中国神经再生研究(英文版) ›› 2026, Vol. 21 ›› Issue (8): 3842-3853.doi: 10.4103/NRR.NRR-D-25-00069
高蛋氨酸饮食诱导组蛋白乙酰化异常和突触可塑性失调引起认知障碍
Aberrant histone acetylation and dysregulated synaptic plasticity in cognitive impairment induced by a high-methionine diet
Jianting Li1, #, Yuan Fu1, #, Xiaolong Gu1, #, Qi Xie1, Zengli Liu1, Zhihua Cao1, Lu Li1, Jiaxin Ren1, Yang Li2, Hailan Yang3, Zhiwei Peng1, Zhizhen Liu1, Jun Xie1, *#br#
- 1Department of Biochemistry and Molecular Biology, College of Basic Medicine, Shanxi Key Laboratory of Birth Defect and Cell Regeneration, MOE Key Laboratory of Coal Environmental Pathogenicity and Prevention, Shanxi Medical University, Taiyuan, Shanxi Province, China;
2Department of Nephrology, First Clinical College of Shanxi Medical University, Taiyuan, Shanxi Province, China;
3Department of Obstetrics, First Clinical College of Shanxi Medical University, Taiyuan, Shanxi Province, China
摘要:
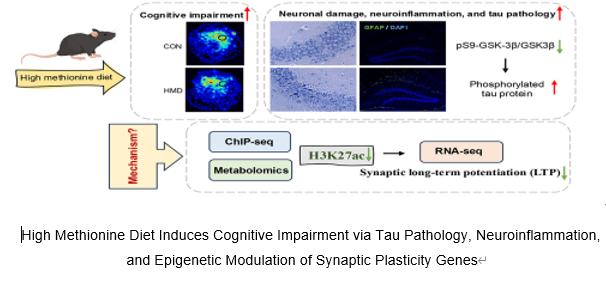
高同型半胱氨酸水平是神经退行性疾病认知障碍主要风险因素。表观遗传修饰,尤其是组蛋白乙酰化,已被证实与认知障碍的进展相关;然而,高同型半胱氨酸血症诱导的认知障碍的具体机制尚不明确。实验通过长期饲喂高蛋氨酸饮食建立了高同型半胱氨酸诱导认知障碍小鼠模型。结果显示,小鼠海马和大脑皮质中组蛋白H3赖氨酸27乙酰化水平显著下降,导致明显的认知缺陷和神经炎症。此外,代谢组学分析和染色质免疫共沉淀测序揭示了高同型半胱氨酸代谢物水平的显著变化,并鉴定出与突触长时程增强相关的组蛋白H3赖氨酸27乙酰化靶向基因,包括Gria1,Gria3,Grin2a, Grin2b,Slc1a1,Slc24a2,Ptk2b和Src。RNA测序证实了高同型半胱氨酸诱导的神经退行性病理变化。最后,体外实验证实高同型半胱氨酸处理的HT-22细胞中组蛋白H3赖氨酸27乙酰化水平下降会下调这些靶基因的表达,从而损害突触可塑性。这些发现表明,由组蛋白H3赖氨酸27乙酰化调控的突触长时程增强相关基因异常表达是高同型半胱氨酸诱导认知障碍的关键驱动因素。靶向组蛋白H3赖氨酸27乙酰化介导的表观遗传失调可能代表一种有前景的治疗策略,为认知障碍患者及更广泛的神经退行性疾病提供潜在干预途径。
https://orcid.org/0000-0002-6161-5696 (Jun Xie)