脑损伤
-
Figure 1 | Intermittent hypoxia (IH) enhances microcirculation within the prefrontal cortex (PFC) and hippocampus without inducing neurovascular damage.

Young adult mice were exposed to 10 5-minute periods of hypoxia (13% O2) interspersed with 5 minutes of normoxia (21% O2), once daily for 14 consecutives (Figure 1A). Compared with the control group, IH significantly increased CBF (P = 0.0119) and oxygen saturation (P = 0.0030), as measured under normoxia post-treatment in both the prefrontal cortex (PFC) and hippocampus (Figure 1B and C). Moreover, these increases in brain perfusion and oxygenation were not accompanied by vascular damage, as evidenced by sodium fluorescein leakage measurements (Figure 1D), or neurocellular degeneration, as evidenced by Nissl staining (Figure 1E), at 14 days post-IH, suggesting that IH can improve brain metabolism without inducing chronic neurovascular damage. To examine if this effect of IH was related to angiogenesis or alterations in neurovascular structure, we labeled the vascular endothelium by injecting lectin into the tail vein and conducted morphometric analyses, both in situ and in brain slices. To enhance tissue transparency while preserving fluorescent signals, the whole brain and the brain tissue sections were subsequently subjected to a clearing process (Additional Figure 1A). Comprehensive examination of cerebral vasculature in situ, via tissue clearing and imaging, revealed notable increases in vascular density and the number of branching points within both the PFC and hippocampus of IH-exposed mice compared with sham-treated controls (Figure 1F and G). In accordance with the findings in intact brains, subsequent quantitative analysis of the microvasculature within brain tissue slices demonstrated marked increases in vessel length, vessel area, vessel volume, and the number of branch points in the PFC (Figure 1H–K) and hippocampus (Figure 1L–O) compared with sham controls. We also analyzed vascular parameters such as mean vessel diameter, vessel resistance, and vessel straightness, but there were no marked group differences in these basic vascular characteristics (Additional Figure 1B–G). Thus, IH enhanced CBF and oxygen saturation by promoting angiogenesis.
Figure 3 | Intermittent hypoxia (IH) after bilateral carotid artery stenosis (BCAS) surgery improves cerebral perfusion and microcirculation within cognition-related brain regions of vascular cognitive impairment and dementia model mice.
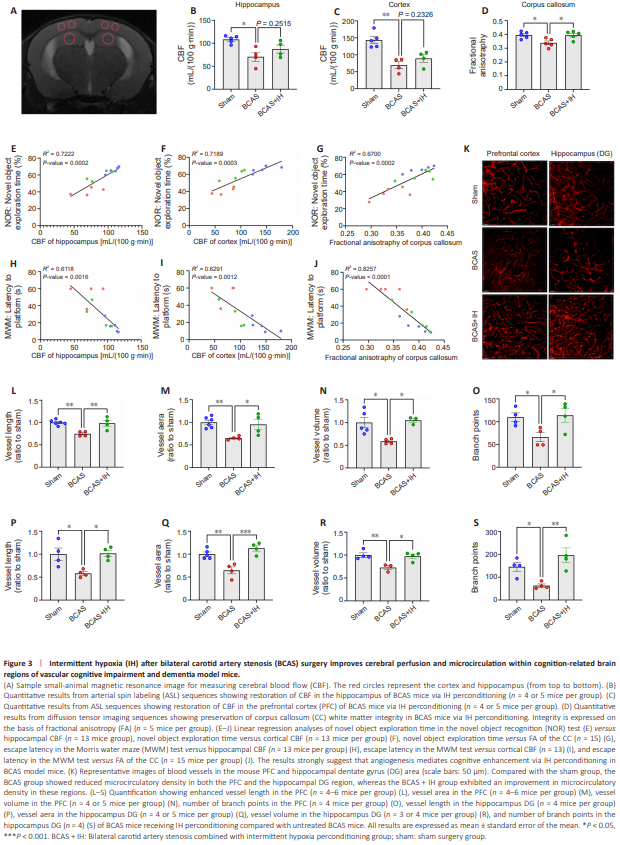
As IH treatment following BCAS was more effective than preconditioning in ameliorating cognitive impairments in VCID mice, we examined the influence of this treatment regimen on CBF and microcirculation in the PFC and hippocampus. Small-animal magnetic resonance imaging revealed a pronounced reduction in CBF within the hippocampus and PFC of VCID model mice versus the sham surgery group, whereas there was no marked reduction in CBF in the VCID + IH group (Figure 3A–C). This suggests IH intervention partially reversed the BCAS surgery-induced reduction in CBF. Furthermore, diffusion tensor imaging revealed damage to corpus callosum fibers in VCID model mice; this damage was also attenuated by IH perconditioning (Figure 3D). Linear regression analyses further revealed positive correlations of CBF in the hippocampus and PFC with both corpus callosum white matter integrity and cognitive performance (Figure 3E–J). In accordance with these in situ results, 3D reconstruction of microvessels in brain slices (Figure 3K) revealed enhanced vessel length, vessel area, vessel volume, and the number of branch points in the PFC and hippocampus dentate gyrus region of VCID + IH group mice versus VCID mice (Figure 3L–S). We also analyzed basic vascular characteristics, including vessel mean diameter, vessel resistance, and vessel straightness; however, no marked differences in these parameters were found among the three groups (Additional Figure 2A–F). Thus, enhanced CBF because of IH treatment following BCAS likely helps mitigate both corpus callosum damage and cognitive dysfunction.