NRR:吉大李炳锦团队发现禁食9小时对卵巢摘除小鼠可产生类似抗抑郁药的效果
中国吉林大学李炳锦团队最新研究发现,9h急性禁食能够改善雌性小鼠抑郁样行为,这一作用与哺乳动物雷帕霉素复合物靶点1(mTORC1)信号通路激活而产生的神经可塑性密切相关。
抑郁是一种常见的复发性精神疾病,已被世卫组织列为全球最大的残疾原因[1]。然而,抗抑郁药的治疗效果仍不令人满意,尤其长期服用抗抑郁药会造成食欲和体重增加。因此能否从饮食调控的角度改善抑郁,具有重要的理论和临床转化意义。既往研究已发现,9h急性禁食可减少正常小鼠强迫游泳不动时间,然而其是否也对雌性抑郁小鼠有治疗作用,仍需进一步探讨。卵巢摘除能迅速降低雌激素和其他卵巢激素水平,这有效地模拟了女性围绝经期[2, 3]。低雌激素水平可诱发啮齿类动物的抑郁样行为[4]。而雌性动物对氯胺酮更加敏感,这一现象与雷帕霉素激活机制靶点的替代机制有关[5]。已有研究指出脑源性神经营养因子-哺乳动物雷帕霉素复合物靶点1信号通路与氯胺酮快速抗抑郁样作用的潜在机制有关,快速抗抑郁药物的机制具体表现为脑源性神经营养因子表达的增加,以及对哺乳动物雷帕霉素复合物靶点1信号通路的直接作用[6-9]。激活哺乳动物雷帕霉素复合物靶点1信号通路最终诱导突触后密度蛋白95水平升高,促进神经发生和突触发生,逆转脑容量萎缩,从而改善抑郁症状[6, 10]。
近期,吉林大学的李炳锦团队在《中国神经再生研究(英文版)》(Neural Regeneration Research)上发表了题为“Fasting produces antidepressant-like effects via activating mammalian target of rapamycin complex 1 signaling pathway in ovariectomized mice” 的研究,发现9h禁食对卵巢摘除小鼠可产生类似抗抑郁药的效果(图1),其可激活哺乳动物雷帕霉素复合物靶点1信号通路,上调脑源性神经营养因子水平,并增加海马中突触蛋白突触后密度蛋白95和树突棘密度,同时增加了海马CA1区的长时程增强(图2),且上述变化可被雷帕霉素逆转,表明禁食可通过激活哺乳动物雷帕霉素复合物靶点1信号通路改变海马CA1区突触可塑性进而产生抗抑郁的作用(图3)。然而这一研究仅对急性禁食对抑郁样行为作用及其机制进行了探讨,长期禁食是否有相似的作用仍需进一步探讨,另外禁食能否应用于临床抑郁的治疗干预还需进一步分析。
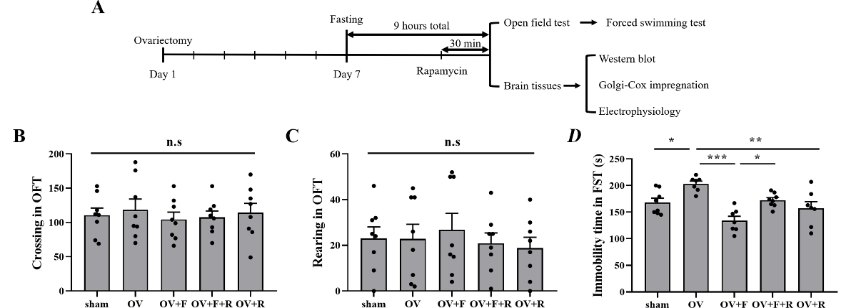
图1 禁食逆转卵巢摘除诱导的抑郁样行为(图源:Cheng et al., Neural Regen Res, 2023)
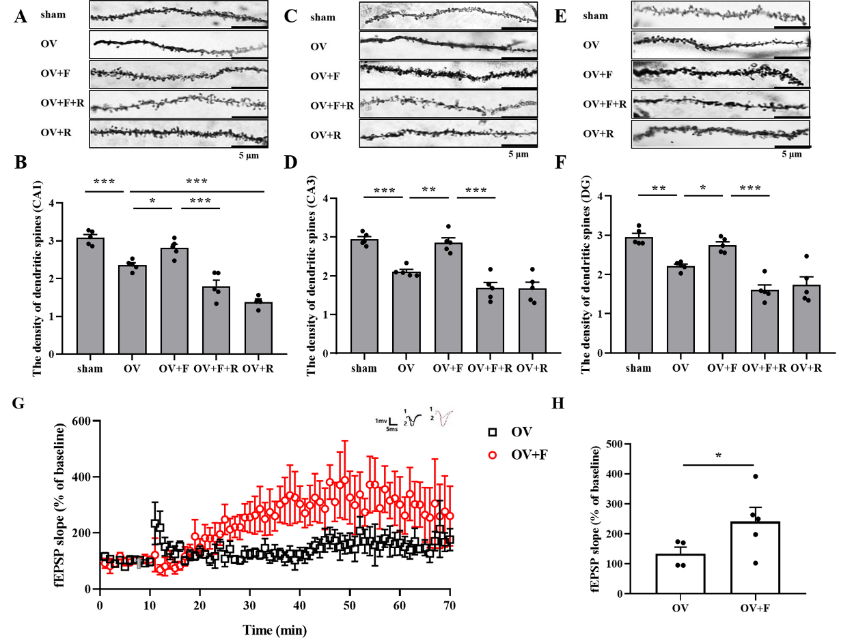
图2 禁食可增加海马CA1、CA3和齿状回区树突棘的密度,并增强海马CA1区长时程增强(图源:Cheng et al., Neural Regen Res, 2023)
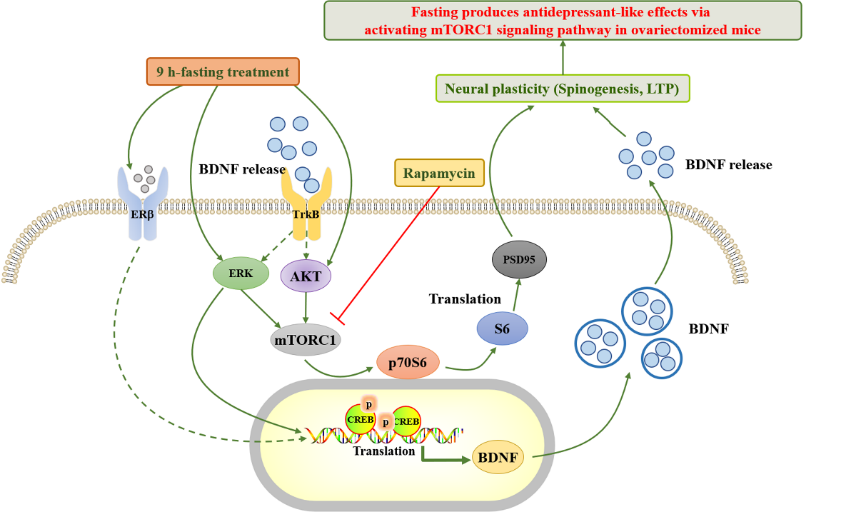
图3 禁食激活哺乳动物雷帕霉素复合物靶点1信号通路(图源:Cheng et al., Neural Regen Res, 2023)
参考文献
[1] World Health Organization. Depression and other common mental disorders: global health estimates. https://apps.who.int/iris/rest/bitstreams/1080542/retrieve. 2017.
