中国神经再生研究(英文版) ›› 2026, Vol. 21 ›› Issue (5): 2021-2030.doi: 10.4103/NRR.NRR-D-24-00676
血浆外泌体Lnc_011797促进铁死亡加重脑白质病变
Lnc_011797 promotes ferroptosis and aggravates white matter lesions
Xiang Xu1, #, Yu Sun2, #, Xiaoyan Zhu3 , Shiyin Ma2 , Jin Wei3 , Chang He3 , Jing Chen4, *, Xudong Pan2, *
- 1 Department of Neurology, Qingdao Central Hospital, University of Health and Rehabilitation Sciences (Qingdao Central Hospital), Qingdao, Shandong Province, China; 2 Department of Neurology, The Affiliated Hospital of Qingdao University, Qingdao, Shandong Province, China; 3 Department of Critical Care Medicine, The Affiliated Hospital of Qingdao University, Qingdao, Shandong Province, China; 4 Department of General Practice, Qingdao Central Hospital, University of Health and Rehabilitation Sciences (Qingdao Central Hospital), Qingdao, Shandong Province, China
摘要:
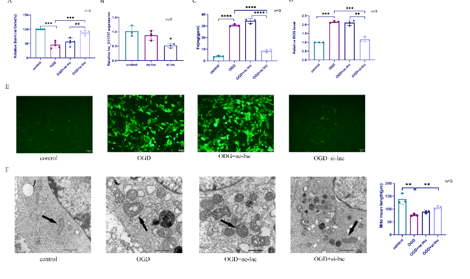
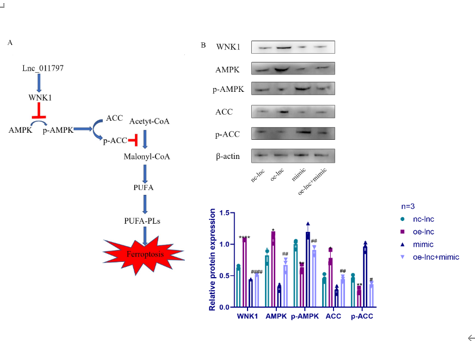
最近有证据表明铁死亡在白质病变的发生发展中起着重要作用。然而,白质病变中铁死亡的机制和调控通路尚不清楚。已有研究发现,长链非编码RNA可通过多种方式影响白质病变的发生和发展。为探索lnc_011797在调控白质病变的机制,实验通过氧糖剥夺人脐静脉内皮细胞来模拟白质病变,继而以lnc_011797过表达或敲减慢病毒影响细胞内lnc_011797表达。结果显示,lnc_011797可促进细胞铁死亡,导致白质病变的发生。此外,lnc_011797可作为miR-193b-3p的竞争性内源性RNA,调控WNK1及其下游铁死亡相关蛋白的表达。而后以双侧颈总动脉狭窄方法建立白质病变小鼠模型,验证了lnc_011797在体内的作用。该结果提示,lnc_011797可通过WNK1调控铁死亡,促进白质病变形成。该结果有助于阐明长链非编码RNA在白质损伤中的调控机制,为治疗脑白质损伤提供新的靶点和策略。
https://orcid.org/0009-0008-7556-6697 (Jing Chen); https://orcid.org/0009-0009-1010-9142 (Xudong Pan)