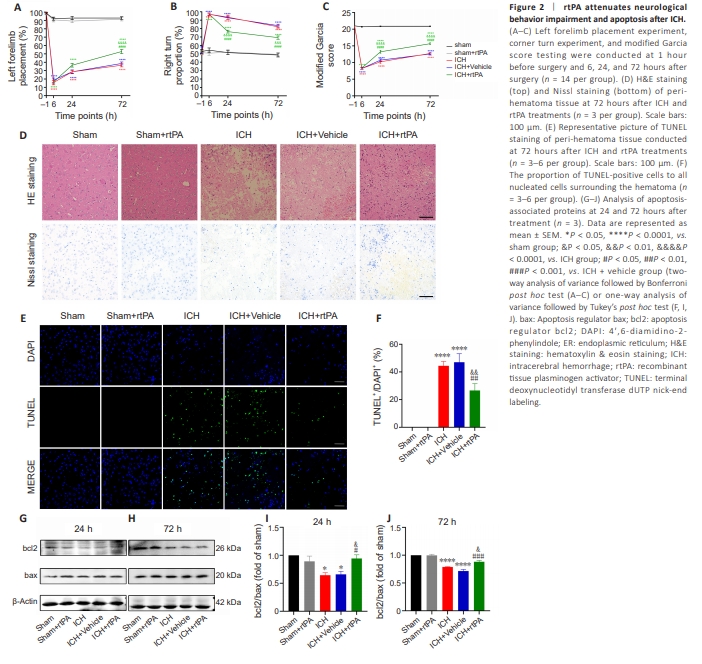
Recombinant tissue plasminogen activator attenuates neurological behavior impairment after intracerebral hemorrhage in mice To investigate the effects of rtPA on neurological behavioral impairment following ICH, we conducted behavioral testing on mice 1 hour prior to surgery and at 6, 24, and, 72 hours post-surgery. Mice in the ICH group exhibited neurological impairment compared with the sham group, and no statistical difference was observed between the ICH and the ICH + vehicle groups at any time point after ICH (Figure 2A–C). There was no significant difference in neurological function in the sham + rtPA group compared with the sham group, indicating that rtPA is likely safe in normal tissue. The ICH + rtPA group showed attenuated neurological impairment at 24 and 72 hours post-ICH compared with the ICH + vehicle group. These results suggest that stereotactic injection of therapeutic doses of rtPA does not induce neurological impairment in normal tissue and that rtPA ameliorates neurological behavioral impairment following ICH. Recombinant tissue plasminogen activator protects against the pathological damage induced by intracerebral hemorrhage in mice To investigate the effects of rtPA on the histopathological damage to perihematoma tissue following ICH, we evaluated the pathomorphological changes in the peri-hematoma tissue (basal ganglia site) using HE staining and Nissl staining at 72 hours post-ICH. The number of neurons in the peri-hematoma tissue was reduced in both the ICH and ICH + vehicle groups compared with the sham group, with a large number of atrophied and deeply stained neurons; no statistical difference was observed between the ICH and ICH + vehicle groups (Figure 2D). No abnormalities were observed in the basal ganglia region in the sham and sham + rtPA groups. Notably, the ICH + rtPA group showed reduced neuropathological damage compared with the ICH + vehicle group. These findings suggest that therapeutic doses of rtPA do not induce pathological damage and can attenuate pathological damage following ICH. Recombinant tissue plasminogen activator attenuates apoptosis caused by intracerebral hemorrhage in mice To assess the effects of rtPA on neuronal apoptosis following ICH, we conducted TUNEL staining at 72 hours post-ICH. A large number of apoptotic cells were observed in the peri-hematoma tissue of the ICH and ICH + vehicle group, with no difference between the groups (Figure 2E and F). No TUNELpositive cells were detected in the sham and sham + rtPA groups. rtPA treatment in ICH model mice resulted in a reduced number of TUNEL-positive cells. We further examined the protein levels of bcl2 and bax by western blot analysis at 24 and 72 hours post-ICH. The bcl2/bax ratio decreased in both the ICH and ICH + vehicle groups at 24 and 72 hours post-treatment compared with that in the sham group (Figure 2G–J). No significant differences were observed between the sham and sham + rtPA groups. The rtPA + ICH group showed a higher ratio of bcl2/bax compared with the ICH and ICH + vehicle groups. These findings suggest that rtPA treatment does not induce apoptosis in normal mouse brains and has the potential to mitigate apoptosis following ICH.
