视神经损伤
-
Figure 2 | VDAC1 oligomerization in R28 retinal cells subjected to OGD/R.
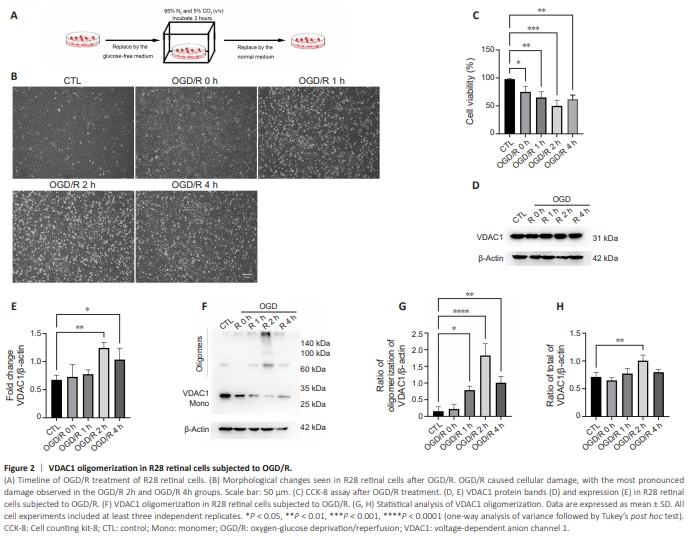
To explore the potential association between PANoptosis and VDAC1 oligomerization, we first investigate the status of VDAC1 oligomerization in R28 retinal cells subjected to OGD/R (Figure 2A). In contrast to the control group, cell injury was detected in the OGD/R group (Figure 2B). The CCK8 assay results also showed that cell viability was decreased in the OGD/R group compared with the control group, especially at 2 hours (Figure 2C). Next, we analyzed VDAC1 expression and oligomerization using ethylene glycol bis (succinimidyl succinate), a chemical cross-linker. VADC1 expression was increased 2–4 hours following OGD/R, especially at 2 hours compared with control group (Figure 2D and E). In addition, an increase in VDAC1 oligomerization was observed at 2 hours in the OGD/R group compared with control group (Figure 2F–H).
Figure 3 | PANoptosis and mitochondrial dysfunction in R28 retinal cells after OGD/R.
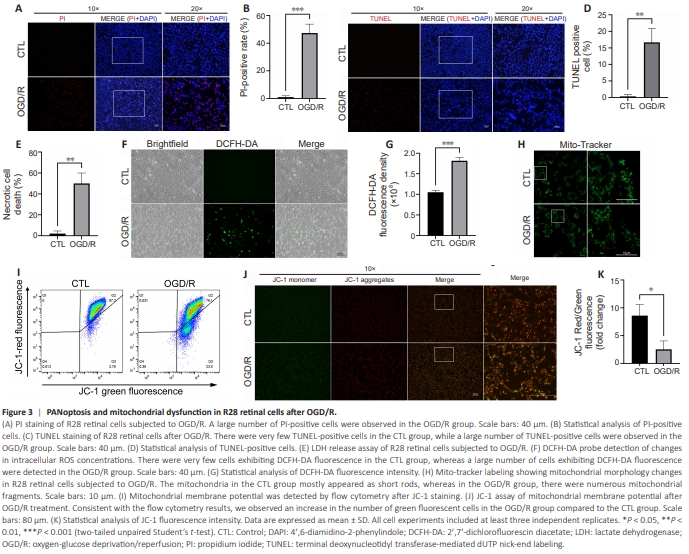
To investigate the potential association between VDAC1 oligomerization and PANoptosis, we characterized the types of RCD induced by OGD/R in vitro. As shown by PI staining, OGD/R induced necrotic cell death in R28 retinal cells (Figure 3A and B). Furthermore, TUNEL staining showed that OGD/R led to apoptotic cell death (Figure 3C and D). Finally, LDH-release assay verified that OGD/R induced necrotic cell death (Figure 3E). Pyroptosis and necroptosis are two major types of programmed necrotic cell death (An et al., 2024). Our results indicate that OGD/R induced PANoptosis in R28 retinal cells. As described in the following section, western blot and co-immunoprecipitation also showed that the three types of cell death signaling cascades were activated and the PANoptosome complex was assembled in response to OGD/R (Figure 4H–K). Next we detected indicators of mitochondrial dysfunction. Staining with the Mito-Tracker Green fluorescent probe showed more divided mitochondria in R28 retinal cells subjected to OGD/R, indicating an increase in mitochondrial fission (as indicated by fragmented mitochondria) compared with control group (Figure 3H). OGD/R also raised the ROS levels in R28 retinal cells compared with control group (Figure 3F and G). We then used a JC-1 kit to assess mitochondrial membrane potential. Monomeric JC-1 (emitting green fluorescence) was increased, and JC-1 aggregates (emitting red fluorescence) were reduced in R28 retinal cells subjected to OGD/R in comparison to the control group, indicating decreased mitochondrial membrane potential (Figure 3I–K). Collectively, these findings demonstrate that OGD/R induced PANoptosis and mitochondrial dysfunction in R28 retinal cells.
