NRR:中国青岛大学附属医院栗世方和万芪团队报道AAV介导的ROCK2下调促进缺血性脑卒中大鼠突触发生和神经发生
撰文:师留留
神经突生长和突触发生是缺血性脑卒中后功能恢复的关键步骤。成年哺乳动物中枢神经系统受损的轴突表现出有限的再生能力,导致持久的神经功能缺陷。这主要是受损的中枢神经系统释放大量的轴突抑制因子,激活RhoA/ROCK信号通路,抑制受损的神经功能恢复[1-3]。RhoA/ROCK信号通路被认为在介导中枢神经系统疾病的发病机制中起重要作用。抑制ROCK2信号通路在不同中枢神经系统疾病的神经保护。前期的研究表明,抑制ROCK信号通路可以促进视神经损伤、脊髓损伤和创伤性脑损伤后的轴突生长及神经元存活,起到神经保护作用[4-6]。目前尚不清楚抑制ROCK2对缺血性脑卒中后神经发生及突触发生的影响。既往的研究表明,应用ROCK抑制剂可以促进缺血性脑卒中后神经功能恢复[7-9],然而由于ROCK抑制剂的不良反应,如低血压、肝肾功能损害以及其缺乏选择性和低效力性,因此限制了其临床应用。因此探索抑制ROCK2表达的新方法并研究其对缺血性脑卒中后神经发生与突触发生的关系,可能成为缺血性脑卒中治疗的新方法。#br#
青岛大学附属医院栗世方和万芪团队在《中国神经再生研究(英文)》(Neural Regeneration Research)发表的研究发现,携带外源基因sh.ROCK2的AAV能够成功转染脑组织并存活至少6周,有效地敲低了ROCK2的表达。这促进了缺血性脑卒中后神经发生、突触发生和神经元存活,从而增加了神经可塑性,促进了神经功能的恢复。因此,利用AAV病毒携带sh.ROCK2基因特异性敲低脑组织中ROCK2的表达,可能为ROCK2基因治疗缺血性脑卒中的远期预后提供一种潜在的方法。师留留和朱婷为论文共同第一作者,栗世方教授和万芪教授为论文共同通讯作者。#br#
既往研究发现,中枢神经系统损伤后突触发生的抑制因子包括髓鞘相关糖蛋白、硫酸软骨素蛋白聚糖、少突胶质髓鞘糖蛋白和神经突生长抑制剂-a (Nogo-A)[10, 11]。这些结构独特的蛋白质都与Nogo受体(NgR)结合,通过激活RhoA及其下游底物ROCK来抑制轴突生长[1-3]。因此,该实验首先观察AAV-sh.ROCK2基因治疗对MCAO大鼠模型后28天不同时间点行为学及体重的影响,实验结果显示AAV-sh.ROCK2基因治疗可以促进缺血性脑卒中大鼠模型中神经行为学改善及体重指数增加(图1),这些结果表明敲低ROCK2的表达可以促进缺血性脑卒中后神经功能的恢复。组织学和行为学结果对于评估缺血性脑卒中的长期预后是必要的,也是决定治疗方案是否可以进入临床试验的主要参数[12-14],接下来他们用尼氏染色评估MCAO大鼠模型28天后的脑萎缩体积。实验结果显示, AAV-sh.ROCK2基因治疗可以减少脑萎缩体积(图2)。以上结果说明, AAV-sh.ROCK2基因治疗可以改善缺血性脑卒中的长期预后。
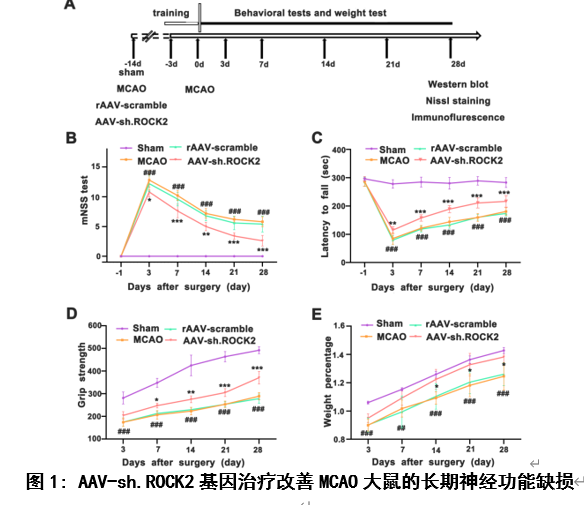 #br#
#br#
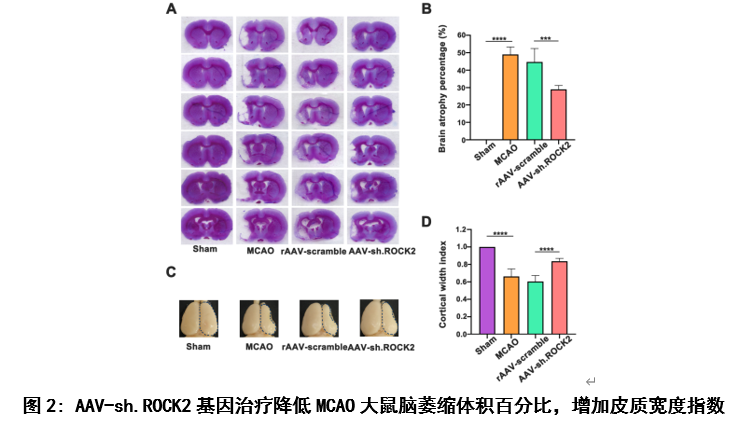
为了评价AAV-sh.ROCK2基因治疗对缺血性脑卒中后神经发生的影响,他们用免疫荧光染色法检测MCAO造模后28天大鼠脑室下区和纹状体中神经发生标志物Nestin(神经干细胞特异性标志物)和DCX(未成熟神经元的标志物)的表达(图3),研究结果显示:AAV-sh.ROCK2基因治疗可以促进缺血性脑卒中大鼠模型脑室下区及纹状体区神经发生,并促进神经元存活。从而促进缺血性脑卒中后的神经可塑性。
