NRR:天津中医药大学第一附属医院赵岚团队揭示神经干细胞治疗阿尔茨海默病的策略与前景
撰文:常军,李玉姣,单晓倩,陈曦,闫栩赫,刘建卫,赵岚
阿尔茨海默病是一种常见的神经退行性疾病,以进行性认知障碍为特征,对患者的生命和健康构成严重威胁[1, 2]。近期研究发现,阿尔茨海默病影响了全球5000多万人,预计2050年将超过1.52亿人[3]。现有药物治疗效果有限,难以逆转突触和神经元的损失,且副作用明显[4, 5]。减少淀粉样蛋白β或tau蛋白的策略对阿尔茨海默病没有明显效果,不能恢复受损的神经组织[4, 5]。最近有研究表明,神经干细胞可自我更新、分化和迁移,增强突触可塑性和神经发生。因此,它们被认为是一种有吸引力的阿尔茨海默病再生治疗方法。
来自中国天津中医药大学第一附属医院赵岚团队在《中国神经再生研究(英文版)》(Neural Regeneration Research)上发表了题为“Neural stem cells promote neuroplasticity: a promising therapeutic strategy for the treatment of Alzheimer’s disease”的综述。该综述发现,神经可塑性可在整个生命期存在,且随着年龄的增长而其能力不断下降,且在衰老和阿尔茨海默病患者中受损明显。因此,促进神经可塑性可能是缓解阿尔茨海默病的一个有前途的策略。神经干细胞不仅能减轻淀粉样蛋白β和tau蛋白水平,也能促进突触可塑性和神经发生以修复哺乳动物大脑的微环境,这被认为是改善阿尔茨海默病有巨大潜力的新疗法。
阿尔茨海默病是一种毁灭性的神经退行性疾病,现有药物只能暂时缓解症状,无法治愈阿尔茨海默病[3],亟需要安全且有效的治疗药物。神经干细胞具有强大的更新、分化、增殖能力,其能否缓解阿尔茨海默病认知以及记忆缺陷值得深入的研究。赵岚等通过对阿尔茨海默病临床和临床前样本研究进行分析,发现阿尔茨海默病神经可塑性显著受损,具体表现为突触可塑性和神经发生受损[6]。突触可塑性受损表现为阿尔茨海默病患者和小鼠模型海马和前额叶皮质长时程增强、突触密度、突触连接和树突棘密度以及突触后致密物95、突触素、脑源性神经营养因子和TrkB蛋白表达均明显降低[6-9]。研究发现,早期阿尔茨海默病神经发生受损,且随着阿尔茨海默病症状的加重,成年海马神经发生急剧下降,特别是神经元丢失,这与神经前体细胞存活、增殖和分化能力降低有关,也与脑源性神经营养因子和TrkB表达减少有关[10-12]。上述研究表明,阿尔茨海默病神经可塑性受损严重,这是其发病的关键原因,恢复和/或促进海马神经可塑性可能是预防和治疗阿尔茨海默病,特别是轻度认知障碍的一种有吸引力的策略。