Figure 4 | Effects of VBIT-12 on PANoptosis in R28 retinal cells subjected to OGD/R.
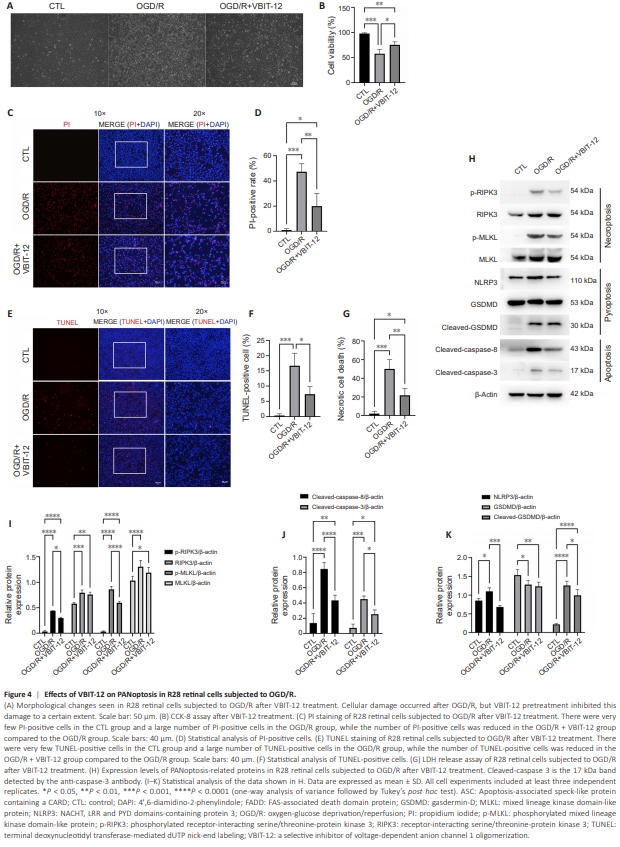
VBIT-12, a potent VDAC1 oligomerization inhibitor, was used to investigate the role of VDAC1 oligomerization in an in vitro model of retinal I/R injury (Verma et al., 2022). As shown in Figure 4A, VBIT-12 treatment suppressed OGD/R-induced injury to R28 retinal cells. CCK-8 assay verified that inhibiting VDAC1 oligomerization protected R28 retinal cells from OGD/R-induced injury (Figure 4B). Next we assessed the effect of VBIT-12 treatment on RCD using PI and TUNEL staining. A decreased proportion of PI-positive cells was observed in the VBIT-12-treated groups compared with the OGD/R group (Figure 4C and D). In addition, VBIT-12 treatment suppressed apoptosis in the VBIT-12-treated groups, as indicated by TUNEL staining (Figure 4E and F). Furthermore, LDH-release assay verified that VBIT-12 protected against OGD/ R-induced necrotic cell death (Figure 4G). Next we assessed activation of the three types of cell death signaling cascades by detecting expression changes in their key regulators. Increased RIPK3 and MLKL expression and phosphorylation (key events for the activation and execution of necroptosis (Zhan et al., 2021)) were detected in R28 retinal cells subjected to OGD/R compared with control group, while VBIT12 treatment reduced RIPK3 and MLKL phosphorylation (Figure 4H and I). OGD/R also elevated NLRP3 expression and GSDMD cleavage (key events for the activation and execution of pyroptosis (Shi et al., 2015)) compared with control group, while VBIT-12 treatment attenuated these trends (Figure 4H and J). Furthermore, VBIT-12 treatment reduced the increase in cleaved-caspase-8 and cleaved-caspase-3 (key events for the activation and execution of apoptosis (Yan et al., 2023)) induced by OGD/R compared with control group (Figure 4H and K). Collectively, these results demonstrated an important role for VDAC1 oligomerization in regulating PANoptosis in R28 retinal cells subjected to OGD/R.
