中国神经再生研究(英文版) ›› 2026, Vol. 21 ›› Issue (6): 2621-2631.doi: 10.4103/NRR.NRR-D-24-01400
延迟小胶质细胞耗竭减轻新生小鼠脑出血后白质损伤
Delayed microglial depletion protects against white matter injury following neonatal cerebral hemorrhage in mice
Xiaoxiao Jing1, 2, #, Xiaoli Zhang2, #, Hongwei Li2, 3, 4, #, Yu Yang2, Zuhang Zhao1, Yuandan Li1, 2, Jinjin Zhu1, 2, Yiran Xu2, Jing Yuan2, Tiantian He2, Chen Zhang1, 2, Juan Song1, Xin Zhao5, Xiaoyang Wang2, 6, Changlian Zhu2, 7, *, Falin Xu1, 2, *
- 1Department of Neonatology, The Third Affiliated Hospital of Zhengzhou University, Zhengzhou, Henan Province, China;
2Henan Key Laboratory of Child Brain Injury and Henan Pediatric Clinical Research Center, Institute of Neuroscience and Third Affiliated Hospital of Zhengzhou University, Zhengzhou, Henan Province, China;
3Department of Laboratory Medicine, Third Affiliated Hospital of Zhengzhou University, Zhengzhou, Henan Province, China;
4Zhengzhou Key Laboratory for In Vitro Diagnosis of Hypertensive Disorders of Pregnancy, Zhengzhou, Henan Province, China;
5Department of Imaging, The Third Affiliated Hospital of Zhengzhou University, Zhengzhou, Henan Province, China;
6Center for Perinatal Medicine and Health, Institute of Clinical Science, University of Gothenburg, Gothenburg, Sweden;
7Center for Bran Repair and Rehabilitation, Institute of Neuroscience and Physiology, University of Gothenburg, Gothenburg, Sweden
摘要:
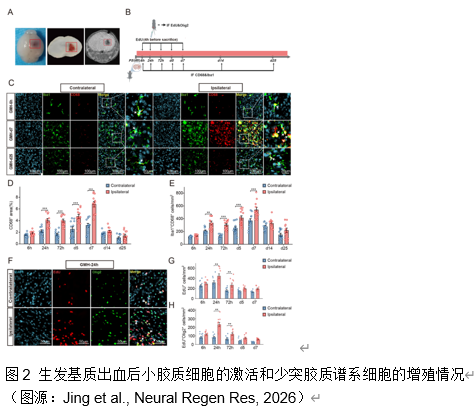
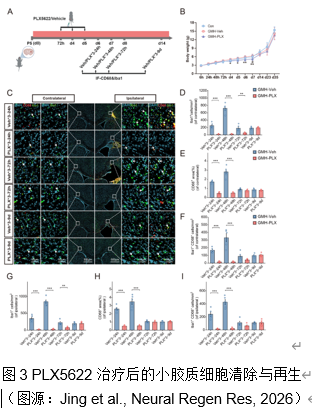
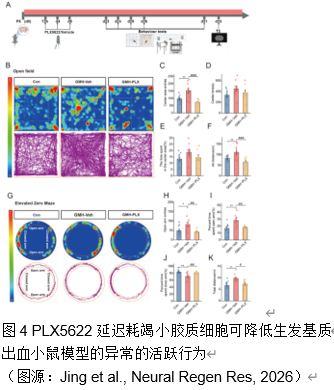
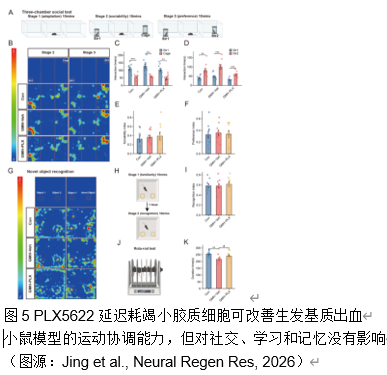
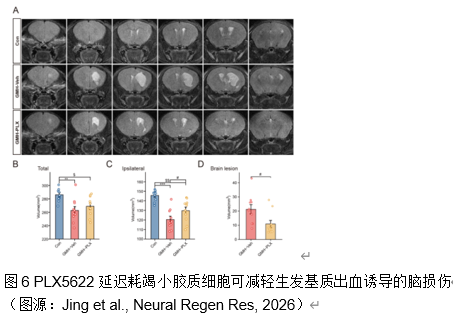
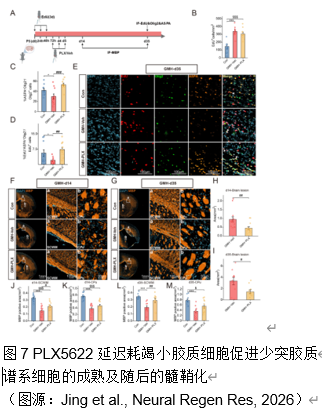
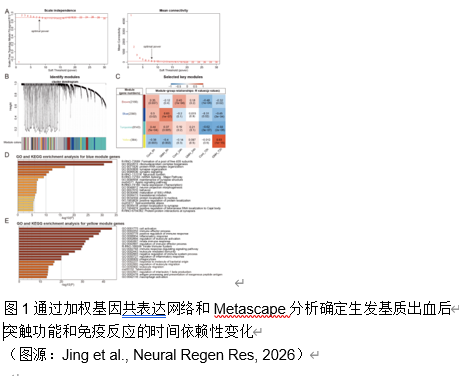
早产儿脑生发基质出血常会导致脑白质损伤。小胶质细胞作为脑内常驻的免疫细胞,在调节神经炎症及修复过程中发挥着复杂而重要的作用。集落刺激因子1受体抑制剂PLX5622,能够选择性地耗竭小胶质细胞,不过其在早产儿脑生发基质出血中的治疗潜力仍未得到充分探索。实验在出生后第 5 天的小鼠中使用胶原酶诱导脑生发基质出血模型。在脑生发基质出血 后 72 小时腹腔注射 PLX5622,以在损伤反应高峰期实现有针对性的、暂时性的小胶质细胞耗竭。实验评估了这种延迟干预对少突胶质谱系细胞成熟、白质完整性和神经行为结果的影响;还分析了脑生发基质出血大鼠模型的RNA测序数据,通过加权基因共表达网络分析确定干预的关键阶段。RNA测序数据揭示了脑生发基质出血后关键突触功能下降而免疫反应增强的关键时期,从而确定了潜在干预措施的关键反应阶段。延迟PLX5622治疗可有效清除活化的小胶质细胞,防止白质损伤,并增强皮层下白质区域的少突胶质谱系细胞成熟和髓鞘化。磁共振成像分析显示,接受治疗的小鼠脑损伤体积缩小。在行为上,与未接受治疗的小鼠相比,接受PLX5622治疗的小鼠在运动协调性和多动性方面都有显著改善。结果表明,用PLX5622对小胶质细胞进行靶向清除,以避免干扰最初的少突胶质谱系细胞增殖,可显著减轻新生儿脑生发基质出血的白质损伤并改善神经行为。这项研究强调了选择性调节小胶质细胞活化以支持脑损伤早产儿神经发育的治疗潜力。
https://orcid.org/0000-0002-5029-6730 (Changlian Zhu); https://orcid.org/0000-0003-1910-112X (Falin Xu)
 #br#
#br#