[2] Greendale GA, Derby CA, Maki PM. Perimenopause and cognition. Obstet Gynecol Clin North Am. 2011;38(3):519-535.
[3] Gore AC, Oung T, Woller MJ. Age-related changes in hypothalamic gonadotropin-releasing hormone and N-methyl-D-aspartate receptor gene expression, and their regulation by oestrogen, in the female rat. J Neuroendocrinol. 2002;14(4):300-309.
[4] Walf AA, Frye CA. A review and update of mechanisms of estrogen in the hippocampus and amygdala for anxiety and depression behavior. Neuropsychopharmacology. 2006;31(6):1097-1111.
[5] Carrier N, Kabbaj M. Sex differences in the antidepressant-like effects of ketamine. Neuropharmacology. 2013;70:27-34.
[6] Wohleb ES, Gerhard D, Thomas A, et al. Molecular and cellular mechanisms of rapid-acting antidepressants ketamine and scopolamine. Curr Neuropharmacol. 2017;15(1):11-20.
[7] Abdallah CG, Averill LA, Gueorguieva R, et al. Modulation of the antidepressant effects of ketamine by the mTORC1 inhibitor rapamycin. Neuropsychopharmacology. 2020;45(6):990-997.
[8] Fogaça MV, Fukumoto K, Franklin T, et al. N-Methyl-D-aspartate receptor antagonist d-methadone produces rapid, mTORC1-dependent antidepressant effects. Neuropsychopharmacology. 2019;44(13):2230-2238.
[9] Duman RS, Shinohara R, Fogaça MV, et al. Neurobiology of rapid-acting antidepressants: convergent effects on GluA1-synaptic function. Mol Psychiatry. 2019;24(12):1816-1832.
[10] Duman RS, Aghajanian GK, Sanacora G, et al. Synaptic plasticity and depression: new insights from stress and rapid-acting antidepressants. Nat Med. 2016;22(3):238-249.
原文链接:https://doi.org/10.4103/1673-5374.367928