Figure 5 | Effects of VBIT-12 on VDAC1 oligomerization and mitochondrial dysfunction in R28 retinal cells subjected to OGD/R.
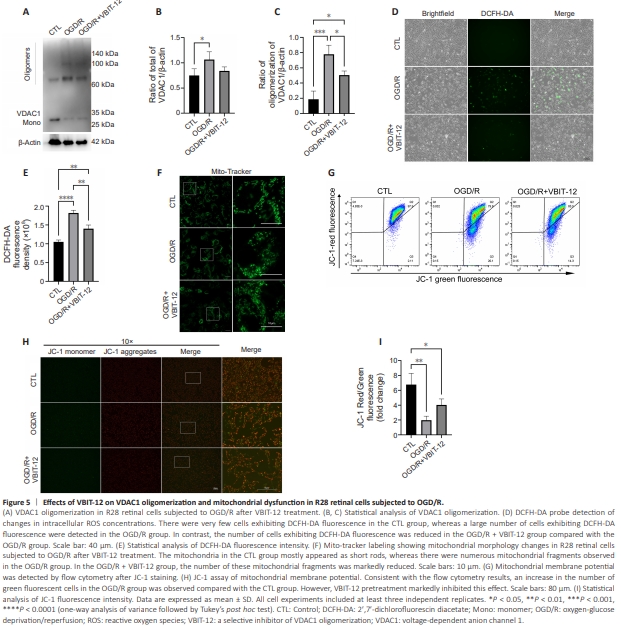
The role of VDAC1 oligomerization in mitochondrial dysfunction was evaluated using VBIT-12 treatment. Firstly, western blotting verified that VBIT12 treatment inhibited OGD/R-induced VDAC1 oligomerization (Figure 5A– C). The increased ROS levels observed in the OGD/R groups compared with control group was also reduced by VBIT-12 treatment, suggesting a critical regulatory role of VDAC1 oligomerization in oxidative stress following OGD/ R (Figure 5D and E). Using MitoTracker Green staining, we found that VBIT-12 treatment reduced mitochondrial fission following OGD/R (Figure 5F). Next, we conducted JC-1 staining to evaluate changes in mitochondrial membrane potential and found that VBIT-12 treatment attenuated the reduction in mitochondrial membrane potential induced by OGD/R (Figure 5G–I). These results demonstrate that VDAC1 oligomerization plays a critical role in regulating mitochondrial dysfunction and oxidative stress following OGD/R.
Figure 6 | Effects of scavenging mitochondria-derived ROS on mitochondrial dysfunction in R28 retinal cells subjected to OGD/R.
