NRR:中国重庆医科大学附属第一医院何朝晖团队揭示磷酸二酯酶4在蛛网膜下腔出血中的新作用机制
#br#
蛛网膜下腔出血(Subarachnoid hemorrhage, SAH)是临床上常见且严重的脑血管疾病,以其极高的致残致死率严重威胁人类生命健康[1]。早期脑损伤(Early brain injury, EBI)是指在蛛网膜下腔出血后72h内大脑发生的一系列直接损伤,已被证实为蛛网膜下腔出血预后不良的重要因素[2,3]。细胞焦亡是一种程序性细胞死亡,表现为细胞不断胀大直至细胞膜破裂,最终导致细胞内容物释放进而激活强烈的炎症反应[4]。NLRP3炎症体是调控细胞焦亡经典途径的关键分子[5]。近年来,越来越多的研究表明神经元的焦亡在早期脑损伤的病理过程中发挥了重要作用,但相关机制未完全阐明。磷酸二酯酶4(Phosphodiesterase 4,PDE4)是一种高亲和力的酶,能特异性水解cAMP引起其浓度变化进而参与下游级联反应,已被证实与多种疾病密切相关[6,7]。既往已有研究表明磷酸二酯酶4参与了蛛网膜下腔出血后早期脑损伤,但是潜在机制尚未完全了解。且目前尚无研究证实磷酸二酯酶4与NLRP3或神经元焦亡之间有无调控关系及相关机制[8]。
中国重庆医科大学附属第一医院的何朝晖团队在《中国神经再生研究(英文)》(Neural Regeneration Research)上发表的研究发现,磷酸二酯酶4通过激活NF-κB通路,并同时引起溶酶体和线粒体功能障碍,三者共同促进NLRP3的激活诱发神经元焦亡参与蛛网膜下腔出血后早期脑损伤。抑制磷酸二酯酶4可通过减轻神经元焦亡改善早期脑损伤。激动NLRP3可以逆转抑制磷酸二酯酶4产生的神经保护作用。研究为蛛网膜下腔出血的治疗提供新的潜在靶点磷酸二酯酶4。谭嘉禾和马寅瑞为论文共同第一作者,何朝晖教授为论文通讯作者。
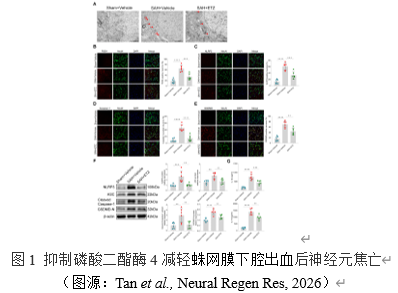
该研究首先是对焦亡表型的探讨。焦亡的特征之一是细胞膜上孔洞形成,因此采用透射电镜观察神经元细胞膜的相关变化[4]。研究结果显示,蛛网膜下腔出血后神经元细胞膜上有明显孔洞形成,而抑制磷酸二酯酶4减轻了这一现象。免疫荧光检测磷酸二酯酶4与焦亡相关分子NLRP3,Caspase-1,GSDMD的表达和定位。结果表明,与假手术组相比,蛛网膜下腔出血组神经元中磷酸二酯酶4和这些焦亡相关分子表达明显增加,抑制磷酸二酯酶4则逆转了这些趋势。此外,与假手术组相比,蛛网膜下腔出血组的焦亡相关分子NLRP3,ASC,cleaved Caspase -1,GSDMD-N的蛋白水平明显升高。与免疫荧光结果一致,抑制磷酸二酯酶4有效地减轻了这些现象。ELISA检测焦亡相关炎症因子IL-1β和IL-18的分泌水平。与蛛网膜下腔出血组相比,抑制磷酸二酯酶4有效地降低了这些炎症因子水平。这些结果证实了抑制磷酸二酯酶4可以减轻蛛网膜下腔出血后神经元的焦亡。
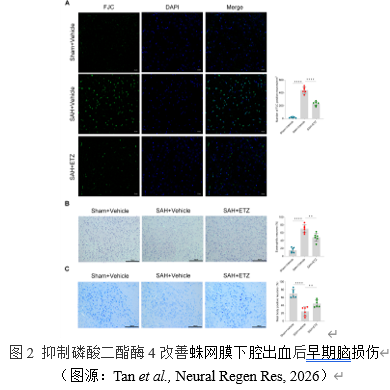
早期脑损伤通常伴随着神经元功能和活性的下降。FJC染色检测神经元变性,测定神经元功能。抑制磷酸二酯酶4显著降低了每平方毫米FJC阳性神经元的数量,即改善了蛛网膜下腔出血小鼠神经元的变性。HE染色、尼氏染色检测神经元活性。与蛛网膜下腔出血组相比,抑制磷酸二酯酶4明显降低嗜酸性神经元比例,而显著增加Nissl体阳性神经元比例。这些结果证实抑制磷酸二酯酶4可恢复神经元功能和活性,改善早期脑损伤。
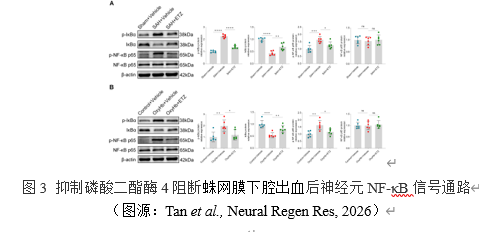
