中国神经再生研究(英文版) ›› 2026, Vol. 21 ›› Issue (8): 3629-3640.doi: 10.4103/NRR.NRR-D-24-00968
肿瘤坏死因子α刺激基因6促进脑出血后血肿清除的作用及机制
Tumor necrosis factor-α–stimulated gene 6 promotes hematoma clearance after intracerebral hemorrhage in a mouse model
Xia Liu1, 2, 3, #, Dabao Yao1, 3, #, Yunjie Li1, 3, Shiling Chen1, 3, Yingxin Tang1, 3, Jingyi Wang1, 3, Jingfei Yang3, 4, Jie Jing4, 5, Jiahui Wang1, 3,#br# Ge Zhang1, 3, Luwei Nie1, 3, Yangyang Feng1, 3, Gaigai Li1, 3, *, Zhouping Tang1, 3, *#br#
- 1Department of Neurology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei Province, China;
2Department of Neurology, Jingzhou Hospital, Yangtze University, Jingzhou, Hubei Province, China;
3Hubei Key Laboratory of Neural Injury and Functional Reconstruction, Huazhong University of Science and Technology, Wuhan, Hubei Province, China;
4Department of Nuclear Medicine, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei Province, China;
5Department of Neurology, Qilu Hospital, Shandong University, Jinan, Shandong Province, China
摘要:
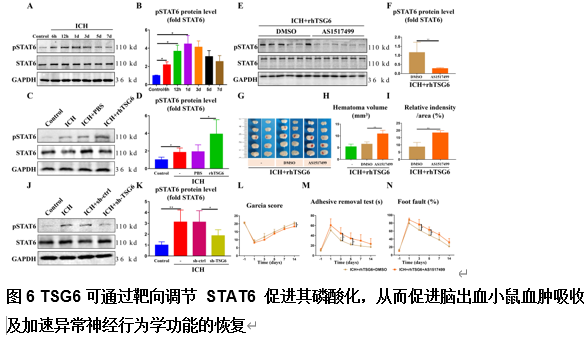