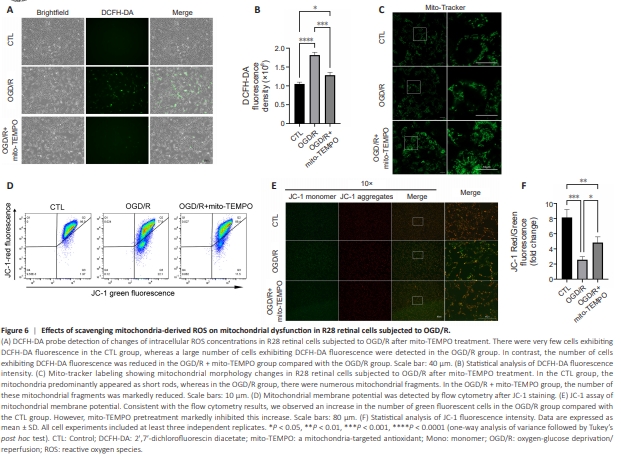
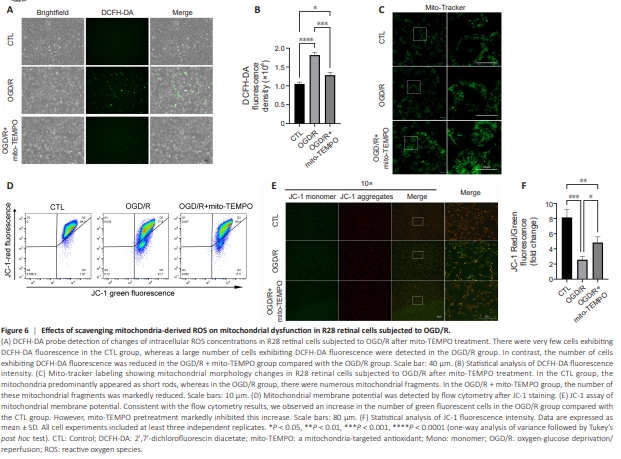
As VDAC1 is a critical regulator of mitochondrial membrane permeability (He et al., 2022), we hypothesized that VDAC1 oligomerization-mediated regulation of cellular ROS mainly depends on its regulation of mitochondria2derived ROS produced in response to OGD/R in R28 retinal cells. To test this, we used mito-TEMPO, a mitochondria-targeted antioxidant, to scavenge mitochondria-derived ROS. As shown in Figure 6A and B, the increase in ROS levels induced by OGD/R compared with control group was decreased by mitoTEMPO to a similar degree as that seen with VBIT-12 treatment (Figure 5D and E), indicating a central role for VDAC1 oligomerization in the regulation of mitochondria-derived ROS and oxidative stress. Next, we evaluated the effect of mito-TEMPO treatment on mitochondrial dysfunction using Mito-Tracker Green staining and JC-1 staining. The results indicated that the increased mitochondrial fission induced by OGD/R was reduced by mito-TEMPO (Figure 6C) to a similar degree as that seen with VBIT-12 treatment (Figure 5F). Regarding the reduction in mitochondrial membrane potential following OGD/R compared with control group, mito-TEMPO treatment showed a protective effect (Figure 6D–F) comparable to that of VBIT-12 treatment (Figure 5G–I). These results demonstrate that VDAC1 oligomerizationmediated regulation of mitochondria-derived ROS plays a central role in mitochondrial dysfunction in R28 retinal cells following OGD/R.
Figure 7 | Effects of scavenging mitochondria-derived ROS on PANoptosis in R28 retinal cells subjected to OGD/R.