鉴于神经干细胞可分化为神经元、星形胶质细胞和少突胶质细胞,并进行自我更新,为脑组织提供大量神经元[13]。这决定了神经干细胞在神经退行性疾病中的治疗潜力。神经干细胞有效治疗疾病的关键阶段包括迁移、存活、定向增殖和分化、逆转丢失神经元和突触。进一步分析发现,神经干细胞的迁移和增殖能力主要由多种因素决定,包括生长因子[14]、细胞因子[15]以及供体细胞的分化状态[16]。因此,调节生长因子和细胞因子水平以及供体细胞分化阶段对神经干细胞迁移、存活和增殖能力有促进作用。但是研究发现阿尔茨海默病内源性神经干细胞数量和功能下降[17]。内源性修复功能不足以补偿中枢神经系统中受损的神经干细胞,这可能与阿尔茨海默病的经典病理淀粉样蛋白β和(或)tau有关[18, 19]。因此,当务之急是探索能够减少淀粉样蛋白β和(或)tau表达的方法,以调节和维持阿尔茨海默病患者脑组织中内源性神经干细胞的正常平衡和再生潜力。
而后,赵岚等对神经干细胞治疗阿尔茨海默病的小鼠模型进行了简述,发现阿尔茨海默病小鼠模型主要包括5×FAD、3×Tg、APP/PS1、Tg2576、SAMP8、CaM/Tet-DTA小鼠等。阿尔茨海默病小鼠模型为药物研发提供了重要的依托。未来有必要在多种动物模型中进行临床前实验,直到有更完整的阿尔茨海默病动物模型,以确保临床前结果能更好地转化进临床。此外,对阿尔茨海默病的关键脑区进行靶向给药仍然是一个尚未解决的重大挑战,鲜有研究对神经干细胞的给药方式进行总结和分析。给药途径对神经干细胞的生物分布和疗效有重要影响。赵岚等发现阿尔茨海默病的主要给药方式包括鼻内、脑室内、静脉内和口服,这在临床应用中各有优缺点,而神经干细胞最佳给药方式仍有待深入的探索。
接下来,赵岚等深入分析了神经干细胞移植对阿尔茨海默病小鼠神经可塑性的影响,结果发现,神经干细胞可逆转阿尔茨海默病小鼠大脑皮质和海马中的突触损失,增加可塑性相关分子表达,但具体机制仍不清楚。有学者发现,敲除神经干细胞来源的脑源性神经营养因子可降低突触素水平[20],但不能改善3xTg阿尔茨海默病小鼠认知障碍和海马中突触的密度[21]。这提示神经干细胞促进突触可塑性的机制可能与脑源性神经营养因子调节密切相关。而过表达脑源性神经营养因子的神经干细胞可增强长时程增强,并增加突触密度[22],提高海马中突触后致密物95、突触素和脑源性神经营养因子蛋白水平,促进更多成熟的颗粒细胞进入颗粒细胞层,促进外源性神经干细胞与内源性细胞形成新的突触连接[22]。综上所述,脑源性神经营养因子在神经干细胞增强海马突触可塑性方面发挥了关键的作用。研究发现,外源性神经干细胞可在阿尔茨海默病小鼠海马中存活,并分化成神经元和星形胶质细胞,迁移到病变部位,修复和替换丢失的神经元,并促进新神经元与功能性神经回路的整合,最终形成功能性神经元并整合到神经回路中,这与神经干细胞介导的神经营养因子促进成年海马神经发生有关[17, 23-25]。
虽然神经干细胞通过调节神经可塑性来改善认知记忆方面显示出了巨大的潜力,但是神经干细胞临床转化却存在一些阻力。赵岚等发现神经干细胞来源的细胞外囊泡可缓解病理特征,包括淀粉样蛋白β和神经炎症,并促进神经可塑性,缓解阿尔茨海默病认知缺陷,且无明显不良反应[26-28]。神经营养因子可促进神经干细胞的生存、增殖和分化能力,提高突触和树突棘密度及可塑性相关分子表达,抑制神经炎症,并促进受损组织的内源性修复[29-31]。胆碱乙酰转移酶或中性内肽酶能促进海马和皮质中神经干细胞迁移、存活和分化能力,逆转神经递质和神经营养因子的不足,并减少淀粉样蛋白β和神经炎症,进而改善学习和记忆能力[4, 32]。#br#
