中国神经再生研究(英文版) ›› 2026, Vol. 21 ›› Issue (8): 3779-3787.doi: 10.4103/NRR.NRR-D-25-00308
阿尔茨海默病病理致轻度认知障碍的认知异质性
Cognitive heterogeneity in mild cognitive impairment due to Alzheimer’s disease pathology
Siyun Chen1, David P. Salmon2, 3, Howard H. Feldman3, 4, Karen Messer5, Mark W. Bondi3, 6, Dongsheng Xu7, Yuqi Qiu8, *, Diane M. Jacobs2, 3, *, The Alzheimer’s Disease Neuroimaging Initiative
- 1Department of Rehabilitation Medicine, Shanghai Jiao Tong University Affiliated Sixth People’s Hospital, Shanghai, China;
2Department of Neurosciences, University of California San Diego, La Jolla, CA, USA;
3UC San Diego Shiley-Marcos Alzheimer’s Disease Research Center, University of California San Diego, La Jolla, CA, USA;
4Alzheimer’s Disease Cooperative Study, Department of Neurosciences, University of California San Diego, La Jolla, CA, USA;
5Division of Biostatistics and Bioinformatics, Herbert Wertheim School of Public Health, University of California San Diego, La Jolla, CA, USA;
6Department of Psychiatry, University of California San Diego, La Jolla, CA, USA;
7College of Rehabilitation Science, Shanghai University of Traditional Chinese Medicine, Shanghai, China;
8KLATASDS-MOE, School of Statistics, East China Normal University, Shanghai, China
摘要:
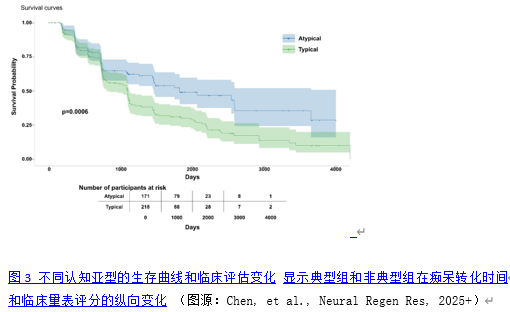
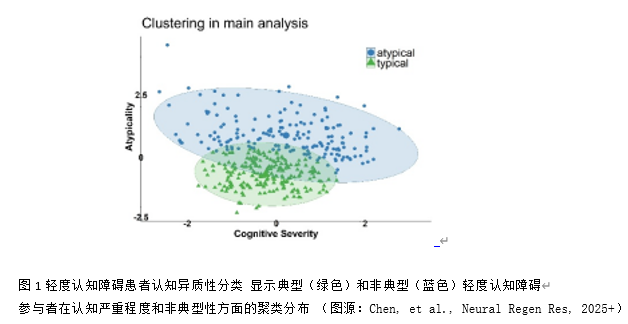
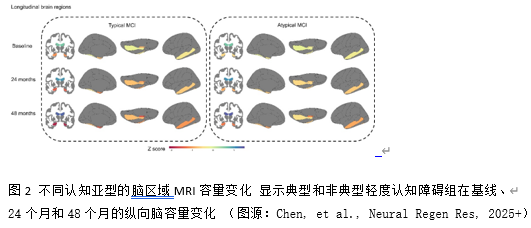
传统的临床亚型分类(如遗忘型和非遗忘型轻度认知障碍)依赖于对认知测试重叠表现模式的主观解释,可能导致不可靠的分类。更精密和客观的轻度认知障碍亚型分类可以通过数据驱动的聚类技术来实现。然而,以往研究并未将队列限制在阿尔茨海默病病理的轻度认知障碍患者中,因此在严格定义的生物标志物阳性前驱期阿尔茨海默病队列中认知变异性的性质及其对疾病进展的影响仍然未知。此研究对阿尔茨海默病致轻度认知障碍患者的认知异质性进行了分析,并评估其预后价值。从阿尔茨海默病神经影像学倡议(ADNI)队列中获取了389例经脑脊液(CSF)生物标志物确诊为阿尔茨海默病的轻度认知障碍患者的神经心理测试数据。通过主成分分析和基于模型的聚类分析识别认知特征,并通过100次Bootstrap验证。成对比较测试了识别出的亚组在参与者特征、认知和临床结局评分、脑脊液生物标志物水平以及MRI检测的脑体积方面的差异。纵向分析评估了48个月内MRI体积测量和临床结局变化率的差异。生存分析评估了转化为痴呆的风险。α-突触核蛋白水平和白质高信号(WMH)体积被纳入敏感性分析。识别出两种不同的认知特征:一个“典型”组(占样本的56.04%),其记忆测试得分相对于非记忆测试得分相对较低;一个“非典型”组(占样本的43.96%),其记忆与非记忆测量之间的差异较小,表明认知领域中损伤模式更为均匀。尽管两组在整体认知障碍程度和脑脊液阿尔茨海默病生物标志物水平上相当,但典型组在多个脑区每6个月的萎缩速率均显著加快(海马区:29.02 mm³,标准误=10.13,p=0.005;全脑:1799.85 mm³,标准误=781.57,P=0.023; 内嗅皮层:22.26 mm³,标准误=11.15,p=0.048;梭状回:66.24 mm³,标准误=28.53,P=0.021)。生存分析显示,典型组的痴呆转化风险显著更高(hazard ratio: 1.70, 95% CI: 1.27, 2.27)且进展时间更短。这些发现经调整共病病理后仍保持一致。总之,这项研究成功识别出阿尔茨海默病相关轻度认知障碍的两种认知亚型,探索这一认知异质性对于理解前驱期阿尔茨海默病的疾病进展模式、改善预后预测以及指导临床试验设计具有重要价值。典型认知亚型识别出的群体具有近两倍的痴呆进展风险,这对临床实践具有重要价值:可以增强预后咨询,指导临床试验分层,且该分类方法可从标准神经心理学评估中获得,具有临床适用性。
https://orcid.org/0000-0003-0886-6784 (Diane M. Jacobs); https://orcid.org/0009-0006-2373-536X (Yuqi Qiu)
 #br#
#br#
 #br#
#br#