NF-κB信号通路对于NLRP3炎性体的激活至关重要[9]。为了明确抑制磷酸二酯酶4是否会影响蛛网膜下腔出血蛛网膜下腔出血后的NF-κB信号传导,实验检测了小鼠和原代神经元相关蛋白的表达水平。结果提示,在蛛网膜下腔出血体内和体外模型中,抑制磷酸二酯酶4可以显著降低IκBα的磷酸化水平,升高其总含量,同时可以显著降低NF-κB p65磷酸化水平,维持其总量不变。这些发现表明,抑制磷酸二酯酶4可以阻断SAH后神经元NF-κB信号通路。
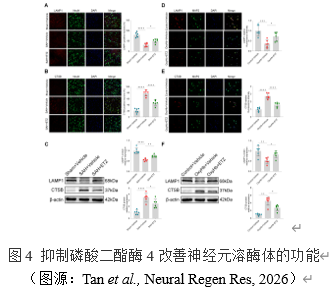
溶酶体功能障碍可诱导NLRP3炎症体的激活[10]。溶酶体功能障碍通常表现为溶酶体膜通透性增强或溶酶体破裂,这都将导致溶酶体膜标志物LAMP1表达下降,溶酶体内容物如CTSB释放入胞质并激活。为此,实验检测了小鼠脑皮质神经元和原代神经元中LAMP1和CTSB的表达和定位。结果提示在SAH体内和体外模型中,抑制磷酸二酯酶4可以显著升高LAMP1表达水平并显著降低CTSB表达水平。这些结果表明,抑制磷酸二酯酶4可通过稳定溶酶体膜或抑制溶酶体破裂来减少CTSB的释放并改善溶酶体的功能。
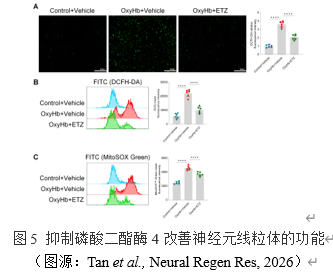
线粒体功能障碍可通过生成过量ROS诱导NLRP3炎症体的激活[11]。通过DCFH-DA荧光探针标记ROS,然后进行免疫荧光和流式细胞术检测。抑制磷酸二酯酶4降低了蛛网膜下腔出血原代神经元中的ROS水平。然而,以上测量的ROS是细胞内ROS总量,包含多个来源。因此,采用MitoSOX线粒体超氧化物检测线粒体特异性ROS,使结果更加严谨。与上述结果一致的是,蛛网膜下腔出血原代神经元线粒体特异性ROS水平明显升高,而抑制磷酸二酯酶4可逆转这一现象。这些结果证明,抑制磷酸二酯酶4可以改善神经元线粒体功能,减少ROS的生成。
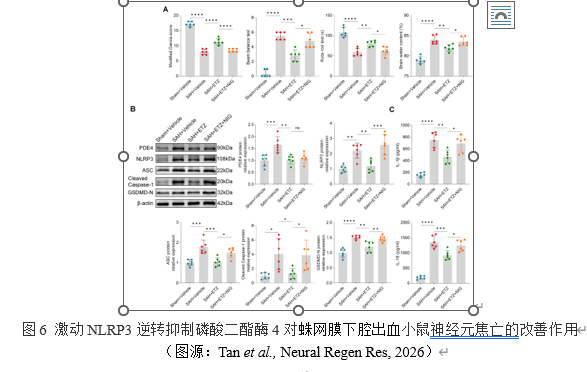
最后,为了明确磷酸二酯酶4是否通过调节NLRP3炎症体介导早期脑损伤和神经元焦亡,以及磷酸二酯酶4与NLRP3在信号通路中的关系,实验给予了NLRP3特异性激动剂NIG。神经行为学实验和脑含水量测量结果显示,激动NLRP3逆转了抑制磷酸二酯酶4对蛛网膜下腔出血小鼠神经功能障碍和脑水肿的改善作用,以及对焦亡相关蛋白和炎症因子水平降低的作用。但并不影响磷酸二酯酶4的表达。这些发现证明,激动NLRP3可以逆转抑制磷酸二酯酶4对蛛网膜下腔出血小鼠神经元焦亡的改善作用,NLRP3在信号通路中位于磷酸二酯酶4的下游。
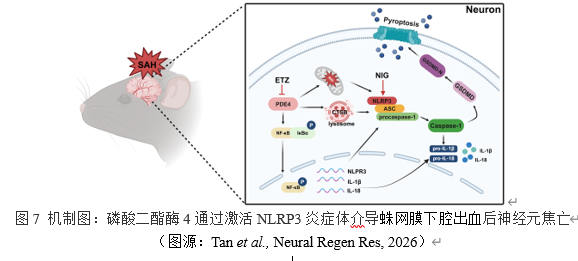
综上所述,本研究明确磷酸二酯酶4通过激活NF-κB通路,并同时引起溶酶体和线粒体功能障碍,三者共同促进NLRP3的激活诱发神经元焦亡参与蛛网膜下腔出血后早期脑损伤。抑制磷酸二酯酶4可通过减轻神经元焦亡改善早期脑损伤。激动NLRP3可以逆转抑制磷酸二酯酶4产生的神经保护作用。研究为蛛网膜下腔出血的治疗提供新的潜在靶点磷酸二酯酶4。
当然该研究也存在一定性局限性。本研究中使用的动物均为雄性,因为雌性小鼠有周期性的激素波动,会显著影响神经科学实验的结果。但单性别实验不可避免地会导致结果出现一定程度的偏差。未来,需要在神经科学相关实验中开发更科学的动物性别管理方法来指导临床前研究。磷酸二酯酶4参与神经元焦亡的机制尚未深入探讨,实验仅在下游通路中调控了NLRP3,未对NF-κB通路、溶酶体、线粒体功能进行调控。这些都值得今后进一步研究。虽然相关药物可以显著抑制磷酸二酯酶4的表达,但仍需要磷酸二酯酶4基因敲除动物模型来进一步验证这一发现。#br#
#br#
参考文献#br#