Figure 4 | rtPA attenuates neuron apoptosis and autophagy after experimental ICH in vitro.
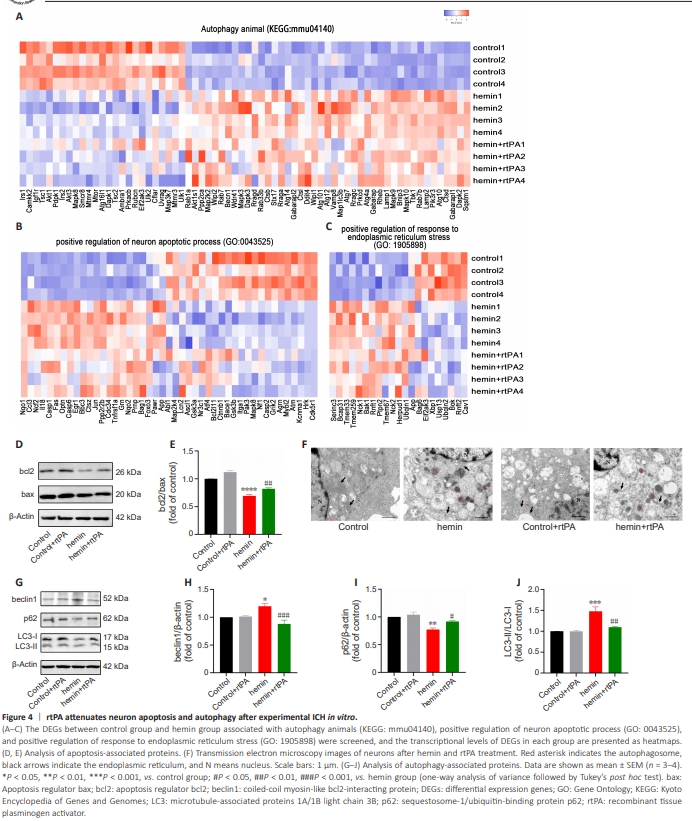
RNA sequencing was performed in cells treated with hemin or hemin with rtPA for 24 hours, and the results are presented in the heatmap of transcriptomics gene expression profile, principal component analysis, and Pearson correlation analysis in Additional Figure 2. We screened for DEGs between the control and hemin groups that were associated with autophagy in animals (KEGG: mmu04140), positive regulation of the neuron apoptotic process (GO: 0043525), and positive regulation of the response to ER stress (GO: 1905898). As shown in Figure 4A–C, rtPA treatment reversed the expression of numerous DEGs in these pathways. Furthermore, rtPA treatment upregulated the bcl2/bax ratio (Figure 4D and E), decreased the number of autophagosomes (Figure 4F), downregulated beclin1 expression and the LC3II/LC3I ratio, and upregulated p62 expression compared with hemin group (Figure 4G–J). These findings indicate the anti-apoptotic and anti-autophagy effects of rtPA in the ICH in vitro model. Recombinant tissue plasminogen activator inhibits endoplasmic reticulum stress in the in vitro intracerebral hemorrhage cell model To observe the morphology and continuity of the ER in primary neurons, we examined TEM images of cells after 24 hours of hemin treatment. In the control and control + rtPA groups, the ER exhibited a folded morphology and was normal in appearance; it was closely arranged and structurally continuous (Figure 4F).
Figure 5 | rtPA ameliorates endoplasmic reticulum stress in the in vitro ICH cell model.