最后,临床前研究已证实神经干细胞治疗阿尔茨海默病的安全性和有效性,但目前临床研究依然较少。临床前研究发现,鼻内移植人神经干细胞可减轻APP/PS1小鼠的认知障碍,且无明显不良反应[24]。这表明鼻内给药人神经干细胞是一种非侵入性的阿尔茨海默病治疗策略。此外,从已发表的研究结果来看,神经干细胞治疗阿尔茨海默病的临床效果是有限的。这可能与一些因素密切相关。首先,最佳给药途径的选择似乎是关键。不同的给药途径,如经脑室内、鼻腔和静脉途径,都会影响干细胞在大脑中的存活和分化[7]。其次,尽管神经干细胞治疗阿尔茨海默病是可行的,但其安全性需要不断验证和完善,在转化为临床试验前仍需开发安全策略。最后,神经干细胞的制备质量也需要进行更深入的研究,应从细胞制备中的酶、蛋白质和基因等多个层面进行探讨。
随着年龄的增长,神经可塑性受损越来越严重,这是早期预测及诊断阿尔茨海默病的一个有效标志(图1)。早期阿尔茨海默病神经可塑性已经受损,所以检测衰老和阿尔茨海默病的神经可塑性是否受损,有利于提高阿尔茨海默病的早期诊断率。神经干细胞具有自我更新、多向分化、免疫原性低、归巢能力强以及免疫调节和旁分泌功能等优点。神经干细胞通过旁分泌作用分泌可塑性相关分子,促进神经发生、突触发生(表1)。重要的是,神经干细胞移植可从根本上逆转阿尔茨海默病神经元和突触的丢失。此外,神经干细胞还可以减轻阿尔茨海默病的典型病理变化,如淀粉样蛋白β和tau及神经炎症。因此,神经干细胞调节神经可塑性可能是一种有前途的阿尔茨海默病再生疗法。
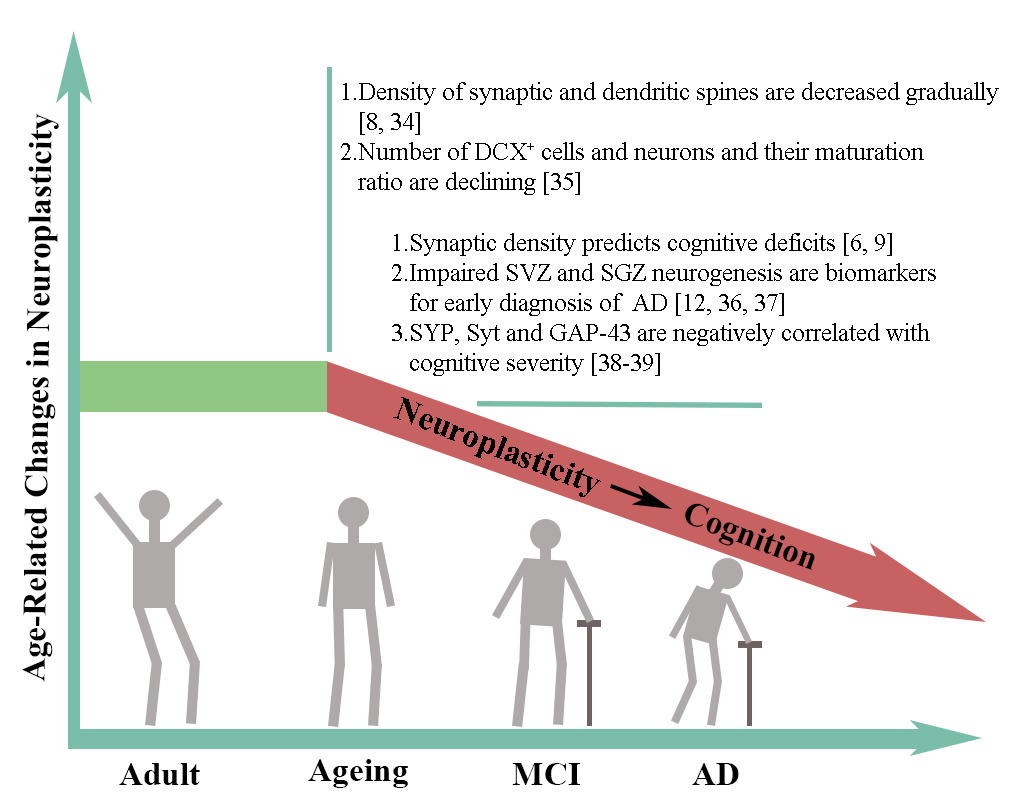
图1 与年龄有关的神经可塑性的变化(图源:Chang et al., Neural Regen Res, 2024)
临床前研究显示神经干细胞疗法是安全有效的,但许多临床试验却失败了。这表明在基础实验临床转化前,仍有许多障碍需要克服,如安全性、细胞来源和最佳治疗时间和给药途径。尽管存在这些障碍,赵岚等仍建议加快神经干细胞的转化研究,这将有利于研究阿尔茨海默病的病因和潜在的治疗方法。神经干细胞来源的细胞外囊泡、神经营养因子调节以及神经干细胞基因修饰可逆转神经元和突触的缺失,缓解阿尔茨海默病病理表现,这将为阿尔茨海默病患者提供安全的、具有临床转化意义的疗法。相信在不远的将来,随着科学技术的不断进步,神经干细胞将成为一种真正有益的临床疗法,为阿尔茨海默病患者带来福音。
原文链接:https://doi.org/10.4103/1673-5374.380874
参考文献
[1] Terreros-Roncal J, Moreno-Jiménez EP, Flor-García M, et al. Impact of neurodegenerative diseases on human adult hippocampal neurogenesis. Science. 2021;374(6571):1106-1113.