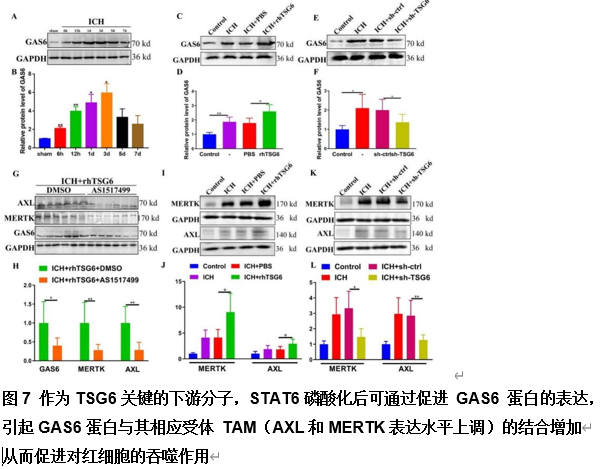
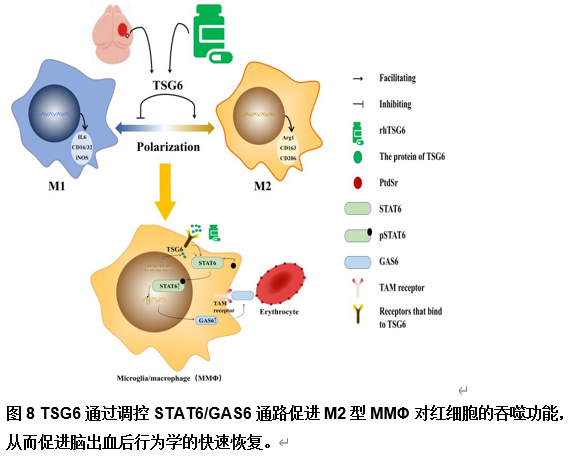
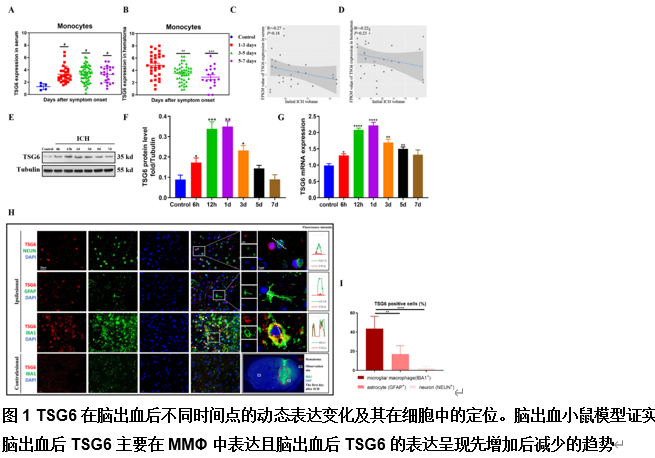
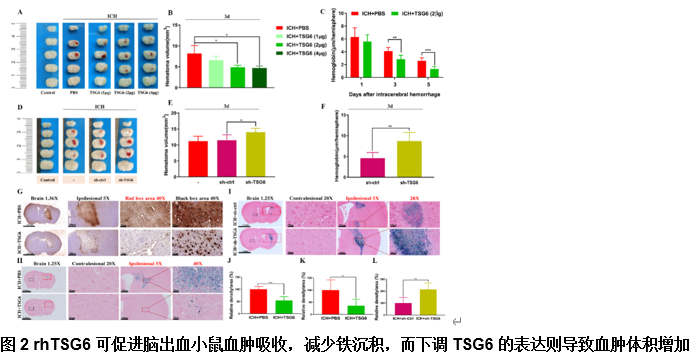
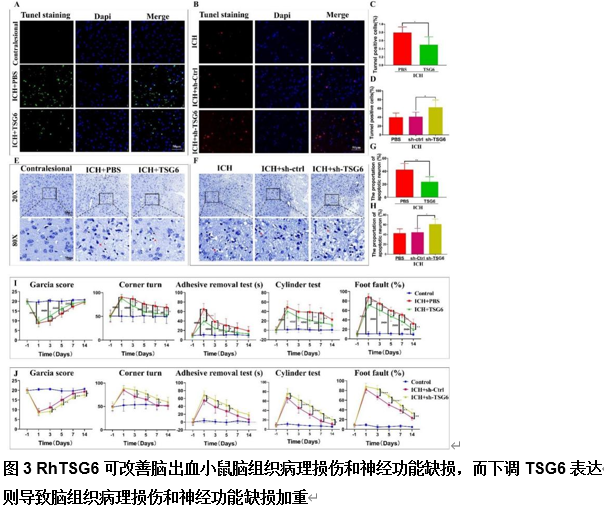
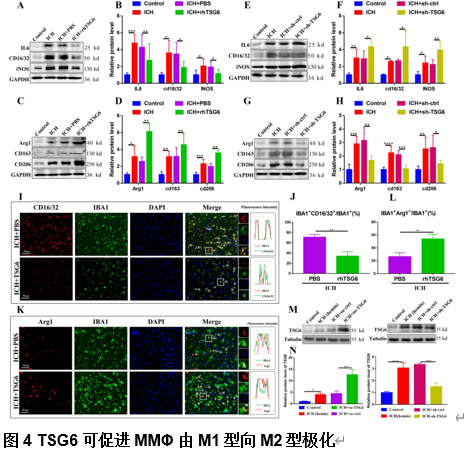
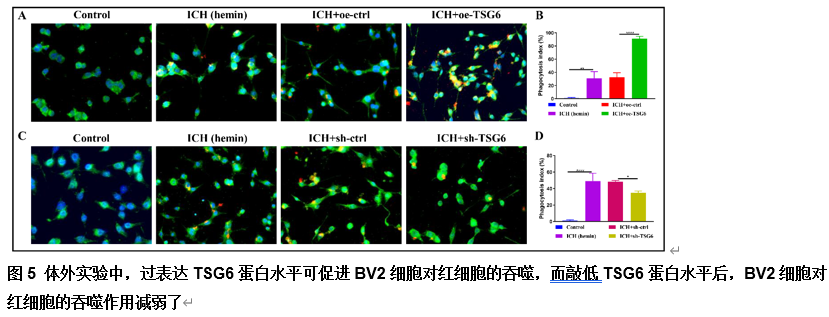
由于脑出血缺乏有效的治疗方法,其患者的预后较差。肿瘤坏死因子α刺激基因6(TSG6)是一种分泌型糖蛋白,可参与多种炎症性疾病的病理过程,并发挥抗炎作用。作者既往研究已发现,在体外脑出血模型中,脂肪来源的干细胞可通过上调TSG6蛋白来抑制炎症。但TSG6对体内血肿清除的直接作用机制仍未得到完全理解。实验探索了TSG6影响脑出血小鼠血肿吸收的潜在机制。实验首先分析了GEO数据库中脑出血患者的基因谱,并检测了脑出血小鼠脑组织中TSP6表达的变化,可见脑出血后TSG6表达呈一过性升高,且初始血肿体积与TSG6水平呈负相关。继而免疫荧光分析结果显示,TSG6主要在小胶质细胞/巨噬细胞中表达。随后发现TSG6可通过加速血肿清除、减少凋亡细胞和退化神经元、增加吞噬小胶质细胞/巨噬细胞的比例和减少铁沉积来促进脑出血小鼠功能的恢复。而后western blot和免疫荧光结果表明,TSG6可促进小胶质细胞/巨噬细胞的M2表型极化。体外吞噬实验进一步证明,TSG6可增强小胶质细胞吞噬红细胞的能力。最后发现实信号转导和转录激活因子6/生长停滞特异性蛋白6信号通路在TSG6介导的血肿吸收中的关键作用。综上,研究证实TSG6在促进脑出血小鼠模型中血肿吸收的关键作用,表明TSG6通过调节小胶质细胞/巨噬细胞细胞向M2型极化,激活信号转导和转录激活因子6/生长停滞特异性蛋白6信号通路,增加吞噬细胞表面相关吞噬受体表达增强其吞噬红细胞能力,从而加速血肿的清除并改善神经功能。
https://orcid.org/0000-0002-4153-8590 (Zhouping Tang); https://orcid.org/0000-0002-4083-8145 (Gaigai Li)
 #br#
#br#
 #br#
#br#
 #br#
#br#
 #br#
#br#
 #br#
#br#