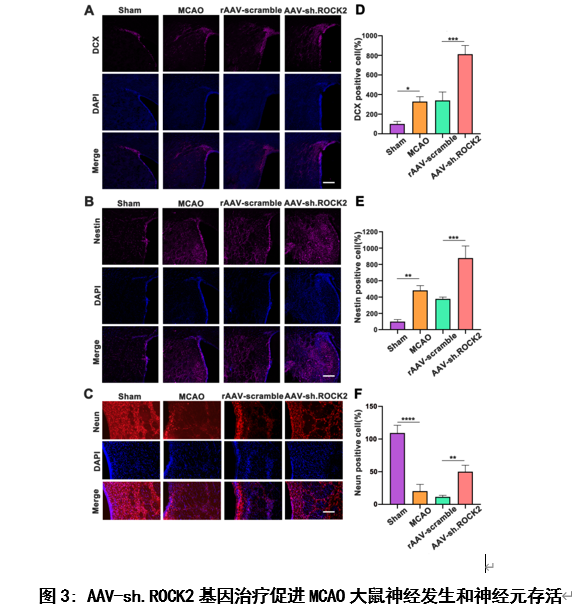 #br#
神经发生后从SVZ向缺血纹状体迁移的NSCs和NPCs具有突触发生的能力,形成突触连接重建神经网络系统。这一过程对缺血性脑卒中后的功能重建至关重要。因此,作者进一步用免疫荧光染色法检测MCAO造模后28天大鼠脑纹状体中生长相关结合蛋白43 (GAP43)和突触素相关蛋白(SYP)的表达表达水平(图4),它们是与突触可塑性相关的重要分子标记物[15]。研究结果显示:AAV-sh.ROCK2基因治疗可以促进缺血性脑卒中大鼠模型中突触发生。
#br#
神经发生后从SVZ向缺血纹状体迁移的NSCs和NPCs具有突触发生的能力,形成突触连接重建神经网络系统。这一过程对缺血性脑卒中后的功能重建至关重要。因此,作者进一步用免疫荧光染色法检测MCAO造模后28天大鼠脑纹状体中生长相关结合蛋白43 (GAP43)和突触素相关蛋白(SYP)的表达表达水平(图4),它们是与突触可塑性相关的重要分子标记物[15]。研究结果显示:AAV-sh.ROCK2基因治疗可以促进缺血性脑卒中大鼠模型中突触发生。
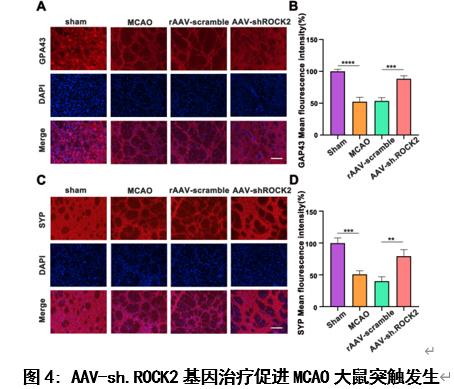 #br#
以上结果表明,AAV-sh.ROCK2基因治疗促进缺血性脑卒中后神经发生及突触发生,改善神经功能预后,为缺血性脑卒中的基因治疗提供可行方法。
#br#
以上结果表明,AAV-sh.ROCK2基因治疗促进缺血性脑卒中后神经发生及突触发生,改善神经功能预后,为缺血性脑卒中的基因治疗提供可行方法。
总之,抑制ROCK2信号通路对缺血性脑卒中神经功能恢复至关重要。作者的研究表明,利用AAV介导的ROCK2下调促进缺血性脑卒中后神经发生和突触发生,促进神经元存活,进而促进神经功能的恢复,另外,AAV可以在脑内长期表达,这为基因治疗提供了有效的载体。该研究将为缺血性脑卒中基因治疗提供参考。当然本实验也存在一定的不足之处,该实验采用成年雄性大鼠制作MCAO模型。缺血性脑卒中多发生在合并糖尿病、高血压、高脂血症的老年患者中,因此后期的研究要考虑老年大鼠。另外他们在MCAO模型前2周注射病毒。这种提前预给药治疗的方法在临床治疗中不易实施。后期他们可以考虑在MCAO造模后注射腺相关病毒来评价疗效。侧脑室内注射可引起继发性损伤,他们可以在减小损伤情况下评价鞘内注射AAV颅内转染的效果或者通过静脉注射靶向转染脑组织。但是,该研究证实了ROCK2抑制对MCAO大鼠的神经发生及突触发生的促进作用,为慢性缺血性脑卒中的基因治疗提供了一种可行的治疗方法。
#br#
参考文献#br#
[1] Fujita Y, Yamashita T. Axon growth inhibition by RhoA/ROCK in the central nervous system. Front Neurosci. 2014;8:338.#br#
[2] Zhang J, Li Z, Liu W, et al. Effects of bone marrow mesenchymal stem cells transplantation on the recovery of neurological functions and the expression of Nogo-A, NgR, Rhoa, and ROCK in rats with experimentally-induced convalescent cerebral ischemia. Ann Transl Med. 2020;8(6):390.#br#