[2] Huang D, Cao Y, Yang X, et al. A nanoformulation-mediated multifunctional stem cell therapy with improved beta-amyloid clearance and neural regeneration for Alzheimer's disease. Adv Mater. 2021;33(13):e2006357.
[3] Livingston G, Huntley J, Sommerlad A, et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet. 2020;396(10248):413-446.
[4] Park D, Choi EK, Cho TH, et al. Human neural stem cells encoding ChAT gene restore cognitive function via acetylcholine synthesis, Aβ elimination, and neuroregeneration in APPswe/PS1dE9 mice. Int J Mol Sci. 2020;21(11):3958.
[5] Zhao L, Liu JW, Shi HY, et al. Neural stem cell therapy for brain disease. World J Stem Cells. 2021;13(9):1278-1292.
[6] Colom-Cadena M, Spires-Jones T, Zetterberg H, et al. The clinical promise of biomarkers of synapse damage or loss in Alzheimer's disease. Alzheimers Res Ther. 2020;12(1):21.
[7] Mango D, Saidi A, Cisale GY, et al. Targeting synaptic plasticity in experimental models of Alzheimer's disease. Front Pharmacol. 2019;10:778.
[8] Montero-Crespo M, Domínguez-Álvaro M, Alonso-Nanclares L, et al. Three-dimensional analysis of synaptic organization in the hippocampal CA1 field in Alzheimer's disease. Brain. 2021;144(2):553-573.
[9] Mecca AP, O'dell RS, Sharp ES, et al. Synaptic density and cognitive performance in Alzheimer's disease: A PET imaging study with [(11) C]UCB-J. Alzheimers Dement. 2022;18(12):2527-2536.
[10] Tobin MK, Musaraca K, Disouky A, et al. Human hippocampal neurogenesis persists in aged adults and Alzheimer's disease patients. Cell Stem Cell. 2019;24:974-982.e3(6).
[11] Toda T, Parylak SL, Linker SB, et al. The role of adult hippocampal neurogenesis in brain health and disease. Mol Psychiatry. 2019;24(1):67-87.
[12] Scopa C, Marrocco F, Latina V, et al. Impaired adult neurogenesis is an early event in Alzheimer's disease neurodegeneration, mediated by intracellular Aβ oligomers. Cell Death Differ. 2020;27(3):934-948.
[13] Mckay R. Stem cells in the central nervous system. Science. 1997;276(5309):66-71.
[14] Ma H, Yu B, Kong L, et al. Neural stem cells over-expressing brain-derived neurotrophic factor (BDNF) stimulate synaptic protein expression and promote functional recovery following transplantation in rat model of traumatic brain injury. Neurochem Res. 2012;37(1):69-83.
[15] Wang FW, Hao HB, Zhao SD, et al. Roles of activated astrocyte in neural stem cell proliferation and differentiation. Stem Cell Res. 2011;7(1):41-53.