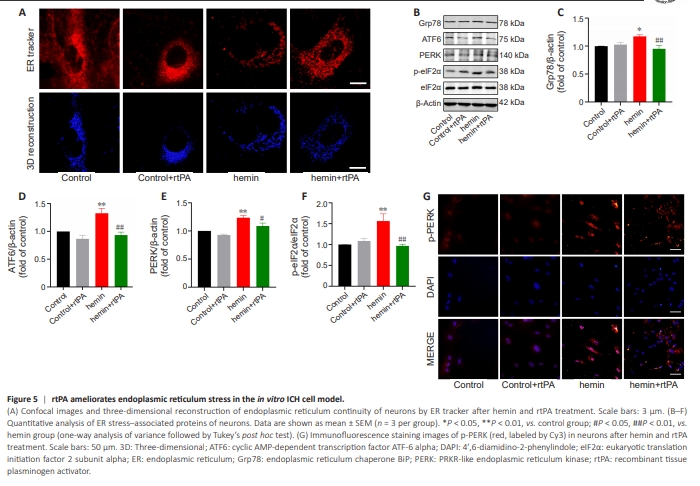
At 24 hours after hemin treatment, the ER lost itsnormal morphology, displaying swelling, severe fractures, and fragmentation. Notably, rtPA treatment alleviated the ER damage induced by hemin. We next used an ER tracker to label the ER at 24 hours after hemin treatment and reconstructed the three-dimensional morphology of the ER using confocal microscopy. The ER remained continuous in the control and control + rtPA groups (Figure 5A). In contrast, the ER was severely disrupted with poor continuity in hemin-treated neurons. Notably, this discontinuity was improved upon rtPA treatment. We further examined the protein levels of Grp78, ATF6, PERK, p-eIF2α, and eIF2α, which are indicators of ER stress, by western blot analysis. The results indicated no significant differences in the protein levels of these markers between the control and control + rtPA groups. While all proteins were elevated in the hemin group, rtPA treatment reduced the expression levels of these indicators (Figure 5B–F). To further clarify the effects of rtPA on ER stress following hemin treatment, we performed immunofluorescence staining to detect p-PERK expression in primary neurons at 24 hours after hemin treatment, as PERK is central regulator of ER stress and p-PERK is its activated form. p-PERK expression was minimal in the control and control + rtPA groups (Figure 5G). Hemin treatment resulted in a remarkable increase in p-PERK fluorescence intensity, and this increase was reduced upon rtPA treatment. These findings indicate that rtPA does not affect ER stress in control neurons but does alleviate hemin-induced ER stress in the in vitro ICH cell model.
Figure 7 | The PI3K inhibitor LY294002 reverses the anti-ER stress effect of rtPA and the EGF domain of rtPA may mediate the PI3K/AKT pathway in the ICH in vitro cell model.

To determine whether the PI3K/AKT/mTOR pathway is involved in the neuroprotective effects of rtPA in the in vitro model, we used the PI3K pathway inhibitor LY294002. LY294002 effectively reversed the upregulation of PI3K p85, p-AKT/AKT, and p-mTOR/mTOR levels induced by rtPA in hemintreated neurons (Figure 6G–J). We further found that LY294002 treatment reversed the regulatory effects of rtPA on apoptosis-related proteins (bcl2 and bax) (Figure 6K–L), autophagy-related proteins (beclin1, p62, and LC3-II/ LC3-I), and ER stress–related proteins (ATF6, p-eIF2α, and eIF2α) (Figure 7A– C), indicating LY294002 reversed the anti-apoptosis, anti-autophagy, and antiER stress effects of rtPA on primary neurons induced by hemin treatment. Additionally, ER tracker and 3D reconstruction of the ER demonstrated that LY294002 attenuated the ability of rtPA to improve ER continuity after hemin treatment (Figure 7D). Furthermore, LY294002 treatment reversed the downregulation of p-PERK fluorescence intensity induced by rtPA (Figure 7E). Collectively, these results suggest that rtPA may exert protective effects against apoptosis, autophagy, and ER stress in the in vitro ICH cell model through the PI3K/AKT/mTOR pathway. The epidermal growth factor domain of rtPA may upregulate the PI3K/AKT pathway in the in vitro ICH cell model To investigate the specific domain of rtPA responsible for exerting its neuroprotective effects, we used various inhibitors to target distinct domains of rtPA: GGACK for the protease domain, AG-1478 for the EGF domain, RAP for the finger domain, and TXA for the Kringle domain. Treatment with AG1478 resulted in a downregulation of p-AKT expression levels and a marginal decrease in PI3K p85 expression levels, although this decrease was not statistically significant (Figure 7F–H). Furthermore, AG-1478 abrogated the neuroprotective effects of rtPA, as evident by the presence of significant neural damage in neurons treated with hemin + rtPA + AG-1478 observed under microscopy (Figure 7I). Our study suggests that AG-1478 might interfere with the neuroprotective benefits and regulatory functions of the EGF domain of rtPA on the PI3K/AKT pathway, given that it did not independently affect the p-AKT/AKT ratio in primary neurons, as previously reported by Zhong et al. (2015).