[3] Gao J, Liu J, Yao M, et al. Panax notoginseng Saponins Stimulates Neurogenesis and Neurological Restoration After Microsphere-Induced Cerebral Embolism in Rats Partially Via mTOR Signaling. Front Pharmacol. 2022;13:889404.#br#
[4] Koch JC, Tönges L, Barski E, et al. ROCK2 is a major regulator of axonal degeneration, neuronal death and axonal regeneration in the CNS. Cell Death Dis. 2014;5(5):e1225.#br#
[5] Challagundla M, Koch JC, Ribas VT, et al. AAV-mediated expression of BAG1 and ROCK2-shRNA promote neuronal survival and axonal sprouting in a rat model of rubrospinal tract injury. J Neurochem. 2015;134(2):261-275.#br#
[6] Willis EF, Kim SJ, Chen W, et al. ROCK2 regulates microglia proliferation and neuronal survival after traumatic brain injury. Brain Behav Immun. 2024;117:181-194.#br#
[7] Tönges L, Frank T, Tatenhorst L, et al. Inhibition of rho kinase enhances survival of dopaminergic neurons and attenuates axonal loss in a mouse model of Parkinson's disease. Brain. 2012;135(Pt 11):3355-3370.#br#
[8] Xu L, Liu X, Guo C, et al. Inhibition of ROCK2 kinase activity improved behavioral deficits and reduced neuron damage in a DEACMP rat model. Brain Res Bull. 2022;180:24-30.#br#
[9] Li L, Liu B. ROCK inhibitor Y‑27632 protects rats against cerebral ischemia/reperfusion‑induced behavioral deficits and hippocampal damage. Mol Med Rep. 2019;20(4):3395-3405.#br#
[10] Schwab ME, Strittmatter SM. Nogo limits neural plasticity and recovery from injury. Curr Opin Neurobiol. 2014;27:53-60.#br#
