中国神经再生研究(英文版) ›› 2026, Vol. 21 ›› Issue (7): 3156-3162.doi: 10.4103/NRR.NRR-D-24-01081
外周免疫在早期肌萎缩侧索硬化轴突功能障碍中的作用:一项基于年龄和性别的分析
Zhuoya Wang1, Wen Cao1, Lu Chen1, Shuo Zhang1, Lu Tang1, Wenjuan Cui1, Mingjun Kong1, Ling Yu1, Dongsheng Fan1, 2, 3, *, Wei Zheng1, *
- 1Department of Neurology, Peking University Third Hospital, Beijing, China;
2Beijing Key Laboratory of Biomarker and Translational Research in Neurodegenerative Diseases, Beijing, China; 3Key Laboratory for Neuroscience, National Health Commission/Ministry of Education, Peking University, Beijing, China
摘要:
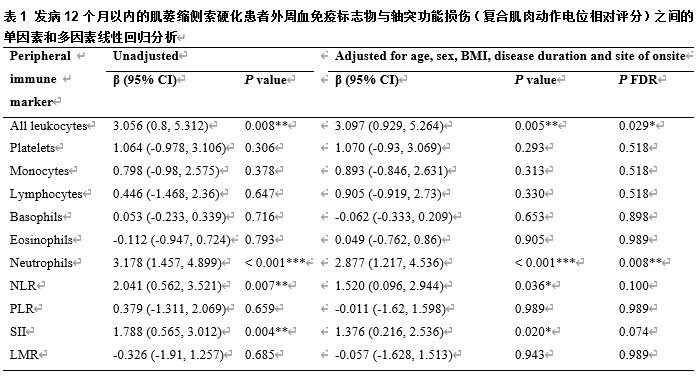
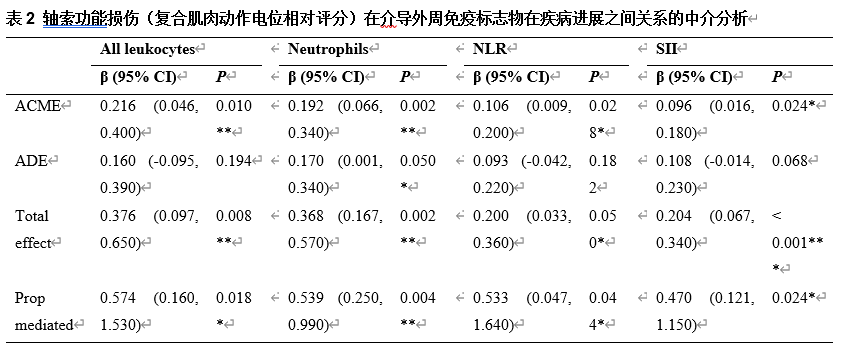
肌萎缩侧索硬化的特征是运动神经元的进行性丧失,且早期轴突功能障碍在疾病过程中起着重要的作用。然而,这种功能障碍背后的分子机制尚不清楚。为了解肌萎缩侧索硬化患者外周免疫失调与轴突功能障碍之间的关系,此次队列研究于2018年1月至2024年5月在北京大学第三医院招募了发病12个月内的372例散发性肌萎缩侧索硬化患者,收集外周血免疫标志物,包括总白细胞、淋巴细胞、单核细胞、中性粒细胞、嗜碱性粒细胞、嗜酸性粒细胞和血小板,并计算4种衍生比值(中性粒细胞与淋巴细胞比值、血小板与淋巴细胞比值、淋巴细胞与单核细胞比值以及全身免疫炎症指数)。调整混杂因素后的多变量分析结果显示,在早期肌萎缩侧索硬化患者中,较高的白细胞计数、中性粒细胞计数以及中性粒细胞相关衍生比值(中性粒细胞与淋巴细胞比值(NLR)和全身免疫炎症指数(SII))与较高的复合肌肉动作电位评分显著相关。进一步分层分析说明,这些关联因年龄和性别而异。此外,中介分析表明,轴突功能障碍在免疫标志物与疾病进展之间的关系中起着重要作用。这些发现提示,外周免疫失调可通过介导周围神经损伤在肌萎缩侧索硬化进展中起重要作用,特别是在疾病的早期阶段。
https://orcid.org/0000-0002-3129-9821 (Dongsheng Fan); https://orcid.org/0009-0008-0445-716X (Wei Zheng)