[16] Ganat YM, Calder EL, Kriks S, et al. Identification of embryonic stem cell-derived midbrain dopaminergic neurons for engraftment. J Clin Invest. 2012;122(8):2928-2939.
[17] Navarro Negredo P, Yeo RW, Brunet A. Aging and rejuvenation of neural stem cells and their niches. Cell Stem Cell. 2020;27(2):202-223.
[18] Hu NW, Corbett GT, Moore S, et al. Extracellular Forms of Aβ and Tau from iPSC Models of Alzheimer's Disease Disrupt Synaptic Plasticity. Cell Rep. 2018;23(7):1932-1938.
[19] Micci MA, Krishnan B, Bishop E, et al. Hippocampal stem cells promotes synaptic resistance to the dysfunctional impact of amyloid beta oligomers via secreted exosomes. Mol Neurodegener. 2019;14(1):25.
[20] Xiong LL, Hu Y, Zhang P, et al. Neural stem cell transplantation promotes functional recovery from traumatic brain injury via brain derived neurotrophic factor-mediated neuroplasticity. Mol Neurobiol. 2018;55(3):2696-2711.
[21] Blurton-Jones M, Spencer B, Michael S, et al. Neural stem cells genetically-modified to express neprilysin reduce pathology in Alzheimer transgenic models. Stem Cell Res Ther. 2014;5(2):46.
[22] De Gioia R, Biella F, Citterio G, et al. Neural stem cell transplantation for neurodegenerative diseases. Int J Mol Sci. 2020;21(9):3103.
[23] Zhao L, Liu JW, Kan BH, et al. Acupuncture accelerates neural regeneration and synaptophysin production after neural stem cells transplantation in mice. World J Stem Cells. 2020;12(12):1576-1590.
[24] Lu MH, Ji WL, Chen H, et al. Intranasal transplantation of human neural stem cells ameliorates Alzheimer's disease-like pathology in a mouse model. Front Aging Neurosci. 2021;13:650103.
[25] Shu H, Guo Z, Chen X, et al. Intracerebral transplantation of neural stem cells restores manganese-induced cognitive deficits in mice. Aging Dis. 2021;12(2):371-385.
[26] Spinelli M, Natale F, Rinaudo M, et al. Neural stem cell-derived exosomes revert HFD-Dependent memory impairment via CREB-BDNF signalling. Int J Mol Sci. 2020;21(23):8994.
[27] Li B, Liu J, Gu G, et al. Impact of neural stem cell-derived extracellular vesicles on mitochondrial dysfunction, sirtuin 1 level, and synaptic deficits in Alzheimer's disease. J Neurochem. 2020;154(5):502-518.
[28] Apodaca LA, Baddour AaD, Garcia C, Jr., et al. Human neural stem cell-derived extracellular vesicles mitigate hallmarks of Alzheimer's disease. Alzheimers Res Ther. 2021;13(1):57.
[29] Lunn JS, Pacut C, Backus C, et al. The pleotrophic effects of insulin-like growth factor-I on human spinal cord neural progenitor cells. Stem Cells Dev. 2010;19(12):1983-1993.
[30] Yan YH, Li SH, Gao Z, et al. Neurotrophin-3 promotes proliferation and cholinergic neuronal differentiation of bone marrow- derived neural stem cells via notch signaling pathway. Life Sci. 2016;166:131-138.
[31] Mcginley LM, Sims E, Lunn JS, et al. Human cortical neural stem cells expressing insulin-like growth factor-I: a novel cellular therapy for Alzheimer's disease. Stem Cells Transl Med. 2016;5(3):379-391.
[32] Park D, Yang YH, Bae DK, et al. Improvement of cognitive function and physical activity of aging mice by human neural stem cells over-expressing choline acetyltransferase. Neurobiol Aging. 2013;34(11):2639-2646.
[33] Berger T, Lee H, Young AH, et al. Adult hippocampal neurogenesis in major depressive disorder and Alzheimer's disease. Trends Mol Med. 2020;26(9):803-818.