[11] Chambel SS, Cruz CD. Axonal growth inhibitors and their receptors in spinal cord injury: from biology to clinical translation. Neural Regen Res. 2023;18(12):2573-2581.#br#
[12] Fisher M, Feuerstein G, Howells DW, et al. Update of the stroke therapy academic industry roundtable preclinical recommendations. Stroke. 2009;40(6):2244-2250.#br#
[13] Fluri F, Schuhmann MK, Kleinschnitz C. Animal models of ischemic stroke and their application in clinical research. Drug Des Devel Ther. 2015;9:3445-3454.#br#
[14] McBride DW, Klebe D, Tang J, et al. Correcting for brain swelling's effects on infarct volume calculation after middle cerebral artery occlusion in rats. Transl Stroke Res. 2015;6(4):323-38.#br#
[15] Yang K, Zhou Y, Zhou L, et al. Synaptic Plasticity After Focal Cerebral Ischemia Was Attenuated by Gap26 but Enhanced by GAP-134. Front Neurol. 2020;11:888.#br#
#br#
#br#
#br#
#br#
#br#
#br#
 #br#
#br#

 #br#
神经发生后从SVZ向缺血纹状体迁移的NSCs和NPCs具有突触发生的能力,形成突触连接重建神经网络系统。这一过程对缺血性脑卒中后的功能重建至关重要。因此,作者进一步用免疫荧光染色法检测MCAO造模后28天大鼠脑纹状体中生长相关结合蛋白43 (GAP43)和突触素相关蛋白(SYP)的表达表达水平(图4),它们是与突触可塑性相关的重要分子标记物[15]。研究结果显示:AAV-sh.ROCK2基因治疗可以促进缺血性脑卒中大鼠模型中突触发生。
#br#
神经发生后从SVZ向缺血纹状体迁移的NSCs和NPCs具有突触发生的能力,形成突触连接重建神经网络系统。这一过程对缺血性脑卒中后的功能重建至关重要。因此,作者进一步用免疫荧光染色法检测MCAO造模后28天大鼠脑纹状体中生长相关结合蛋白43 (GAP43)和突触素相关蛋白(SYP)的表达表达水平(图4),它们是与突触可塑性相关的重要分子标记物[15]。研究结果显示:AAV-sh.ROCK2基因治疗可以促进缺血性脑卒中大鼠模型中突触发生。
 #br#
以上结果表明,AAV-sh.ROCK2基因治疗促进缺血性脑卒中后神经发生及突触发生,改善神经功能预后,为缺血性脑卒中的基因治疗提供可行方法。
#br#
以上结果表明,AAV-sh.ROCK2基因治疗促进缺血性脑卒中后神经发生及突触发生,改善神经功能预后,为缺血性脑卒中的基因治疗提供可行方法。