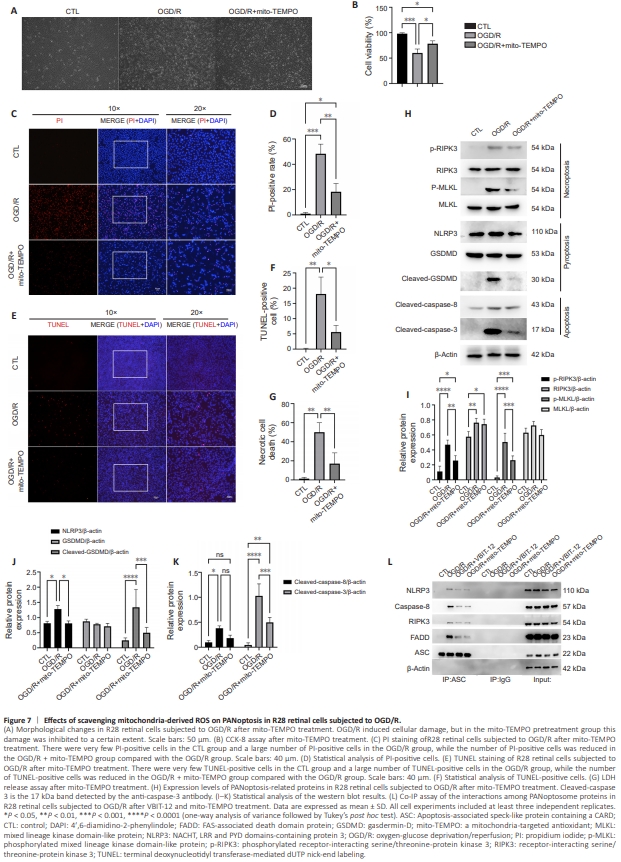
To explore the role of VDAC1 oligomerization-mediated regulation of mitochondria-derived ROS on PANoptosis, we evaluated the effect of mitochondria-derived ROS scavenging on PANoptosis in R28 retinal cells following OGD/R. As shown in Figure 7A, mito-TEMPO decreased the cell injury induced by OGD/R compared with control group. Furthermore, CCK8 assay indicated that mito-TEMPO had a similar protective effect on cell viability (Figure 7B) as did inhibition of VDAC1 oligomerization (Figure 4B). Next, PI and TUNEL staining were used to assess the effect of mito-TEMPO on cell death. We found that mito-TEMPO treatment reduced the proportion of PI-positive cells following OGD/R (Figure 7C and D). The proportion of TUNELpositive cells was also reduced in the mito-TEMPO group in comparison with the OGD/R group (Figure 7E and F). mito-TEMPO treatment showed similar protective effects as VBIT-12 treatment (Figure 4D and F). In addition, LDHrelease assay verified the protective effect of mito-TEMPO on necrotic cell death induced by OGD/R (Figure 7G). Then, we investigated the effect of mitochondria-derived ROS scavenging on the three cell death signaling cascades involved in PANoptosis. The increase in RIPK3 and MLKL phosphorylation was reduced by mito-TEMPO treatment in R28 retinal cells subjected to OGD/R, indicating an inhibitory effect of mitochondria-derived ROS scavenging on necroptosis activation and execution (Figure 7H and I). mito-TEMPO treatment also decreased the NLRP3 upregulation and GSDMD cleavage (key events for the activation and execution of pyroptosis) induced by OGD/R compared with control group (Figure 7H and J). Regarding apoptosis, mito-TEMPO treatment decreased the cleavage of caspase-8 and caspase-3 in R28 retinal cells induced by OGD/R compared with control group (Figure 7H and K). As PANoptosome complex assembly is considered a key event in PANoptosis induction (Wang et al., 2022b), we conducted a co-immunoprecipitation assay to assess complex assembly. As shown in Figure 7L, the key components of the PANoptosome complex (NLRP3, caspase-8, RIPK3, and FADD), were immunoprecipitated from lysates of R28 retinal cells subjected to OGD/R by an antibody to ASC (a key adapter of PANoptosome complex (Wang and Kanneganti, 2021)), indicating that OGD/R induced assembly of thePANoptosome complex compared with control group. Treatment with VBIT12 suppressed interactions between ASC and the other components of the PANoptosome complex, which demonstrated that inhibition of VDAC1 oligomerization reduced PANoptosis induction in R28 retinal cells subjected to OGD/R compared with control group. mito-TEMPO treatment also decreased assembly of PANoptosome complex, indicating that mitochondria-derived ROS scavenging suppressed PANoptosis induction in R28 retinal cells subjected to OGD/R, and the level of inhibition was comparable to that seen with VBIT-12 treatment. Collectively, these results demonstrate that mitochondria-derived ROS are critical to PANoptosis induction and play a central role in VDAC1 oligomerization-mediated regulation of PANoptosis following OGD/R.