[34] Moreno-Jiménez EP, Terreros-Roncal J, Flor-García M, et al. Evidences for Adult Hippocampal Neurogenesis in Humans. J Neurosci. 2021;41(12):2541-2553.
[35] Hayashi Y, Lin HT, Lee CC, et al. Effects of neural stem cell transplantation in Alzheimer's disease models. J Biomed Sci. 2020;27(1):29.
[36] Zhang T, Ke W, Zhou X, et al. Human neural stem cells reinforce hippocampal synaptic network and rescue cognitive deficits in a mouse model of Alzheimer's disease. Stem Cell Reports. 2019;13(6):1022-1037.
[37] Zhang W, Wang PJ, Sha HY, et al. Neural stem cell transplants improve cognitive function without altering amyloid pathology in an APP/PS1 double transgenic model of Alzheimer's disease. Mol Neurobiol. 2014;50(2):423-437.
[38] Gómez-Isla T, Frosch MP. Lesions without symptoms: understanding resilience to Alzheimer disease neuropathological changes. Nat Rev Neurol. 2022;18(6):323-332.
[39] Kim JA, Ha S, Shin KY, et al. Neural stem cell transplantation at critical period improves learning and memory through restoring synaptic impairment in Alzheimer's disease mouse model. Cell Death Dis. 2015;6(6):e1789.
[40] Zhang HA, Yuan CX, Liu KF, et al. Neural stem cell transplantation alleviates functional cognitive deficits in a mouse model of tauopathy. Neural Regen Res. 2022;17(1):152-162.
[41] Wang S, Yao H, Xu Y, et al. Therapeutic potential of a TrkB agonistic antibody for Alzheimer's disease. Theranostics. 2020;10(15):6854-6874.
[42] Babcock KR, Page JS, Fallon JR, et al. Adult hippocampal neurogenesis in aging and Alzheimer's disease. Stem Cell Reports. 2021;16(4):681-693.
[43] Wu CC, Lien CC, Hou WH, et al. Gain of BDNF function in engrafted neural stem cells promotes the therapeutic potential for Alzheimer's disease. Sci Rep. 2016;6:27358.
[44] Yamasaki TR, Blurton-Jones M, Morrissette DA, et al. Neural stem cells improve memory in an inducible mouse model of neuronal loss. J Neurosci. 2007;27(44):11925-11933.
[45] Zhang W, Wang GM, Wang PJ, et al. Effects of neural stem cells on synaptic proteins and memory in a mouse model of Alzheimer's disease. J Neurosci Res. 2014;92(2):185-194.
[46] Walker CK, Herskowitz JH. Dendritic spines: mediators of cognitive resilience in aging and Alzheimer's disease. Neuroscientist. 2021;27(5):487-505.
[47] Liu XY, Yang LP, Zhao L. Stem cell therapy for Alzheimer's disease. World J Stem Cells. 2020;12(8):787-802.
[48] Zhao L, Zhou C, Li L, et al. Acupuncture improves cerebral microenvironment in mice with Alzheimer's disease treated with hippocampal neural stem cells. Mol Neurobiol. 2017;54(7):5120-5130.
[49] Ager RR, Davis JL, Agazaryan A, et al. Human neural stem cells improve cognition and promote synaptic growth in two complementary transgenic models of Alzheimer's disease and neuronal loss. Hippocampus. 2015;25(7):813-826.
[50] Kizil C, Bhattarai P. Is Alzheimer's also a stem cell disease? - The zebrafish perspective. Front Cell Dev Biol. 2018;6:159.
[51] Lu HX, Hao ZM, Jiao Q, et al. Neurotrophin-3 gene transduction of mouse neural stem cells promotes proliferation and neuronal differentiation in organotypic hippocampal slice cultures. Med Sci Monit. 2011;17(11):BR305-311.
[52] Wu L, Sluiter AA, Guo HF, et al. Neural stem cells improve neuronal survival in cultured postmortem brain tissue from aged and Alzheimer patients. J Cell Mol Med. 2008;12(5a):1611-1621.