[1]Macdonald RL, Schweizer T A. Spontaneous subarachnoid haemorrhage. Lancet. 2017;389:655-666.#br#
[2]Cahill J, Calvert JW, Zhang JH. Mechanisms of early brain injury after subarachnoid hemorrhage. J Cereb Blood Flow Metab. 2006;26:1341-1353.#br#
[3] Rass V, Helbok R. Early brain injury after poor-grade subarachnoid hemorrhage. Curr Neurol Neurosci Rep. 2019;19:78.#br#
[4]Yu P, Zhang X, Liu N, et al. Pyroptosis: mechanisms and diseases. Signal Transduct Target Ther. 2021;6:128.#br#
[5] Coll RC, Schroder K, Pelegrín P. NLRP3 and pyroptosis blockers for treating inflammatory diseases. Trends Pharmacol Sci. 2022;43:653-668.#br#
[6] Fertig BA, Baillie GS. PDE4-mediated cAMP signalling. J Cardiovasc Dev Dis. 2018;5:8.#br#
[7] Gancedo JM. Biological roles of cAMP: variations on a theme in the different kingdoms of life. Biol Rev Camb Philos Soc. 2013;88:645-668.#br#
[8] Li Q, Peng Y, Fan L, et al. Phosphodiesterase-4 inhibition confers a neuroprotective efficacy against early brain injury following experimental subarachnoid hemorrhage in rats by attenuating neuronal apoptosis through the SIRT1/Akt pathway. Biomed Pharmacother. 2018;99:947-955.#br#
[9] Lamkanfi M, Dixit VM. Mechanisms and functions of inflammasomes. Cell. 2014;157:1013-1022.#br#
[10] Yang Y, Wang H, Kouadir M, Song H, Shi F.Recent advances in the mechanisms of NLRP3 inflammasome activation and its inhibitors. Cell Death Dis. 2019;10:128.#br#
[11] Swanson KV, Deng M, Ting JP. The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat Rev Immunol. 2019;19:477-489.#br#