Figure 8 | Effects of VBIT-12 on PANoptosis in the retinas of aHIOP rats.
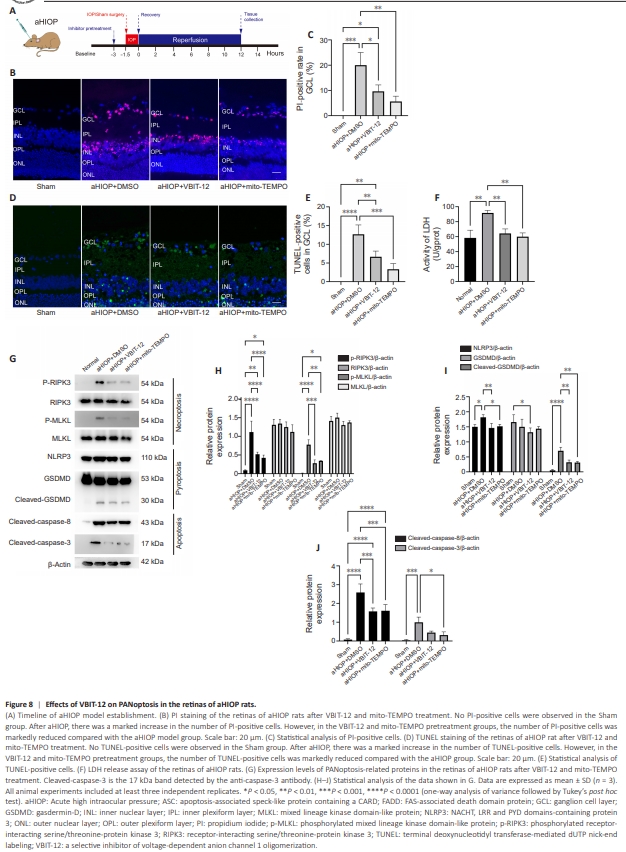
To evaluate the role of VDAC1 oligomerization in vivo, we established the aHIOP model, a commonly used model of retinal I/R injury (Figure 8A). As shown by PI staining, necrotic cell death was induced in the ganglion cells layer and inner nuclear layer in the aHIOP group compared with sham group (Figure 8B). The proportion of PI-positive cells in the aHIOP group was decreased by VBIT-12 treatment (Figure 8B and C). mito-TEMPO treatment also decreased necrotic cell death in the aHIOP group (Figure 8B and C). As indicated by TUNEL staining, both VBIT-12 treatment and mito-TEMPO treatment reduced apoptotic cell death in the aHIOP group (Figure 8D and E). LDH activity assay also verified the protective effect of VBIT-12 treatment and mito-TEMPO treatment against necrotic cell death induced by aHIOP (Figure 8F). Next, we analyzed changes in the expression of key regulators in the cell death signaling cascades involved in PANoptosis in the retinas of aHIOP rats. Increased levels of RIPK3 and MLKL phosphorylation were detected in the aHIOP group compared with sham group, indicating activation of necroptoticcell death signaling (Figure 8G and H). Both VBIT-12 treatment and mitoTEMPO treatment reduced RIPK3 and MLKL phosphorylation, indicating an in vivo protective effect of inhibition of VDAC1 oligomerization and mitochondria-derived ROS scavenging on necroptosis activation and execution (Figure 8G and H). Additionally, NLRP3 expression and GSDMD cleavage were increased in the aHIOP group compared with sham group, while VBIT-12 treatment and mito-TEMPO treatment suppressed activation of pyroptotic cell death signaling (Figure 8G and I). Regarding key apoptotic regulators, cleavage of caspase-8 and caspase-3 was detected in the aHIOP group (Figure 8G and J). Furthermore, both VBIT-12 treatment and mito-TEMPO treatment decreased cleavage of these regulators, indicating reduced activation of apoptotic cell death signaling. Collectively, these results demonstrate that inhibition of VDAC1 oligomerization and mitochondria-derived ROS scavenging suppresses PANoptosis following retinal I/R injury in vivo.