[53] Duncan T, Valenzuela M. Alzheimer's disease, dementia, and stem cell therapy. Stem Cell Res Ther. 2017;8(1):111.
[54] Boldrini M, Fulmore CA, Tartt AN, et al. Human hippocampal neurogenesis persists throughout aging. Cell Stem Cell. 2018;22:589-599.e5(4).
[55] Baglietto-Vargas D, Sánchez-Mejias E, Navarro V, et al. Dual roles of Aβ in proliferative processes in an amyloidogenic model of Alzheimer's disease. Sci Rep. 2017;7(1):10085.
[56] Yang J, Yan Y, Xia Y, et al. Neurotrophin 3 transduction augments remyelinating and immunomodulatory capacity of neural stem cells. Mol Ther. 2014;22(2):440-450.
[57] Li Puma DD, Piacentini R, Grassi C. Does Impairment of adult neurogenesis contribute to pathophysiology of Alzheimer's disease? A still open question. Front Mol Neurosci. 2020;13:578211.
[58] Moreno-Jiménez EP, Flor-García M, Terreros-Roncal J, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer's disease. Nat Med. 2019;25(4):554-560.
[59] Zhou Y, Su Y, Li S, et al. Molecular landscapes of human hippocampal immature neurons across lifespan. Nature. 2022;607(7919):527-533.
[60] Bustos FJ, Ampuero E, Jury N, et al. Epigenetic editing of the Dlg4/PSD95 gene improves cognition in aged and Alzheimer's disease mice. Brain. 2017;140(12):3252-3268.
[61] Martin SB, Dowling AL, Lianekhammy J, et al. Synaptophysin and synaptojanin-1 in Down syndrome are differentially affected by Alzheimer's disease. J Alzheimers Dis. 2014;42(3):767-775.
[62] Masliah E, Mallory M, Alford M, et al. Altered expression of synaptic proteins occurs early during progression of Alzheimer's disease. Neurology. 2001;56(1):127-129.
[63] Zhou CL, Zhao L, Shi HY, et al. Combined acupuncture and HuangDiSan treatment affects behavior and synaptophysin levels in the hippocampus of senescence-accelerated mouse prone 8 after neural stem cell transplantation. Neural Regen Res. 2018;13(3):541-548.
[64] Park D, Lee HJ, Joo SS, et al. Human neural stem cells over-expressing choline acetyltransferase restore cognition in rat model of cognitive dysfunction. Exp Neurol. 2012;234(2):521-526.
[65] Gao L, Zhang Y, Sterling K, et al. Brain-derived neurotrophic factor in Alzheimer's disease and its pharmaceutical potential. Transl Neurodegener. 2022;11(1):4.
[66] Wu Z, Chen C, Kang SS, et al. Neurotrophic signaling deficiency exacerbates environmental risks for Alzheimer's disease pathogenesis. Proc Natl Acad Sci U S A. 2021;118(25):e2100986118.
[67] Blurton-Jones M, Kitazawa M, Martinez-Coria H, et al. Neural stem cells improve cognition via BDNF in a transgenic model of Alzheimer disease. Proc Natl Acad Sci U S A. 2009;106(32):13594-13599.
赵岚,博士,研究员,博士研究生导师。主要从事针刺治疗老年性疾病、脑病的机制研究。本论文获得国家自然科学基金(82074533)的支持。主持课题7项,参与课题11项;发表论文62篇,其中SCI收录23篇;获得科研成果7项;获得科研奖项2项;参编专著1项。2016年度天津市“131”创新型人才培养工程二层次人才。2017年“十三五”期间天津市高校“中青年骨干创新人才”。
 #br#
#br#
常军,2022级天津中医药大学第一附属医院中西医结合临床专业博士,主要从事针刺治疗脑病的机制研究。第一作者书写文章5篇,中文文章3篇,英文文章2篇;申请专利1项。于2020年获天津中医药大学优秀共青团员,2021年获天津中医药大学优秀共青干部,2022年获天津中医药大学优秀毕业研究生。

 #br#
#br#