中国神经再生研究(英文版) ›› 2026, Vol. 21 ›› Issue (8): 3769-3778.doi: 10.4103/NRR.NRR-D-25-00493
多层组学整合分析揭示阿尔茨海默病的潜在治疗靶点
Integrated multi-omics reveal potential therapeutic targets for Alzheimer's disease
Hongli Li1, #, Jin Kang2, #, Zilin Liang3, Xiaowei Wang4, Lemei Zhu1, 5, Hanfen Tang6, *, Weijun Peng1, 5, *
- 1Department of Integrated Traditional Chinese & Western Medicine, The Second Xiangya Hospital, Central South University, Changsha, Hunan Province, China;
2Department of Rheumatology and Immunology, The Second Xiangya Hospital of Central South University, Changsha, Hunan Province, China;
3School of Integrated Chinese and Western Medicine, Hunan University of Chinese Medicine, Changsha, Hunan Province, China;
4Department of Pathology, The Second Xiangya Hospital, Central South University, Changsha, Hunan Province, China;
5Academician Workstation, Changsha Medical University, Changsha, Hunan Province, China;
6Department of Nutrition, Second Xiangya Hospital, Central South University, Changsha, Hunan Province, China
摘要:
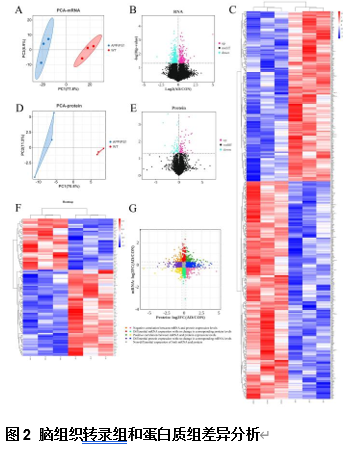
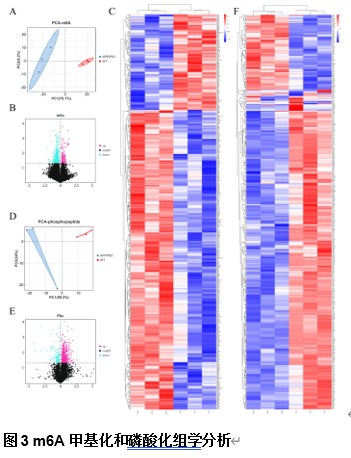
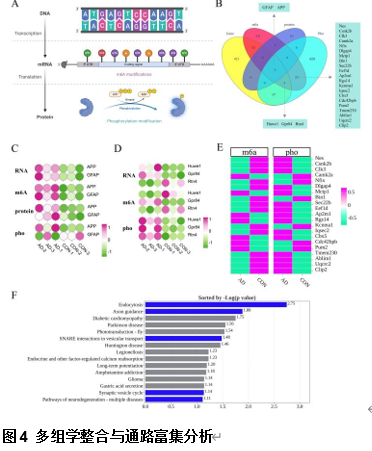
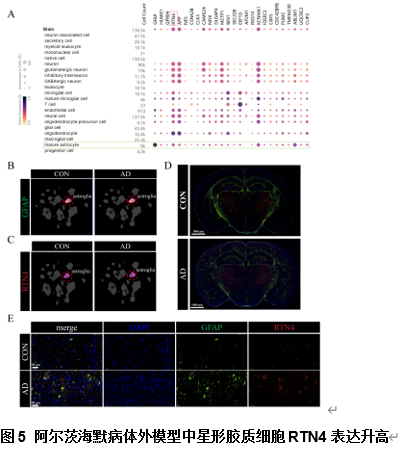
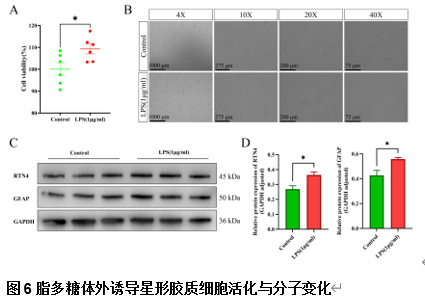
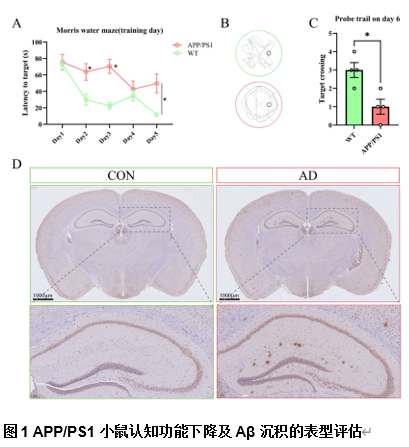
由于阿尔茨海默病发病机制的多因素复杂性,多层组学整合分析对于全面阐明其分子变化至关重要。实验利用成熟的APP/PS1小鼠模型,通过转录组学、蛋白质组学、m6A表观转录组学和磷酸化蛋白质组学分析进行了多层组学整合分析。结果显示,阿尔茨海默病小鼠模型在多个生物学维度上存在实质性的分子变化,以及GFAP、APP和RTN4等关键基因的表达变化。其中反应性星形胶质细胞中RTN4的显著升高表明其参与了阿尔茨海默病的发病机制。实验确定了与内吞作用相关的失调通路,这些通路在疾病进展中发挥了重要的作用。研究还关注了转录后m6A甲基化和翻译后蛋白质磷酸化修饰的重大影响,但这些修饰在阿尔茨海默病相关研究中的代表性不足。此次实验的重要贡献在于提出了采取多层组学整合方法来揭示阿尔茨海默病复杂的生物学变化很有必要,新的见解确定了潜在的治疗靶点,如RTN4。
https://orcid.org/0000-0002-4506-0942 (Weijun Peng); https://orcid.org/0000-0001-8104-5101 (Hanfen Tang)
 #br#
#br#